

Original Article - Year 2013 - Volume 28 -
Treatment of nasal valves in secondary rhinoplasty
Tratamento das válvulas nasais em rinoplastia secundária
ABSTRACT
BACKGROUND: Rhinoplasty is currently accepted as an operation for nasal remodeling, and not simply as a nose reduction procedure. Increasing attention has been given to preservation and repair of the function of nasal valves. The impairment of nasal valves is a distinct cause of symptomatic nasal obstruction; however there are disagreements regarding the forms of treatment.
METHOD: In the period between June of 2009 and January of 2011, 26 patients underwent secondary rhinoplasty to correct post-rhinoplasty insufficient function of internal nasal valves. Nasal septum autologous cartilage and auricular concha and rib cartilages were used for the treatments.
RESULTS: The treatments were effective in resolving nasal obstruction in 96.2% of the cases operated with autologous cartilage.
CONCLUSIONS: The use of expanding grafts from the nasal middle third and supporting grafts from lateral crura alar autologous cartilages proved to be effective in the treatment of post-rhinoplasty valvular impairment providing appropriate support to the nasal valves and resolution of clinical cases of nasal obstruction.
Keywords: Rhinoplasty. Cartilage. Nose/surgery.
RESUMO
INTRODUÇÃO: A rinoplastia é atualmente aceita como uma operação para remodelamento nasal, e não simplesmente um procedimento de redução do nariz. Cada vez mais atenção tem sido dispensada à preservação e à reparação da função das válvulas nasais. O comprometimento da válvula nasal é uma causa distinta de obstrução nasal sintomática, embora discordâncias existam com relação às formas de tratamento.
MÉTODO: No período de junho de 2009 a janeiro de 2011, 26 pacientes foram submetidos a rinoplastia secundária para correção de insuficiência de válvulas nasais internas pós-rinoplastia. Cartilagens autólogas de septo nasal, concha auricular e costela foram utilizadas para o tratamento.
RESULTADOS: Houve efetividade na resolução da obstrução nasal em 96,2% dos casos operados com cartilagens autólogas.
CONCLUSÕES: O uso de enxertos expansores de terço médio nasal e de enxertos de suporte das cruras laterais alares com cartilagens autólogas se mostrou efetivo no tratamento do comprometimento valvular pós-rinoplastia, proporcionando adequado suporte às válvulas nasais e resolução dos quadros de obstrução nasal.
Palavras-chave: Rinoplastia. Cartilagem. Nariz/cirurgia.
Initially depicted with exclusively reducing tactics, rhinoplasty is currently accepted as an operation for nasal remodeling. Increasing attention has been given to the preservation of function in nasal valves since the pioneering work of Sheen1-3.
The impairment of nasal valves is a distinct cause of symptomatic nasal obstruction; however, there is no consensus with respect to the forms of treatment4.5.
The nasal valve, with its internal and external components, has been described anatomically as the cross sectional area with the greatest resistance to the inspiratory respiratory flow, in the entire upper and lower respiratory system. Therefore, the prevention and treatment of impairments in this region during rhinoplasty is essential.
The external nasal valve is defined as the vestibule area under the wing, formed by the caudal septum, medial cruras of alar cartilages, edges of the nasal wing, and nose floor4 (Figure 1).
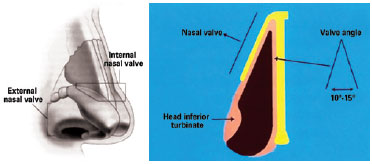
Figure 1 - Diagrams of internal and external nasal valves.
The internal nasal valve is located at about 1.3 cm from the nostrils' entrance and corresponds to the area below the lateral portion of the upper lateral cartilages (caudal portion), medially corresponding to the dorsal region of the septum, and inferiorly to the head of the inferior turbinate4 (Figure 1).
The aim of this study is to assess the quality of aesthetic and functional reconstruction of noses with post-rhinoplasty valvular impairment using autologous cartilage grafts.
Inter-relationship Between Aesthetics and Nasal Function
Bulbous nasal tip and nasal hump are among the most frequent complaints presented by patients who are candidates for rhinoplasty. The surgeon needs to adapt the procedure to each patient to obtain good results taking into consideration their anatomy, race, and gender.
The removal of the nasal tip cartilage and reduction of the nasal hump in predisposed patients, and when inappropriately carried out, can lead to aesthetic and functional changes in the nose such as:
The surgeon must individualize the treatment for each nose to prevent or minimize these potential complications by restructuring the cartilaginous skeleton manipulated during rhinoplasty and taking into consideration the aesthetic canons, prior anatomy, and the patient's desire 5-7.dorsum irregularities after the edema regression; nasal obstruction due to worsening internal nasal valve function, generally associated with a disconnection fostered between the upper lateral cartilages and the septum, and between the upper and lower lateral cartilages; nasal obstruction associated with resection of variable quantities of cartilage support that is aggravated by osteotomies that result in narrowing the piriformis opening; collapse of the middle third nasal cavity with the formation on an inverted "V".
METHOD
In the period between June of 2009 and January of 2011, 26 patients with chronic nasal obstruction associated to post-rhinoplasty internal nasal valve insufficiency were operated.
The patients' age ranged from 22 to 75 years old, averaging 37.4 years; 22 (84.6%) were females and 4 (15.4%) were males.
Nasal septum was the graft of choice for the repair of nasal valves due to its characteristics of thickness and resistance. Ear and costal cartilages were only used when the quantity of septum cartilage was inadequate for the treatment.
The sample included patients who complained of post-rhinoplasty nasal obstruction. Autologous cartilage grafts were used for the reconstruction of nasal valves and collapsing of the middle third. Patients with uncontrolled hypertension, decompensated diabetes, heart diseases, or other important systemic diseases were not included in this study.
The technique used for placement of grafts was that described by Sheen1, Gunter & Friedman8, Toriumi et al.9, and Rohrich et al.10.
All patients underwent open rhinoplasty in the hospital environment. Patients were positioned with their backs elevated to 30 degrees to reduce venous return and less transoperative bleeding. Total intravenous general anesthesia with controlled hypotension was employed. In addition, local topical and infiltrative anesthesia was used associated with topical and infiltrative vasoconstriction. All patients received thermoformable external immobilization for 7 days. Smooth hemostasis with hemostatic absorbable mesh, only involving the cornets, was used in cases of associated turbinectomies.
All patients were followed up for 12 months.
RESULTS
A resolution of clinical nasal obstruction was achieved in 25 out of the 26 operated patients (96.2%) according to the patients' reports.
The inspection of nasal valves was performed with rigid endoscopes to avoid valve distortion; valves were shown to be suitable for normal respiratory flow, with the exception of one patient in whom the nasal obstruction persisted. In this case, a clear persistence of valve collapsing during inspiration was observed.
In the 26 operated patients, septal cartilage was used in 18 (69.2%), costal cartilage was used in 6 (23.1%), and auricular concha cartilage was used in 2 (7.7%).
Although 13 (50%) of the patients included in the study had already undergone prior septoplasties and turbinectomies, 20 (76.9%) presented significant septal deviation of the nasal valve and/or persistence of hypertrophy of some region in the turbinates requiring review, which was performed during the same surgical time.
Ten out of the thirteen patients who underwent previous septoplasty presented residual deviations and needed re-intervention.
Some of the cases documented in this study's sample are reported as follows.
Case 1
Female patient, 23 years of age, who underwent closed rhinoplasty 15 months before this study and presented aesthetic and functional complaints of irregularities in the dorsum and nasal obstruction.
Septoplasty and turbinectomy had not been performed previously. The septum was the graft donor area (Figure 2).
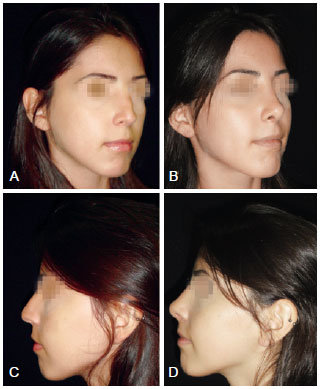
Figure 2 - Case 1. In A and C, preoperative aspect, respectively, in right oblique view and left profile. In B and D, 6 months postoperative aspect of nasal valve treatment with septal cartilage graft, respectively, in right oblique view and left profile.
Case 2
Female patient, 35 years of age, who has previously undergone rhinoplasty associated to septoplasty, performed by two different surgeons, one being a plastic surgeon and one being an otorhinolaryngologist, at the same surgical time and about 2 years before this study. The patient presented aesthetic and functional complaints related to tip deviations and nasal obstruction (Figures 3 and 4).
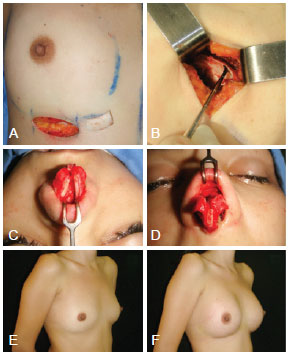
Figure 3 - Case 2. Obtaining and positioning costal cartilage grafts, obtained through the same incision in the breast groove where breast implants were placed. In A and B, obtaining the costal cartilage graft. In C and D, intraoperative aspects of rhinoplasty associated with septoplasty. In E, preoperative aspect of breast. In F, aspect after breast implant.
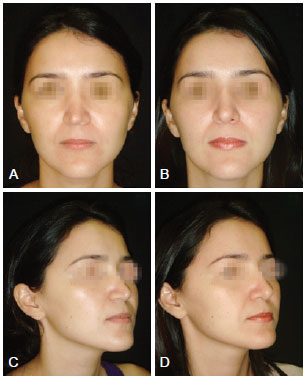
Figure 4 - Case 2. In A and C, preoperative aspect, respectively, in right front and oblique view. In B and D, postoperative aspect of nasal valve treatment with costal cartilage graft, respectively, in front and right oblique view.
Case 3
Female patient, 46 years of age, who previously underwent rhinoplasty 12 years before this study, presented sealed and collapsing nasal middle third, pinched nasal valves, concealed columella, nose tip without definition, or support and chronic nasal obstruction resulting from internal nasal valve insufficiency. We opted to grafting with costal cartilage due to the requirement of large amounts of cartilage to treat the deformities (Figures 5 to 7).
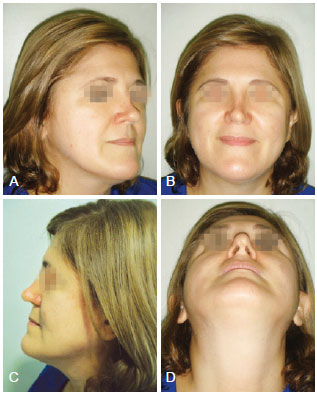
Figure 5 -Visualization of various deformities described in case 3, respectively in right oblique and front views, and left profile and nose base.
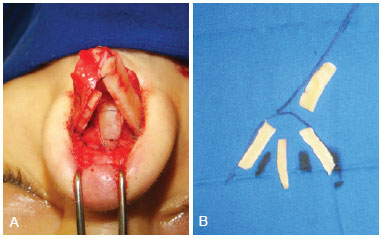
Figure 6 - Case 3. In A, intraoperative aspect demonstrating the need for using multiple cartilaginous grafts due to total absence of lateral crura right alar cartilage. In B, trans-operative schedule for use of grafts.
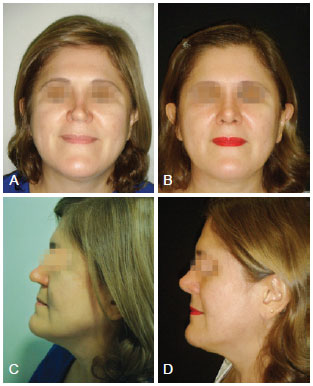
Figure 7 - Case 3. In A and D, preoperative aspect, respectively, in front view and left profile. In B and C, 6 months post-operative aspect of secondary rhinoplasty with multiple cartilaginous grafts, including ribs, respectively in front view and left profile.
Case 4
Male patient, 56 years of age, previously subjected to rhinosseptoplasty 20 before this study, evolved with nasal obstruction associated with internal nasal valve insufficiency and low quality of sleep (Figure 8).
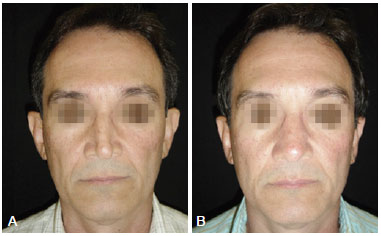
Figure 8 - Case 4. In A preoperative aspect in front view. In B, 12 months postoperatively aspect of treatment of nasal valves with auricular concha grafts in frontal view.
Case 5
Female patient, 38 years of age, previously submitted to rhinoplasty 3 years before this study, complained about right nasal obstruction. Nasal valves contour asymmetry was observed in the external examination. The functional result was outstanding, however, at the expense of a wider middle third (Figure 9).
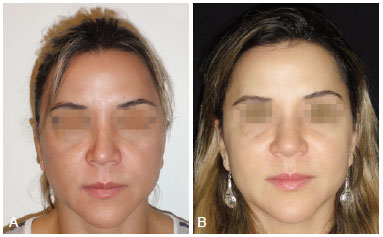
Figure 9 - Case 5. In A, preoperative aspect in front view. In B, 90 days postoperative aspect of nasal valve treatment with costal cartilage grafts, however, with large nasal enlargement, in front view.
DISCUSSION
Increasing attention has been given to nasal function during the performance of rhinoplasty since the works of Sheen1 in early 1980. In 1984, Sheen1 described the "spreader graft", indicating those patients with higher risk of presenting functional problems namely: Caucasian noses with thin skin, narrow nasal valves, and short nasal bones. The risk is even higher when these characteristics are associated with nasal septum deviation on the nasal valve region and presence of chronic-inflammatory diseases in the nasal mucosa, such as rhinitis5.
The descriptions of "lateral crural strut graft" by Gunter & Friedman8, and of "alar batten grafts" by Toriumi et al.9 are other milestones of equal importance in modern rhinology, both in primary and secondary rhinoplasty cases7.
In 2008, Rhee et al.6 conducted a 25 years review on treatment of nasal valvular dysfunction, including prospective and retrospective works, most of them with a level 4 of scientific evidence according to the Oxford Center for Evidence-Based Medicine. This study demonstrated that, regardless of the technique used, all modernly described surgical approaches were effective6.
In the cases presented in this study, septal cartilage was preferably used in the majority of grafts used in rhinoplasties because of its appropriate characteristics in thickness and resistance. Rib and auricular concha cartilages were only used when septal cartilage was not available.
The techniques described by Sheen1-3 and Gunter7.8, considered to be part of the "modern rhinoplasty", were basically used in this study because of their strong functional awareness and by being highly effective when properly carried out.
In this series of cases, a resolution of the clinical nasal obstruction was achieved in 96.2% of the operated patients, which represents an index that is similar to those reported in the literature6.
In the case of nasal valve failed repair, a costal graft of inadequate thickness was used, larger than the indicated, causing bulging and obstruction of the nasal valve.
With regard to the aesthetic result, the costal cartilage grafts must be carefully carved to not excessively bulge the middle third and nasal tip, solving the functional problem, however, creating an unaesthetic nose.
Two points are important to prevent functional changes during primary rhinoplasty: modeling the nasal tip through suture points associated to minor and resections, de-structuring the nasal tip cartilage; and gradual reduction of the dorsum with adequate preservation of the upper lateral cartilages, as described by Rohrich10.
The trans-operative bleeding control is guaranteed through general anesthesia with controlled hypotension, intense topical vasoconstriction associated to little infiltrative anesthesia, and finally, by positioning the patient with their backs elevated to 30 degrees.
In the postoperative period, the most important measure to prevent bleeding is the absolute control of blood pressure because we do not use traditional nasal tamponade in any case. A mild hypotension is desirable in the immediate postoperative period.
CONCLUSIONS
The surgical techniques that use "spreader grafts", "lateral crural strut grafts", and "alar batten grafts" with autologous cartilage grafts have proven effective in the treatment of post-rhinoplasty valvular impairment providing appropriate support to nasal valves and showing great effectiveness in the resolution of clinical cases of nasal obstruction.
Extreme care must be taken in sculpting rib and auricular concha grafts to avoid excessive thickness in the middle third and in the nasal tip because these can become obstructive and unaesthetic.
REFERENCES
1. Sheen JH. Spreader graft: a method of reconstructing the roof of the middle nasal vault following rhinoplasty. Plast Reconstr Surg. 1984;73(2):230-9.
2. Sheen JH. Rhinoplasty: personal evolution and milestones. Plast Reconstr Surg. 2000;105(5):1820-52.
3. Sheen JH, Sheen AP. Aesthetic rhinoplasty. 2nd ed. St. Louis: Mosby; 1998.
4. Rhee JS, Weaver EM, Park SS, Baker SR, Hilger PA, Kriet JD, et al. Clinical consensus statement: diagnosis and management of nasal valve compromise. Otolaryngol Head Neck Surg. 2010;143(1):48-59.
5. Almeida GS, Pessoa BBGP, Oliveira NGS, Gomes AAR, Crisóstomo MR, Pessoa SGP. Reconstrução do terço médio nasal em rinoplastia primária. Rev Soc Bras Cir Plást. 2008;23(2):124-7.
6. Rhee SJ, Arganbright JM, McMullin BT, Hannley M. Evidence supporting functional rhinoplasty or nasal valve repair: a 25-year systematic review. Otolaryngol Head Neck Surg. 2008;139(1):10-20.
7. Gunter JP, Rohrich RJ, Adams Jr WP. Dallas rhinoplasty: nasal surgery by the masters. St. Louis: Quality Medical Publishing; 2002.
8. Gunter JP, Friedman RM. Lateral crural strut graft: technique and clinical application in rhinoplasty. Plast Reconstr Surg. 1997;99(4):943-52.
9. Toriumi DM, Josen J, Weinberger M, Tardy ME Jr. Use of alar batten grafts for correction of nasal valve collapse. Arch Otolaryngol Head Neck Surg. 1997;123(8):802-8.
10. Rohrich RJ, Muzaffar AR, Janis JE. Component dorsal hump reduction: the importance of maintaining dorsal aesthetic lines in rhinoplasty. Plast Reconstr Surg. 2004;114(5):1298-308.
Plastic surgeon, member of the Brazilian Society of Plastic Surgery, Fortaleza, CE, Brazil
Correspondence to:
Glauco Soares de Almeida
Av. Rui Barbosa, 343 - ap. 2.002 - Meireles
Fortaleza, CE, Brazil - CEP 60115-220
E-mail: glaucosoaresalmeida@ig.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: 5/20/2012
Article accepted: 10/1/2012
Work performed at the author's private clinic, Fortaleza, CE, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter