

Original Article - Year 2012 - Volume 27 -
The effectiveness of the adipofascial retroauricular flap in otoplasty
Eficácia do retalho adipofascial retroauricular em otoplastia
ABSTRACT
BACKGROUND: Otoplasty techniques using permanent sutures to mold cartilage have high rates of incidence of granulomatous issues (up to 22%) during the postoperative period. In order to control these complications, Horlock et al. developed the adipofascial retroauricular flap in 2001. In the present study, we compared patients who underwent otoplasty with or without the use of this flap. We aimed to quantify the incidence of complications affecting the sutures, evaluate the efficacy of the retroauricular adipofascial flap in the prevention of problems associated with the permanent sutures applied to the auricular cartilage, and analyze if the preparation of this flap increased the rates of other types of complications not related to the sutures, particularly suture dehiscence or defective wound healing during the postoperative period.
METHODS: We consulted the medical records of 24 patients who underwent otoplasties from 2007 to 2009 at the Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (Clinics Hospital of Ribeirão Preto, Faculty of Medicine of the University of São Paulo). The patients were divided into 2 groups, one where a "classic" technique was used (n = 24 ears) and another where the "flap" technique was used (n = 24 ears). Patients were reviewed in a retrospective manner and had a minimal postoperative period of 24 months.
RESULTS: The total incidence of important complications related to the sutures was zero in the "flap" group and 20.8% in the "classic" group. Edema was more pronounced in the first 14 postoperative days, and was identified in 41.6% of patients in the flap group versus 12.5% of the patients in the classic group. The technical steps involved in preparing the adipofascial retroauricular flap did not increase surgical time. It is possible that the retroauricular tissue covering the sutures (which is stronger) and the tissue connecting these sutures (preserved subcutaneous tissues) were the main factors responsible for the significant decrease in granulomatous complications after otoplasties in which the flap was used.
CONCLUSIONS: After the retroauricular adipofascial flap was introduced, the incidence of complications was reduced to zero, demonstrating the efficacy of this flap in the prevention of complications related to granulomas. This benefit was obtained without an increase in the incidence of other complications or surgical time. This study suggests that the retroauricular adipofascial flap may be an important addendum to the technical tools used during otoplasty.
Keywords: Ear, external/surgery. Otologic surgical procedures/methods. Surgical flaps.
RESUMO
INTRODUÇÃO: As técnicas de otoplastia que utilizam suturas permanentes para moldar as cartilagens apresentam elevada taxa (de até 22%) de incidência de problemas granulomatosos no pós-operatório. Para controlar essas complicações, Horlock et al., em 2001, desenvolveram o retalho adipofascial retroauricular. Neste trabalho, são comparados pacientes submetidos a otoplastia com e sem o uso desse retalho, com o objetivo de identificar a incidência de complicações com suturas, avaliar a eficácia do retalho adipofascial retroauricular na prevenção dos problemas com as suturas definitivas aplicadas na cartilagem auricular, e analisar se a confecção desse retalho envolveu aumento da taxa de outras complicações de otoplastia que não as relacionadas às suturas, especialmente deiscência de suturas ou má cicatrização no pós-operatório.
MÉTODO: Foram revisados os prontuários de 24 pacientes submetidos a otoplastias realizadas no período de 2007 a 2009 no Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (Ribeirão Preto, SP, Brasil). Os pacientes foram divididos em 2 grupos, um deles compreendendo a técnica "clássica" (grupo clássico, n = 24 orelhas) e o outro, a técnica de "retalho" (grupo retalho, n = 24 orelhas), de forma retrospectiva e com tempo pós-operatório mínimo de 24 meses.
RESULTADOS: A incidência total de complicações graves de suturas foi de zero no grupo retalho e de 20,8% no grupo clássico. Edema mais acentuado nos primeiros 14 dias de pós-operatório também foi identificado no grupo retalho em 41,6% dos pacientes vs. 12,5% dos pacientes do grupo clássico. O passo técnico de confecção do retalho adipofascial retroauricular não aumentou o tempo cirúrgico. É plausível que a cobertura tecidual retroauricular mais robusta sobre as suturas e o fato de que o tecido em contato com essas suturas seja subcutâneo íntegro sejam os possíveis fatores responsáveis pela queda da incidência de problemas granulomatosos significativos nas otoplastias em que se utilizou o retalho.
CONCLUSÕES: Após a introdução do retalho adipofascial retroauricular, a incidência de complicações relacionadas a granulomas caiu para zero, demonstrando a eficácia do retalho na prevenção dessas complicações. Esse benefício foi obtido sem que houvesse aumento da incidência de outras complicações ou do tempo cirúrgico. Este estudo sugere que o retalho adipofascial retroauricular pode ser uma importante adição ao armamentário técnico dispensado à otoplastia.
Palavras-chave: Orelha externa/cirurgia. Retalhos cirúrgicos. Procedimentos cirúrgicos otológicos.
Auricular prominence (dumbo ears) is the most common congenital deformity affecting the auricular pavilion. It affects about 5% of the population and is more common in Caucasians. The exact cause of auricular prominence is not clear, and it may be related to postural differences in newborns, muscle hypotonia, structural alterations of collagen, or genetic disease; it can also display familial incidence with dominant autosomal genetic patterns1-5.
Auricular prominence is defined by an angle of at least 40° between the auricular pavilion and the plane of the mastoid or when the external border of the helix is at least 21 mm away from this plane1-4. It is mainly caused by anti-helix malformation, protrusion and pronounced development of the concha, or a combination of both. Additional factors may contribute to auricular protrusion, such as prominence of the structures of the lower pole of the ear (the tail of the helix, the concha cavum, and the lobe), a more pointed upper pole, third cross, and pronounced Darwin's tubercle, amongst other deformities1,5,6.
The first description of otoplasty is credited to Dieffenbach7, who performed the excision of skin and conchalmastoid fixation, joining the perichondrium from the concha to the galea, in a case of auricular trauma in 1845. In 1881, Ely8 added the idea of resecting the conchal cartilage when performing the excision of skin and conchal-mastoid fixation. Several authors improved otoplasty in the following years, and in 1910, Luckett9 introduced the important concept of anti-helix restoration. For this, he used the technique of incision of the cartilage along the margins of the new anti-helix and horizontal sutures for repair. In 1952, Becker10 observed the importance of creating a conical anti-helix and his technique of multiple incisions and sutures of the cartilage was improved by Converse & Wood-Smith11 in 1963. In the same year, Mustarde12 described his technique for repair of the anti-helix, applying a series of conchal-scaphal sutures with non-absorbable thread. Still in 1963, 2 techniques involving cartilage abrasion to obtain the anti-helix were independently published, the Chongchet technique13 (abrasion with a scalpel) and the Stenström technique14 (abrasion with a scraper). Both techniques were based on the then recent work of Gibson & Davis15, published in 1958, describing the ability of cartilage to fold towards the side opposite to the side that is injured. Furnas16 initiated the popularization of conchalmastoid sutures with the publication of his article in 1968.
Since then, otoplasty has undergone several changes and technical additions, including several tactical variations1. Although there have been changes in the surgical approach, patient satisfaction is frequently emphasized, regardless of the technique used1,5.
The division of otoplasty according to techniques that mold the cartilage with non-absorbable sutures and those that do not includes the main variations of the procedure and maintains active discussion on the incidence of complications specifically related to non-absorbable sutures. There are reports of several types of complications arising from sutures in otoplasty, such as granulomas, extrusion, fistulas, and cartilage laceration with prominence relapse4,17-19.
The combination of technical variations is possible and, by considering the advantages and disadvantages of each one of these variations, the surgeon has several options for an individualized approach in each patient4-6,18,20,21. Among these different possibilities, we preferentially use shaping techniques with non-absorbable sutures, because they allow good and predictable control of the final result. However, these sutures may be associated with specific complications, especially granulomas and thread extrusion4,18. The retroauricular adipofascial flap described by Horlock et al.22 was created to prevent these problems. In the current study, we compared patients who underwent otoplasty with or without this flap. We aimed to identify the incidence of complications with the sutures, evaluate the efficacy of the retroauricular adipofascial flap in preventing the problems caused by the use of permanent sutures applied at the auricular cartilage, and analyze if the preparation of the flap increased the incidence of other otoplasty complications, not related to sutures, particularly dehiscence of the sutures or defective wound healing during the postoperative period. In addition, this study aimed to evaluate whether adding retroauricular adipofascial flap preparation as a technical step significantly increased otoplasty operative time.
METHOD
Sample Characterization
We reviewed the medical records of patients who underwent otoplasty from 2007 to 2009 at the Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (Clinics Hospital of Ribeirão Preto, Faculty of Medicine of the University of São Paulo, Ribeirão Preto, SP, Brazil).
Twenty-four patients were selected according to the inclusion and exclusion criteria presented in Charts 1 and 2, respectively.
The patients were divided into 2 groups, the "flap" group and the "classic" group comprising individuals operated on by using the adipofascial retroauricular flap or without it, respectively. The flap was used by the author from 2008 onwards and, therefore, patients who underwent the operation before this time formed the classic group and those who underwent the operation after this time were included in the flap group.
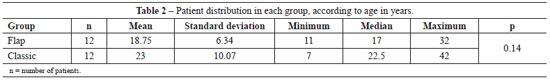
Twenty-four patients were selected, 12 from the classic group and 12 from the flap group, with a total of 24 ears in each group (Tables 1 and 2). The groups did not show statistical differences in gender or age (Student's t test).
Clinical Evaluation Procedures
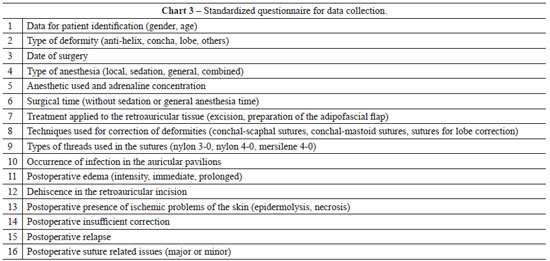
The medical records of all selected patients were evaluated using a specially formulated questionnaire to collect all information relevant to the study (Chart 3).
Since the institutional standard for postoperative follow-up after otoplasty was 12 months at the time these patients were treated and the inclusion criteria predicted a 24-month evaluation, all patients were asked to attend a new consultation for active investigation of points 14-16 of the questionnaire shown in Chart 3.
For additional clarification of item 16 in the questionnaire ("postoperative presence of issues related to the sutures"), we proposed the following definitions for the problems we encountered:
1. Major problems with the sutures were defined as retroauricular alterations in the position of the permanent sutures that showed persistent signs of inflammation (pain, hypersensitivity, hyperemia, hyperthermia), fistulas, or extrusion.
2. Minor problems with the sutures were defined as retroauricular alterations in the position of the permanent sutures with visible fine threads or even palpable knots that were asymptomatic but detectable upon clinical examination.
Surgical Technique
All patients were operated on in a surgical unit within a hospital, with continuous venous access and continuous monitoring of vital signs. For antibiotic prophylaxis, cephazolin was administered 30 min prior to the beginning of surgery, with the dose recommended by the Commission for Control of Institutional Hospital Infection.
In both groups, surgery started with the application of 2% Xylocaine with 1:200.000 epinephrine, independently from simultaneous sedation or general anesthesia. Guidepoints were tattooed using a 21-G needle and sterile 4% methylene blue (Figure 1). A cutaneous incision in the shape of an ellipse was made on the posterior side of the concha. In the patients in the classic group, the epidermis, dermis, and subcutaneous tissue were excised and discarded. The posterior face of the ear was then detached as needed in the plane above the perichondrium. In the patients in the flap group, only the epidermis and dermis were initially excised and detached, similar to the removal of a retroauricular skin flap (Figure 2); these tissues were then discarded. We then proceeded to subdermal detachment of the posterior skin, laterally, until about 3 mm beyond the most lateral guide-points (Figures 3 and 4). After the dermal flap was created, the subcutaneous tissue was longitudinally excised in its most lateral position (Figure 5). The adipofascial flap was then prepared, detaching the tissues superficial to the perichondrium en bloc in a medial direction until the mastoid fascia22 (Figures 6 to 8).
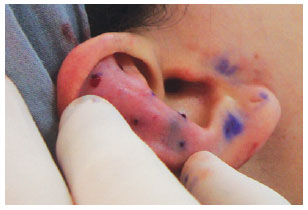
Figure 1 - Guide-points: methylene blue tattoos on the skin and cartilage served to guide accurate application of sutures in the cartilage when shaping the anti-helix and concha.
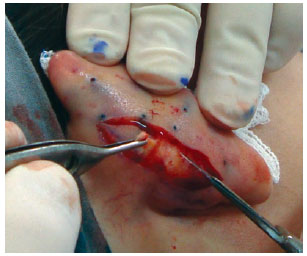
Figure 2 - Limited removal of the retroauricular tissue. Only the epidermis and dermis were incised and detached, as when removing a retroauricular skin graft. The subcutaneous tissue was totally maintained in this region.
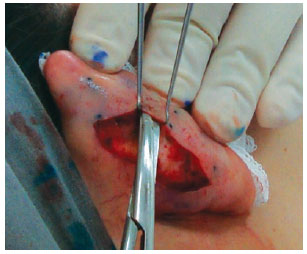
Figure 3 - Subcutaneous detachment for flap preparation.
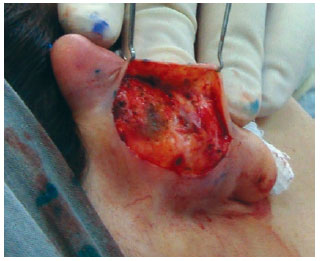
Figure 4 - Complete subcutaneous detachment, surpassing the guide points.
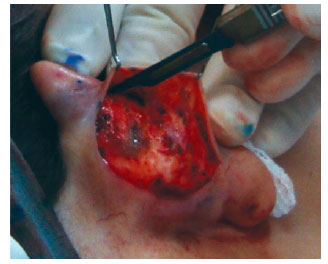
Figure 5 - Incision of the most lateral portion of the flap.
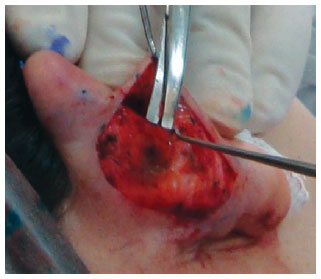
Figure 6 - Detachment of the flap en bloc, above the perichondrium.
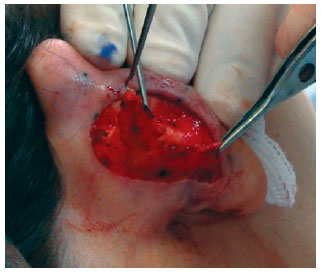
Figure 7 - Flap in its original position.
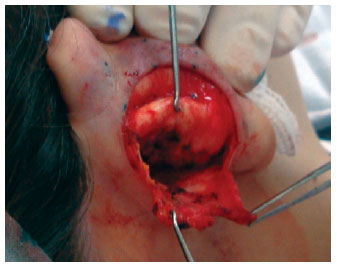
Figure 8 - Reflected flap.
The cartilage corresponding to the new anti-helix was shaped with 2-5 conchal-scaphal "U" sutures using 4-0 colorless nylon6,17. Prominence of the concha was treated with 1-2 "U" sutures by using black 3-0 nylon6,16. In the patients in the flap group, the flap was entirely replaced in its original position and fixed to the cartilage of the scapha with 3-4 simple 4-0 catgut sutures, covering all of the nylon sutures (Figures 9 and 10). Figures 11 and 12 summarize the main steps adopted in each group.
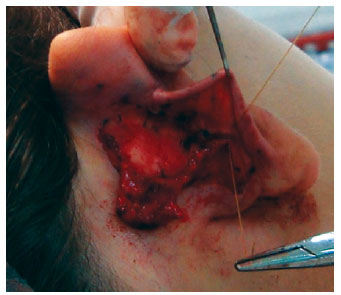
Figure 9 - Fixation of the flap protecting the permanent sutures.
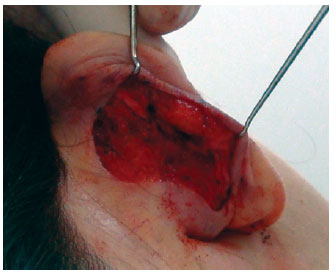
Figure 10 - Conclusion of flap fixation.
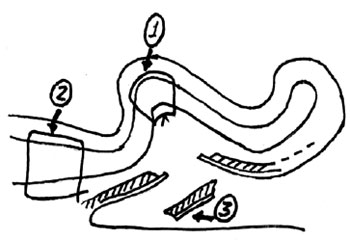
Figure 11 - Summary of the technique used in the classic group. The points indicated by 1, 2, and 3 represent, respectively, anti-helix repair (nylon 4-0), concha-mastoid closure (nylon 3-0), and the retroauricular cutaneous ellipse excision.
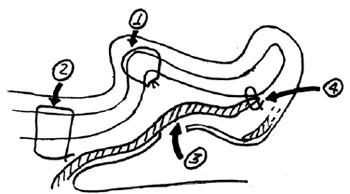
Figure 12 - Summary of the technique used in the flap group. Points indicated by 1, 2, 3, and 4 represent, respectively, anti-helix repair (nylon 4-0), concha-mastoid closure (nylon 3-0), adipofascial retroauricular flap, and the point for fixation of the flap to the cartilage (catgut 4-0).
For both groups, surgery was concluded by closing the retroauricular skin by using continuous 4-0 black nylon sutures, applying neomycin and bacitracin ointment over the incisions, placing gauze and sterile cotton in the anterior face of the ears for retroauricular wound care, and dressing with a wrap.
Variable Characterization and Statistical Methods
The following statistical evaluations were performed for the data obtained from the questionnaire:
1. Comparison of the proportion of ears that had complications with sutures;
2. Comparison of the proportion of ears with other problems; and
3. Comparison of the duration of surgery.
For the first and second evaluations, we used the exact Fisher's test, which is used in data analysis where the aim is to verify the association between 2 categorical variables. This statistical analysis was performed using the procedure PROC FREQ of the software SAS® 9.0.
For the third evaluation described above, we used the Student's t test, in order to compare 2 mean values from unpaired samples. To use this test, it is necessary to test if the 2 groups under comparison have statistically identical variances, and if the data follows an equal distribution. To perform this analysis, the PROC TTEST procedure of the SAS® 9.0 software was used.
RESULTS
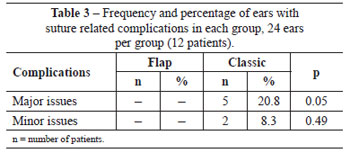
Considering the number of ears in each group, the total incidence of complications related to sutures was zero in the flap group and 29.3% in the classic group (20.8% major issues and 8.3% minor issues). For this comparison, there was a statistical difference between the groups but only for the major complications (Table 3).
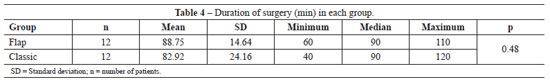
Mean surgical time was 89 min in the flap group and 83 min in the classic group, and this difference was not statistically significant (Table 4).
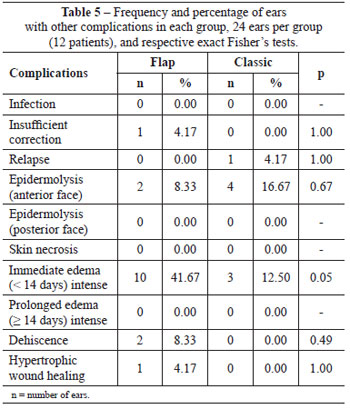
Table 5 shows the incidence of other problems related to otoplasty, with the only significant difference being that the presence of immediate edema was more intense in the flap group.
Figures 13 and 14 illustrate patients from the flap group and Figures 15 and 16 show patients in the classic group.
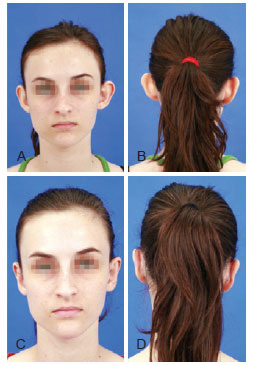
Figure 13 - Patient in the flap group. In A and B, preoperative appearance. In C and D, postoperative appearance.
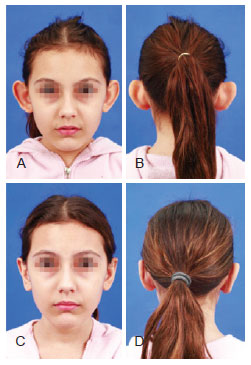
Figure 14 - Patient in the flap group. In A and B, preoperative appearance. In C and D, postoperative appearance.
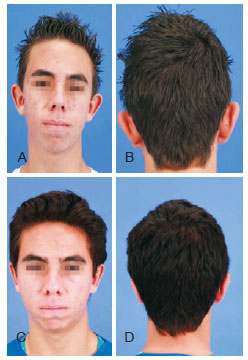
Figure 15 - Patient in the classic group. In A and B, preoperative appearance. In C and D, postoperative appearance.
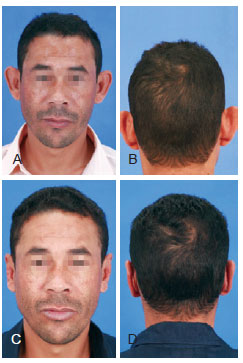
Figure 16 - Patient in the classic group. In A and B, preoperative appearance. In C and D, postoperative appearance.
DISCUSSION
Auricular prominence is a benign medical condition, but affected individuals are frequently the target of bullying in social situations, which may lead to psychological trauma4,5. Constant social stress negatively affects the life of these patients and the psychological benefits of correcting auricular prominence have been highlighted previously23,24. In this context, surgical correction of auricular prominence is one of the few purely aesthetic surgeries whose indication is widely accepted, not only in adults but also in children and adolescents4,6,21.
In the present work, we followed the standardizing suggestions proposed by Limandjaja et al.4, only selecting primary and bilateral cases and assessing the complications of otoplasty statistically by comparing ears as individuals. In this way, it was possible to compare our results with those from other studies.
Despite the fact that granulomatous complications of the sutures in otoplasty are easily identified clinically by plastic surgeons, the lack of clarity in several otoplasty studies concerning what is considered a "granuloma", or even whether "granulomas" are considered among the complications of otoplasty16,24-30, makes it difficult to compare previous findings in this respect. Formation of a foreign body granuloma surrounding the non-absorbable sutures is, from a histopathological point of view, practically inevitable, but there are no otoplasty studies that quantify the granulomatous reaction of suture threads. Considering these difficulties, in this study we chose to avoid nomenclature complications due to the use of the histopathological classification of degrees of granuloma, and instead used the terminology "major" or "minor" issues to describe complications of the sutures.
Conditions that may eventually have required clinical intervention or re-operation were considered as major problems, represented by persistent inflammatory signs (pain, heat, swelling, and redness) over the knot of a non-absorbable suture, and evolution towards fistulas or even extrusion of the suture material. We considered "minor" issues as situations in which there were no complaints from the patient, such as retroauricular threads that were visible through the transparency of the skin or palpable asymptomatic nodules in the position of the knots. In these situations, the possible need for surgical intervention of any kind was much lower.
The mean time until the emergence of symptomatic issues related to sutures after otoplasty is not clearly defined, but some studies report the appearance of complications 2-24 months after surgery; this time also depends on the type of material used18,29,30. For this reason, we included patients with at least 24 months of postoperative follow-up in this study.
The data in the literature reports incidence ranging from 0 to 22.2% for extrusion and symptomatic issues related to permanent sutures in otoplasty4,17,18,19. In the 48 ears analyzed in the current study, the incidence of major problems was 20.8% in the classic group and zero in the flap group; this difference was statistically significant. The rate of symptomatic complications related to cartilage sutures in the classic group was consistent with that found in the literature. We highlight that the absence of these issues in the flap group strongly suggests that the retroauricular adipofascial flap was effective in preventing problems with permanent sutures applied in the auricular cartilage.
The adipofascial retroauricular flap has 2 characteristics that are responsible for its benefits: firstly, it provides a better and stronger tissue cover over the sutures, as previously mentioned by Horlock et al.22. Contrary to the method used by these authors, who resected the most lateral portion of the flap in order to adjust it exactly to its new position in the ear, in the present study, the flap was maintained as a whole, which provided additional retroauricular volume, making available even more tissue for coverage of the sutures. Since we did not observe any additional complaints, this maneuver appeared advantageous. The second benefit of the flap is that it causes the tissue in contact with the suture material to be integrated subcutaneously rather than as cicatricial tissue. It is possible that subcutaneously integrated tissue is less prone to the processes that lead to major problems with sutures in otoplasties.
It is also important to highlight that there was no statistically significant difference between the surgical times of the 2 groups, which indicates that adding preparation of the flap to otoplasty is easily executed and does not influence surgical time.
When considering preparation of the adipofascial retroauricular flap, one should remember that the retroauricular skin that covers the flap becomes very thin. This could eventually cause a higher incidence of dehiscence, necrosis, or problems of cicatricial hypertrophy in the retroauricular skin. However, these complications did not occur significantly in the current study. There was dehiscence in 2 ears in the same patient of the flap group, which was attributed to the fact that the patient insistently scratched the incisions during the early postoperative period, despite our instructions to not do so. With regards to possible problems that the flap may eventually introduce to otoplasty, in this study we observed more extensive immediate edema (< 14 days) in the patients in the flap group, which was possibly associated with surgical trauma in the posterior face of the auricular pavilion.
Controversy relating to the type and diameter of nonabsorbable suture used in otoplasty exists. The most commonly used threads are nylon and mersilene, ranging in diameter from 2-0 to 5-04. There are reports suggesting that nylon is more prone to extrusion4 while mersilene is more prone to granulomatosis31. However, there have been no recent studies specifically designed to compare the use of these 2 threads in otoplasty. Therefore, the choice to use nylon 3-0 and 4-0 in the current study, as well as in other studies similar to this one, is essentially related to preference and surgical experience.
The data from this study are useful as pilot data, and the flap described may be an interesting tool available for otoplasty.
CONCLUSIONS
Throughout the professional life of a plastic surgeon, it is common to learn new techniques and change practices with the aim of improving surgical results, and that is exactly the situation presented in this study.
With regards to the granulomatous issues caused by cartilage sutures with non-absorbable threads in otoplasty, the complication rate before the use of the retroauricular adipofascial flap was 20.8%, which is comparable to that found in the literature. After introduction of the flap, complication rates dropped to 0%, demonstrating the efficacy of the flap in preventing complications related to granulomas. This benefit was obtained without an increase in the incidence of other complications or in surgical time.
REFERENCES
1. Kelley P, Hollier L, Stal S. Otoplasty: evaluation, technique, and review. J Craniofac Surg. 2003;14(5):643-53.
2. Hunter AG, Yotsuyanagi T. The external ear: more attention to detail may aid syndrome diagnosis and contribute answers to embryological questions. Am J Med Genet A. 2005;135(3):237-50.
3. Janis JE, Rohrich RJ, Gutowski KA. Otoplasty. Plast Reconstr Surg. 2005;115(4):60e-72e.
4. Limandjaja GC, Breugem CC, Mink van der Molen AB, Kon M. Complications of otoplasty: a literature review. J Plast Reconstr Aesthet Surg. 2009;62(1):19-27.
5. Caouette-Laberge L, Guay N, Bortoluzzi P, Belleville C. Otoplasty: anterior scoring technique and results in 500 cases. Plast Reconstr Surg. 2000;105(2):504-15.
6. Furnas DW. Otoplasty for prominent ears. Clin Plast Surg. 2002;29(2):273-88.
7. Dieffenbach JE. Die ohrbildung otoplastik. Die operative chirurgie. Leipzig: F.A. Brockhause; 1848. p. 395.
8. Ely ET. An operation for prominence of the auricles. Arch Otolaryngol. 1881;10:97.
9. Luckett WH. A new operation for prominent ears based on the anatomy of the deformity. Surg Gynaecol Obstet. 1910;10:635.
10. Becker OJ. Correction of the protruding deformed ear. Br J Plast Surg. 1952;5(3):187-96.
11. Converse JM, Wood-Smith D. Technical details in the surgical correction of the lop ear deformity. Plast Reconstr Surg. 1963;31:118-28.
12. Mustarde JC. The correction of prominent ears using simple mattress sutures. Br J Plast Surg. 1963;16:170-8.
13. Chongchet V. A method of antihelix reconstruction. Br J Plast Surg. 1963;16:268-72.
14. Stenström SJ. A "natural" technique for correction of congenitally prominent ears. Plast Reconstr Surg. 1963;32:509.
15. Gibson T, Davis W. The distortion of autogenous cartilage grafts: its cause and prevention. Br J Plast Surg. 1958;10:257.
16. Furnas DW. Correction of prominent ears by conchamastoid sutures. Plast Reconstr Surg. 1968;42(3):189-93.
17. Mustardé JC. The treatment of prominent ears by buried mattress sutures: a ten-year survey. Plast Reconstr Surg. 1967;39(4):382-6.
18. Nuara MJ, Mobley SR. Nuances of otoplasty: a comprehensive review of the past 20 years. Facial Plast Surg Clin North Am. 2006;14(2):89-102.
19. Aki F, Sakae E, Cruz DP, Kamakura L, Ferreira MC. Complicações em otoplastia: revisão de 508 casos. Rev Soc Bras Cir Plást. 2006;21(3):140-4.
20. Richards SD, Jebreel A, Capper R. Otoplasty: a review of the surgical techniques. Clin Otolaryngol. 2005;30(1):2-8.
21. Burstein FD. Cartilage-sparing complete otoplasty technique: a 10-year experience in 100 patients. J Craniofac Surg. 2003;14(4):521-5.
22. Horlock N, Misra A, Gault DT. The postauricular fascial flap as an adjunct to Mustardé and Furnas type otoplasty. Plast Reconstr Surg. 2001;108(6):1487-90.
23. Cooper-Hobson G, Jaffe W. The benefits of otoplasty for children: further evidence to satisfy the modern NHS. J Plast Reconstr Aesthet Surg. 2009;62(2):190-4.
24. Calder JC, Naasan A. Morbidity of otoplasty: a review of 562 consecutive cases. Br J Plast Surg. 1994;47(3):170-4.
25. Shokrollahi K, Cooper MA, Hiew LY. A new strategy for otoplasty. J Plast Reconstr Aesthet Surg. 2009;62(6):774-81.
26. Weerda H, Siegert R. Complications in otoplastic surgery and their treatment. Facial Plast Surg. 1994;10(3):287-97.
27. Stucker FJ, Vora NM, Lian TS. Otoplasty: an analysis of technique over a 33-year period. Laryngoscope. 2003;113(6):952-6.
28. Bauer BS, Song DH, Aitken ME. Combined otoplasty technique: chondrocutaneous conchal resection as the cornerstone to correction of the prominent ear. Plast Reconstr Surg. 2002;110(4):1033-40.
29. Vuyk HD. Cartilage-sparing otoplasty: a review with long-term results. J Laryngol Otol. 1997;111(5):424-30.
30. Scharer SA, Farrior EH, Farrior RT. Retrospective analysis of the Farrior technique for otoplasty. Arch Facial Plast Surg. 2007;9(3):167-73.
31. Rigg BM. Suture materials in otoplasty. Plast Reconstr Surg. 1979;63(3):409-10.
Plastic surgeon, full member of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery), assistant physician of the Plastic Surgery Unit of the Clinics Hospital of Ribeirão Preto, Faculty of Medicine of the University of São Paulo, Ribeirão Preto, SP, Brazil.
Correspondence to:
Luis Fernando Ungarelli
Av. Norma Valério Correa, 571 - casa 85 - Jardim Botânico
Ribeirão Preto, SP, Brazil - CEP 14021-593
E-mail: lfungarelli@hotmail.com
Article submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: February 15, 2012
Article accepted: April 7, 2012
Work carried out at the Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo (Clinics Hospital of Ribeirão Preto, Faculty of Medicine of the University of São Paulo), Ribeirão Preto, SP, Brazil.



 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter