

Original Article - Year 2012 - Volume 27 -
Results of resection of infantile nasal hemangiomas in the profiferative phase: a safe approach for central face tumors
Resultados da ressecção de hemangiomas infantis nasais em fase proliferativa: abordagem segura para os tumores centrais da face
ABSTRACT
BACKGROUND: Infantile hemangioma is the most common benign tumor in infancy and occurs most often in the cervicofacial region. Its course can be divided into 3 phases with frequent spontaneous regression. However, residual sequelae or anatomical structure deformities can occur. An early and definitive surgical approach aiming at good aesthetic results and anatomical preservation is indicated in such cases because of the localization of the nasal hemangiomas and their capacity to disfigure. This study analyzed the results of the definitive surgical approach for proliferative nasal hemangiomas according to an objective evaluation.
METHODS: From 1997 to 2009, 20 patients suffering from nasal hemangiomas in the proliferative phase were treated surgically. The lesions were analysed according to the area affected and type of treatment. Complication rates and the need for additional procedures were analyzed. The aesthetic results were evaluated by independent evaluators.
RESULTS: The lesions were localized in the tip of the nose in 50% of patients, dorsal area in 20%, all subunits in 15%, paranasal areas in 10%, and alar area in 5%. Resection was total and subtotal in 60% and 40% of the patients, respectively. The mean follow-up period was 42.6 months. The mean number of surgical procedures per patient was 1.3 ± 0.7. No significant complications were observed. The results were positively evaluated with respect to the reduction of lesion volume and improved face shape, corroborating the proposed approach.
CONCLUSIONS: Definitive surgical treatment is a safe and effective alternative for the management of nasal hemangiomas and has low complication rates.
Keywords: Hemangioma. Nose. Child. Plastic surgery. Outcome assessment.
RESUMO
INTRODUÇÃO: O hemangioma infantil é o tumor benigno mais comum da infância, predominando na região cervicofacial. É caracterizado por apresentar 3 fases distintas, observando-se frequentemente regressão espontânea dessas lesões. No entanto, sequelas residuais ou deformidades das estruturas anatômicas em crescimento podem ocorrer. A abordagem cirúrgica precoce e definitiva é indicada, em decorrência da localização dos hemangiomas nasais e seu potencial desfigurante, visando à obtenção de bons resultados estéticos e preservação anatômica. O objetivo do presente estudo foi analisar os resultados da abordagem cirúrgica definitiva para hemangiomas proliferativos nasais, com base em uma avaliação objetiva.
MÉTODO: No período de 1997 a 2009, 20 pacientes portadores de hemangiomas nasais em fase proliferativa foram submetidos a tratamento cirúrgico. As lesões foram avaliadas segundo local de acometimento e tratamento realizado. Foram analisados índices de complicações e necessidade de procedimentos adicionais. Os resultados estéticos foram avaliados por avaliadores independentes.
RESULTADOS: As lesões estavam localizadas na ponta nasal em 50% dos pacientes; no dorso, em 20%; em todas as subunidades, em 15%; nas áreas paranasais, em 10%; e na unidade alar, em 5%. A ressecção foi total em 60% dos pacientes e subtotal em 40%. O período médio de acompanhamento foi de 42,6 meses. A média de procedimentos cirúrgicos por paciente foi de 1,3 ± 0,7. Nenhuma complicação importante foi observada. Os resultados foram positivamente avaliados quanto a redução do volume da lesão e melhora do contorno facial, corroborando a conduta proposta.
CONCLUSÕES: No manejo dos hemangiomas nasais, o tratamento cirúrgico definitivo pode ser considerado uma alternativa segura e eficaz, com baixas taxas de complicação.
Palavras-chave: Hemangioma. Nariz. Criança. Cirurgia plástica. Avaliação de resultados.
Infantile hemangioma is the most common vascular tumor in childhood. It is a true neoplastic disorder composed of endothelial cells: it proliferates after birth, growing via hyperplasia and cellular hypertrophy, with microscopic characteristics of neoplastic tissues and it is easily cultured in in vitro cultures1-3. Infantile hemangioma is characterized by rapid proliferation in the first months of life, followed by spontaneous involution4.
The natural history of hemangioma is divided into 3 well-characterized phases: initial growth (proliferative), spontaneous regression (involution), and the final involuted phase4,5. The clinical presentation varies depending on the evolution phase. Infantile hemangioma typically appears between birth and the 8th week of life. Precursor marks such as spots or telangiectasias are frequent6. The proliferative phase is more pronounced during the first 3 to 6 months of life, peaking around 12 months. After that, the hemangioma begins a phase of spontaneous regression or involution characterized by increased cell apoptosis and progressive replacement of neoplastic tissue with stromal fibroadipose tissue2,3,6-9.
The incidence of hemangioma in Caucasians ranges from 4% to 12%2,3; it is 3 times more frequent in female than male patients and is more common in premature and lowweighted newborns10. Anatomical distribution is variable, but hemangiomas occur in the head and neck region in approximately 60% of cases. Patients with infantile hemangioma in the cervicofacial region frequently have psychosocial disturbances due to associated aesthetic and functional deformities2,6,11,12.
Approximately 50% of infantile hemangiomas are completely involuted by 5 years of age. The potential for spontaneous involution makes active treatment of non life threatening lesions controversial. The side effects of current medical therapy and definitive deformities caused by surgical treatment must be critically considered before indicating active treatment1,3,4,6,13 .
Treatment options for infantile hemangiomas include conservative (expectant) treatment, pharmacological or surgical treatment.
Active treatment during the proliferative phase is indicated more often for hemangiomas in the center of the face, particularly nasal hemangiomas, due to the localization and potential for disfiguration. The particular complications of such locations, includes risks of airway obstruction, bleeding, ulceration, pain, and potential deformity of the nasal cartilages as well as psychological problems both for the child and family members, prompting an early and definitive surgical approach2,3,8,14.
Several studies also indicate that the rate of spontaneous resolution of nasal hemangiomas is relatively lower even after the end of proliferative phase. This is because of the presence of fibro-adipose tissue deposition and remaining excess skin, which may deform facial shape, contributing to unsatisfactory results frequently produced by these lesions3,9,15.
In this sense, proliferative nasal hemangiomas are particularly challenging because of need for short-term resolution, counteracting with the risk of adverse effects caused by early treatment. In this context, surgical treatment for nasal proliferative hemangiomas must be indicated and evaluated through objective criteria aiming at satisfactory outcomes, including the removal of affected tissues and preservation of anatomy.
Therefore, in the present study, we analyzed the results of the definitive surgical approach for nasal proliferative hemangiomas according to an objective evaluation.
METHOD
From 1997 to 2009, 20 patients being 16 females with nasal infantile hemangiomas were treated during the proliferative phase by the same surgeon. The mean age of the patients at the time of surgery was 41 months. The present study was approved by the ethics committee of our institution.
The lesions were evaluated according to anatomical localization (according to the aesthetic subunits of the nose). Infantile hemangioma was classified as superficial (only the skin was affected), deep (only affected subcutaneously), or mixed, depending on depth (Figures 1 to 4).

Figure 1 - Mixed infantile hemangioma in the proliferative phase affecting the entire nose. The patient underwent primary resection and 2 additional procedures. Evolution over 5 years.

Figure 2 - Deep infantile hemangioma in the proliferative phase affecting the tip of the nose. Transcolumellar access and 8-year evolution after a single procedure.

Figure 3 - Deep infantile hemangioma in the proliferative phase affecting the tip of the nose. Transcolumellar access and 13-year evolution after a single procedure.
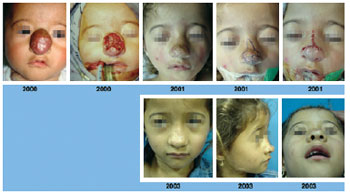
Figure 4 - Superficial infantile hemangioma in the proliferative phase affecting the tip and dorsal sides of the nose. Transtumoral access and evolution after 3 years and 2 surgical procedures. The beginning of conservative cutaneous resection is highlighted.
From a technical perspective, surgical planning aimed at definitive treatment. The surgical access were transtumoral direct approach (Figures 1 and 4), with incisions on the hemangioma or its limits, or a transcolumellar approach, similar to that used in open rhinoplasty (Figures 2, 3, and 5). In deep hemangiomas, the approach was always transcolumellar, but it varied in superficial and mixed cases.
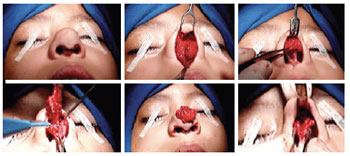
Figure 5 - Transcolumellar access, resection, and simultaneous reconstruction of anatomical structures.
Non affected skin was not removed in any case, even when there was excess after resection of the hemangioma. Resection was classified as partial (up to 50% of the lesion), subtotal (50-90% of the lesion), or total (91-100% of the lesion).
To evaluate the results and validate the indications, we considered the complication rates, need for additional procedures, and aesthetic evaluation.
To evaluate the aesthetic results, pre-and postoperative photos were analyzed by 3 plastic surgeons not involved in the treatment of the patients. Shape, lesion volume, scar quality, general appearance of the face, and functional involvement were scored from 1 to 4 as follows: 1, worsening; 2, small improvement; 3, improvement; 4, large improvement.
The statistical package STATA (Stata Statistical Software: version 10.0, StataCorp LP, College Station, TX, USA) was used for statistical analysis.
The distribution of frequencies was used to describe categorical variables (i.e., gender, ethnicity, localization, treatment categories, and evaluations), and the measures of central tendency (mean and median) and variability (minimum and maximum values, and standard deviations) were used for numerical variables (i.e., age and follow-up). The χ2 frequency test was used to verify independence between scores from different evaluators concerning each item, and the kappa concordance index was used to verify the degree of concordance between evaluators. The level of significance was set at 5% for all statistical tests.
RESULTS
The lesions were located in the nose tip in 10 (50%) cases, in the dorsum in 4 (20%), paranasal areas in 2 (10%), and ala in 1 (5%). In 3 (15%) patients, the entire nose was affected (Figure 6). Nine (45%) hemangiomas were deep, 8 (40%) were mixed, and 3 (15%) were superficial (Figure 7).
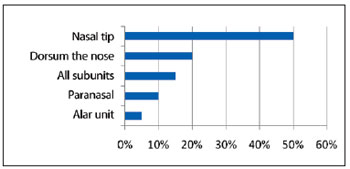
Figure 6 - Lesion incidence by nasal unit
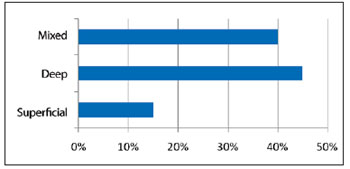
Figure 7 - Lesion incidence by depth.
The transcolumellar and direct approaches were used in 9 (45%) and 11 (55%) cases, respectively. Total and subtotal resection were performed in 12 (60%) and 8 (40%) patients, respectively.
The mean follow-up period was 42.6 months (median 41 months, ranging from 3 months to 8 years).
The mean number of surgical procedures per patient was 1.3 ± 0.7. No postoperative complications compromising the final result were observed.
The 3 evaluators considered the results adequate with respect to the reduction of volume of the lesion and improvement of facial shape; the mean aesthetic score was 3.3. Large improvements were observed in 12 (60%) patients, and moderate improvements were observed in 4 (20%) cases. The results were considered insufficient in 4 (20%), patients for whom additional surgical procedures were indicated (Figure 8).
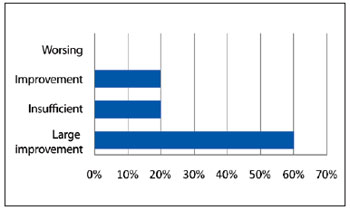
Figure 8 - Evaluation of aesthetic results.
DISCUSSION
Volumetric reduction of facial hemangiomas through surgery can be effective, avoiding several complications including visual and airway obstruction, bleeding ulcerations, and anatomical distortion. Treating infantile nasal hemangiomas early is advantageous because it prevents sequelae resulting from the lesion enlargement, which can compromise growing structures. In contrast with the classical treatment philosophy, which recommends late approach, we indicate the treatment of patients with hemangiomas still in the proliferative phase in order to achieve the proposed aims.
However, the long-term predictability of the results is very limited since the patient is still developing and growing during treatment. It is important to consider surgical planning and measures aiming to reduce the potential for negative side effects.
Resection of healthy skin is formally contraindicated. In extensive lesions with superficial involvement, partial resections may be indicated, which can also avoid growth restrictions due to a lack of tissue malleability. Conservative planning in skin resection helps maintain enough tissue for nasal growth and simultaneously prevents the hemangioma from compressing growing structures. The fast skin adaptation characteristic of infancy allows the maintenance of excessive normal tissue as evidenced by the favorable results obtained in the present study, validating the criteria of surgical indication. The findings of the long-term evaluation also corroborate this, showing such results with only 1 or 2 surgical procedures.
Evaluation by independent examiners appears to be the best way to objectively analyze the sample. Some questions such as the difficulty of the case and need for reoperation must be answered by independent plastic surgeons with evaluation philosophies and sufficiently high standards who lack the bias of the primary surgeon on the case. Nevertheless, it is important that there is a consensus among the evaluators so that the results will be relevant from a statistical perspective. In other words, when the precise indications and criteria are followed, surgical removal of Figure 8 - Evaluation of aesthetic results. hemangiomas provides favorable results for both simple and complicated cases.
When weighing the risks and benefits of surgical treatment and considering the scar at the location of the hemangioma, the present study support the current treatment philosophy. The proportion of cases evaluated and considered as not requiring new procedures or requiring small simple procedures indicates that the treatment aims were achieved. Early surgery has established itself as a treatment option and is being performed more often.
CONCLUSIONS
For the management of nasal hemangiomas, definitive surgical treatment is a safe and effective option with low complication rates.
REFERENCES
1. Beck DO, Gosain AK. The presentation and management of hemangiomas. Plast Reconstr Surg. 2009;123(6):181e-91e.
2. Boye E, Jinnin M, Olsen BR. Infantile hemangioma: challenges, new insights, and therapeutic promise. J Craniofac Surg. 2009;20(Suppl 1):678-84.
3. Goldenberg DC. Estudo crítico dos resultados obtidos no tratamento dos hemangiomas cutâneos cérvico-faciais [Dissertação de mestrado]. São Paulo: Faculdade de Medicina da Universidade de São Paulo; 2002.164p.
4. Bruckner AL, Frieden IJ. Hemangiomas of infancy. J Am Acad Dermatol. 2003;48(4):477-93.
5. Achauer BM, Chang CJ, Vander Kam VM. Management of hemangioma of infancy: review of 245 patients. Plast Reconstr Surg. 1997;99(5):1301-8.
6. Brown TJ, Friedman J, Levy ML. The diagnosis and treatment of common birthmarks. Clin Plast Surg. 1998;25(4):509-25.
7. Chen MT, Yeong ER, Horng SY. Intralesional corticosteroid therapy in proliferating head and neck hemangiomas: a review of 155 cases. J Pediatr Surg. 2000;35(3):420-3.
8. Frieden IJ, Eichenfield LF, Esterly NB, Geronemus R, Mallory SB. Guidelines of care for hemangiomas of infancy. J Am Acad Dermatol. 1997;37(4):631-7.
9. Hiraki PY, Goldenberg DC. Diagnóstico e tratamento do hemangioma infantil. Rev Bras Cir Plást. 2010;25(2):388-97.
10. Tanner JL, Dechert MP, Frieden IJ. Growing up with a facial hemangioma: parent and child coping and adaptation. Pediatrics. 1998;101(3 pt 1):446-52.
11. Bauland CG, van Steensel MA, Steijlen PM, Rieu PN, Spauwen PH. The pathogenesis of hemangiomas: a review. Plast Reconstr Surg. 2006;117(2):29e-35e.
12. Schiavon G, Fusaro F, Carmignola G, Zoppellaro F, Marzaro M, Bastasin F, et al. Considerazioni sulla terapia steroidea degli emangiomi infantili. Minerva Pediatr. 1998;50(4):105-9.
13. Léauté-Labrèze C, Taïeb A. Effecacité des bêtabloquants dans les hémangiomes capillaires infantiles: signification physiopathologique et conséquences thérapeutiques. Ann Dermatol Venereol. 2008;135(12):860-2.
14. Enjolras O, Deffrennes D, Borsik M, Diner P, Laurian C. Vascular tumors and the rules of their surgical management. Ann Chir Plast Esthet. 1998;43(4):455-89.
15. McCarthy JG, Borud LJ, Schreiber JS. Hemangiomas of the nasal tip. Plast Reconstr Surg. 2002;109(1):31-40.
1. Associate Professor of Plastic Surgery at the Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo (Faculty of Medicine of the University of São Paulo - FMUSP), full member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery) - SBCP, São Paulo, SP, Brazil.
2. Resident physician of Plastic Surgery at the Plastic Surgery Unit of FMUSP, member in training of SBCP, São Paulo, SP, Brazil.
3. Specialist in Plastic Surgery, collaborator physician of the Plastic Surgery Unit at FMUSP, Associate member of SBCP, São Paulo, SP, Brazil.
4. Master in Plastic Surgery, collaborator physician of the Plastic Surgery Unit at FMUSP, associate member of SBCP, São Paulo, SP, Brazil.
5. Professor of Plastic Surgery at the FMUSP, full member of SBCP, São Paulo, SP, Brazil.
Correspondence to:
Dov Charles Goldenberg
Rua Pedro de Toledo, 960 - cj. 124 - Vila Mariana
São Paulo, SP, Brazil - CEP 04039-002
E-mail: drdov@terra.com.br
Article submitted to SGP (Sistema de Gestão de Publicações/ Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: January 27, 2012
Article accepted: May 26, 2012
Work conducted at the Plastic Surgery Unit, Faculdade de Medicina da Universidade de São Paulo (Faculty of Medicine, University of São Paulo), São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter