

Ideas and Innovation - Year 2012 - Volume 27 -
Practical training model for microvascular anastomosis
Modelo prático para treinamento de anastomose microvascular
ABSTRACT
The microsurgical anastomosis technique is practically challenging and requires extensive training, dedication, and time for inexperienced surgeons to perfect. In the present report, we describe an accessible, practical, and simple training model, which involves the use of an abdominal flap obtained from abdominoplasties. The caliber of the superficial epigastric vessels found in the excised abdominal flaps ranged from 1.2 mm to 2.0 mm, depending on the preoperative body mass index of the patient. This flap facilitated the training of microsurgical anastomosis in blood vessels with different calibers. These vessels had a small amount of remnant blood within the lumen, which enabled the testing of the quality and patency of the anastomosis. This training model involving human abdominal vessels allows for a quicker transition to actual operative situations, compared to the use of animal or inanimate models. Moreover, effective practice of dissection and endtoend and endtoside anastomoses using our model helps in the development of skill and surgical dexterity.
Keywords: Anastomosis, surgical. Microsurgery. Abdomen/surgery. Training.
RESUMO
A técnica de anastomose microcirúrgica é desafiadora e requer treinamento extenso, dedicação e tempo. Os autores descrevem um modelo de treinamento acessível, prático e fácil, que utiliza retalho abdominal proveniente de abdominoplastias. O calibre dos vasos epigástricos superficiais encontrados nos retalhos abdominais excisados variou de 1,2 mm a 2 mm, dependendo do índice de massa corporal da paciente no préoperatório. Esse retalho permitiu o treinamento de anastomoses microcirúrgicas em vasos de diferentes calibres. Esses vasos permaneciam com pequena quantidade de sangue em seu lúmen, o que permitia testar a qualidade e a patência das anastomoses. Esse modelo de treinamento em vasos abdominais humanos, quando comparado aos modelos animais ou inanimados, permite transição mais rápida e real aos pacientes. A prática de dissecção e de anastomoses terminoterminais e terminolaterais de uma maneira efetiva e prática aperfeiçoa a destreza cirúrgica.
Palavras-chave: Anastomose cirúrgica. Microcirurgia. Abdome/cirurgia. Capacitação.
Microvascular anastomosis is a challenging technique that requires extensive training, dedication, time, and effort to be mastered. Microsurgery is expanding the surgical treatment options for cancer, trauma, and aesthetic plastic surgery, and it is therefore imperative that trainees be well prepared for complicated reconstructive interventions in the future1.
Young surgeons have few opportunities to gain experience in the microvascular anastomosis technique by practicing on actual patients. Therefore, several experimental animals (rats, mice, and swine), in which the size and consistency of vessels is similar to that of humans, have been used for many years for training purposes. However, the stringent laws of the Institutional Animal Care and Use Committees have triggered the need to find alternatives. Artificial materials (such as silicone tubes and surgical gloves), roboticassisted methods, interactive simulators, and polyvinyl alcohol hydrogel models have been helpful for training in the basic skills of microvascular anastomosis2,3. However, each of these modalities has room for improvement in terms of accessibility, repeatability, and cost effectiveness.
THE PROPOSED MODEL
We describe an accessible and readily available model for microsurgical training using human subcutaneous tissue obtained from abdominal dermolipectomies of patients undergoing abdominoplasty or panniculectomy4. Surgery is performed through a suprapubic incision by which the superficial epigastric vessels are identified on the surface of both sides of the flap, carefully dissected near their origin, piercing the deep layer of the fascia of Scarpa above the inguinal ligament, and then sectioned (Figures 1 and 2).

Figure 1 - Dissection of the right superficial inferior epigastric artery indicating the length of the pedicle.

Figure 2 - Dissection of the perforators of the rectus abdominal muscle.
The excess abdominal flap is undermined as usual, removed, and placed on a table outside the surgical field, facing upward and exposing the vessels. It is then split into 2 halves and juxtaposed under the microscope to perform different anastomosis (end-to-end, end-to-side) and suture techniques, using the appropriate microsurgical instruments and materials (Figures 3 and 4).
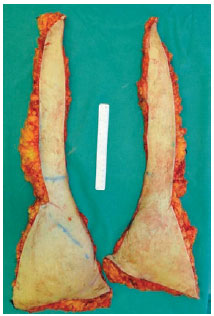
Figure 3 - The 2 halves of the abdominal flap.
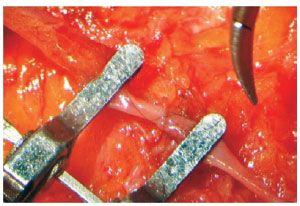
Figure 4 - Close-up view of an end-to-end anastomosis of the superficial inferior epigastric artery from both sides.
The caliber of the inferior superficial epigastric arteries ranged from 1.2 mm to 2.0 mm, depending on the pre operative body mass index (BMI) of the patients, (a higher BMI was correlated with a greater diameter of the vessels). This model allows the surgeon to practice the techniques of anastomosis in vessels of different caliber, simulating the real situation in the operating room. This microsurgical flap model also enabled dissection of vascular pedicles (including the perforators). In addition, these natural flaps retained a certain amount of blood in their lumen even after resection, which enabled the assessment of the patency and quality of the anastomosis created.
The technical skills of our trainees in plastic surgery, general surgery, and orthopedic surgery can be tested using the abdominal flap model. This model enables testing of surgical dexterity and the overall timing of the performance of different types of anastomosis on tissues similar to the ones encountered in patients without the added cost of expansive models or animals. In addition, the trainees found the abdominal flaps to be a much more realistic simulation of arterial microvessel repair. They also showed increased confidence transitioning from this natural flap model to the actual patient, allowing them to perform dissections, sections, and anastomoses in an effective and timely manner.
REFERENCES
1. Cigna E, Bistoni G, Trignano E, Tortorelli G, Spalvieri C, Scuderi N. Microsurgical teaching: our experience. J Plast Reconstr Aesthet Surg. 2010;63(6):e529-31.
2. Kligman BE, Haddock NT, Garfein ES, Levine JP. Microsurgery trainer with quantitative feedback: a novel training tool for microvascular anastomosis and suggested training exercise. Plast Reconstr Surg. 2010;126(6):328-30e.
3. Mutoh T, Ishikawa T, Ono H, Yasui N.A new polyvinyl alcohol hydrogel vascular model (KEZLEX) for microvascular anastomosis training. Surg Neurol Int. 2010;1:74.
4. Esteban D, Fraga MF, Shimba LG, Kikuchi W, Junior AH. Basic plastic surgery training using human skin. Plast Reconstr Surg. 2009;123(2):90-2e.
1. Doctor in medicine from the Faculdade de Ciências Médicas da Santa Casa de São Paulo (Faculty of Medical Sciences of Santa Casa de São Paulo) - FCMSCSP, Associate Professor of FCMSCSP, São Paulo, SP, Brazil.
2. Master in medicine from FCMSCSP, Associate Professor of FCMSCSP, São Paulo, SP, Brazil.
3. Specialist member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery) - SBCP, Plastic surgeon at the Universidade de Maryland, Baltimore, USA.
4. Plastic surgeon, specialist member of SBCP, Associate Physician of Plastic surgery at FCMSCSP, São Paulo, SP, Brazil.
5. Doctor in medicine from the FCMSCSP, Chief of plastic surgery at FCMSCSP, São Paulo, SP, Brazil.
Correspondence to:
Murillo Fraga
Av. Lineu de Paula Machado, 738 - Cidade Jardim
São Paulo, SP, Brazil - CEP 05601-000
E-mail: murifraga@ig.com.br
Article submitted to SGP (Sistema de Gestão de Publicações/ Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: March 29, 2012
Article accepted: April 21, 2012
Study performed at the Faculdade de Ciências Médicas da Santa Casa de São Paulo (Faculty of Medical Sciences of Santa Casa de São Paulo), São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter