

Original Article - Year 2013 - Volume 28 -
Head and neck microvascular reconstruction: retrospective analysis of 60 free flaps
Reconstrução microcirúrgica em cabeça e pescoço: análise retrospectiva de 60 retalhos livres
ABSTRACT
BACKGROUND: The microsurgical reconstruction of the head and neck usually is the first indication for high tissue losses. The surgical team should have a hand full of technical options for any such loss. In addition, knowing how to manage each complication is important. The aim of this study was to evaluate possibilities of reconstruction and complications in patients submitted to reconstruction using free flaps.
METHODS: A retrospective study of patients with loss of tissue in the head and neck, and submitted to immediate or delayed microsurgical reconstruction was performed between March 2010 and March 2012.
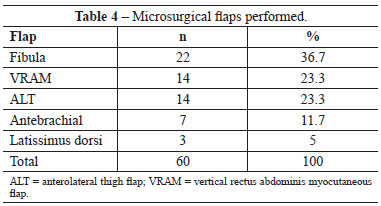
RESULTS: Sixty patients that received free flaps, submitted to surgery between March 2010 and March 2012, were analyzed. Of these, 31 (52.7%) patients were women and 39 (65.0%) Caucasians. Immediate reconstructions were performed in 65% of the cases. Malignant tumours were the most common diagnoses, representing 86.7% of the cases. The frequency of the flaps was as follows: fibula in 36.7% of the cases, rectus abdominis in 23.3%, anterolateral thigh in 23.3%, antebrachial in 11.7%, and latissimus dorsi in 5%. The average operative time was 8.6 ± 2.1 hours. The ischemia period was 107.5 ± 27.6 minutes. Complications were observed in 45% of the cases: dehiscence in 18.3%, salivary fistula in 16.7%, infection in 16.7%, death during the first week after surgery in 5%, and extrusion of the synthesized material in 1.7%. Among the 60 flaps, a reintervention was required in 20 (33.3%), and the loss of 13 (21.7%) reconstructions occurred. The venous thrombosis was the main cause of reintervention and loss. Six salvage flaps were performed, three of the pectoralis pedicle and three of the latissimus dorsi muscle; two of the latter were microsurgeries.
CONCLUSIONS: Microsurgical flaps are important in head and neck reconstructions; these can be better than the pediculated ones in most of the situations; when well succeeded, they promote a decrease of morbidity, a promotion of rehabilitation, and a decrease in costs.
Keywords: Microsurgery. Head/surgery. Neck/surgery. Surgical flaps.
RESUMO
INTRODUÇÃO: A reconstrução microcirúrgica de cabeça e pescoço costuma ser a primeira indicação para grandes perdas teciduais. A equipe cirúrgica deve ter um arsenal de opções técnicas para qualquer dessas perdas. Além disso, é importante conhecer o manejo de cada complicação. O objetivo deste estudo é avaliar as condutas de reconstrução e as complicações de pacientes submetidos a reconstruções com retalhos livres.
MÉTODO: Foi realizado estudo retrospectivo de pacientes com perda de substância em cabeça e pescoço, submetidos a reconstruções microcirúrgicas imediatas ou tardias, no período de março de 2010 a março de 2012.
RESULTADOS: Foram analisados 60 pacientes que receberam retalhos livres, dos quais 31 (52,7%) eram mulheres e 39 (65%), caucasianos. As reconstruções foram imediatas em 65% dos casos. Os tumores malignos foram o diagnóstico mais comum, representando 86,7% dos casos. A frequência dos retalhos foi a seguinte: fíbula, em 36,7% dos casos; reto abdominal, em 23,3%; ântero-lateral da coxa, em 23,3%; antebraquial, em 11,7%; e grande dorsal, em 5%. O tempo cirúrgico médio foi de 8,6 ± 2,1 horas. O tempo de isquemia foi de 107,5 ± 27,6 minutos. Foram observadas complicações em 45% dos casos: deiscência em 18,3%, fístula salivar em 16,7%, infecção em 16,7%, óbito na primeira semana pós-operatória em 5%, e extrusão de material de síntese em 1,7%. Dos 60 retalhos, houve reintervenção em 20 (33,3%) e perda de 13 (21,7%) reconstruções. A trombose venosa foi a causa mais comum de reintervenção e de perda. Foram realizados 6 retalhos de resgate, 3 de peitoral pediculado e 3 de músculo grande dorsal, sendo 2 microcirúrgicos.
CONCLUSÕES: Os retalhos microcirúrgicos têm grande importância em reconstrução de cabeça e pescoço, podem ser superiores aos pediculados na maioria das situações, e determinam, quando bem-sucedidos, diminuição da morbidade, aceleração da reabilitação e diminuição de custos.
Palavras-chave: Microcirurgia. Cabeça/cirurgia. Pescoço/cirurgia. Retalhos cirúrgicos.
The surgical treatment of advanced neoplasia, great trauma and serious infections in the head and neck region usually causes large functional and esthetic sequels. Furthermore, tissue gaps may be extensive and difficult to reconstruct1.
Microsurgical reconstruction, also known as microvascular autologous transplantation, is the preferred procedure in the reconstruction of large tissue losses.
This type of reconstruction may be performed immediately or delayed. However, in the oncological cases, the immediate reconstruction is usually preferred, to enable a faster functional and esthetic rehabilitation of the patient, and to enable the patient to be directed to radiation therapy within a short period of time.
Diverse variables should be taken into account in order to succeed in these reconstructions. A trained team is required, ideally including two microsurgeons, an anesthesia team able to maintain stable hemodynamic parameters to enable adequate flow through the anastomosis, selection of patients with controlled surgical risk, effective monitoring during post-operative management, prevention and management of situations that lead to an unsuccessful procedure, and the promptness for reintervention, whenever it is necessary.
The main objective of this work was to describe a retrospective series of microsurgical reconstruction cases in patients with extensive tissue loss in the head and neck. The secondary objectives were to analyze the recommended flaps used for each case, as well as the complications resulting from each flap.
METHODS
This is a retrospective study of patients presenting loss of tissue in the head and neck, submitted to immediate or delayed microsurgical reconstructions, from March 2010 to March 2012 at the National Cancer Institute, in the Hospital das Clínicas of Porto Alegre, or at the Santa Casa of Porto Alegre. In all cases, the author performed the dissection of the flap and the microvascular anastomoses.
Selection of the Surgical Technique
The selection routines for the microsurgical flaps used in reconstruction are shown in Table 1. The reconstructions are illustrated in Figures 1 to 9.
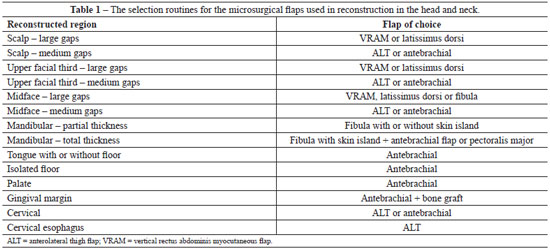
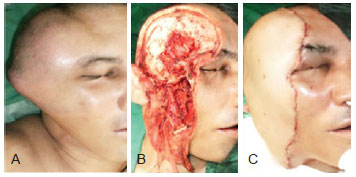
Figure 1 - Reconstruction with latissimus dorsi flap. In A, Pre-operative view. In B, Intra-operatory view. In C, Post-operative view.
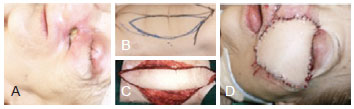
Figure 2 - Reconstruction with vertical rectus abdominus myocutaneous flap. In A, Pre-operative view. In B, Pre-operative marking. In C, Intra-operatory view. In D, Post-operative view.
Figure 3 - Reconstruction with anterolateral thigh flap. In A, Pre-operative view. In B, Pre-operative marking. In C, Post-operative view.
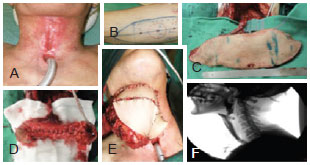
Figure 4 - Reconstruction cervical esophagus with anterolateral thigh flap. In A and B, Pre-operative views. In C and D, Intra-operatory views. In E, Immediate post-operative view. In F, esophagogram demonstrating the passage of contrast through the reconstructed segment.
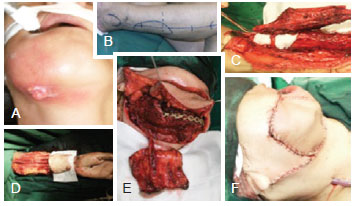
Figure 5 - Mandibular reconstruction with fibula flap with skin island and antebrachial flap. In A and B, Pre-operative views. In C, D and E, Intra-operatory views. In F, Immediate post-operative view.
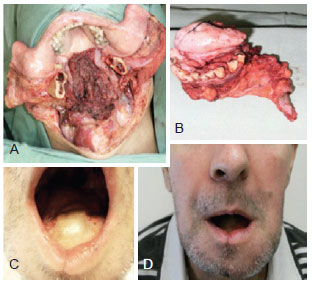
Figure 6 - Patient subjected to resection of relapsed squamous cell carcinoma. In A and B, we can observe that it is a commando resection and that involved the mandibular central arch. The reconstruction was performed with a free flap of fibula with skin island. In C, Post-operative view showing the skin flap used to reconstruct the oral cavity. In D, Satisfactory post-operative view of the lower facial third. The lip scar presents a fold, which is a consequence of the diastasis of the orbicularis muscle fibers. This deformity should be corrected in the future.
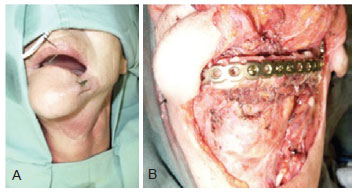
Figure 7 - Delayed and secondary reconstruction in a female patient previously reconstructed only with titanium plate. The plate presented extrusion and was successfully replaced by another and a fibula microsurgical flap without skin island. In A, Pre-operative view. In B, Intra-operatory view with the reconstruction and anastomosis already finished.
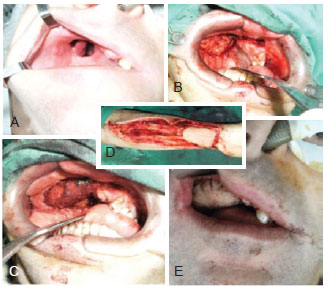
Figure 8 - Complex reconstruction of the maxilla. In A, Pre-operative view showing loss of substance by resection of a fibrous dysplasia. In B, Intra-operatory aspects of the resconstruction performed with facial fat flap (Bichat's ball). In C, iliac crest flap. In D and E, microsurgical antebrachial flap.
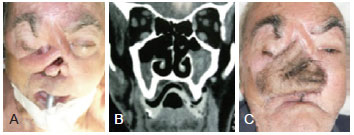
Figure 9 - Reconstruction of the midface with antebrachial flap. In A, Pre-operative view. In B, Tomography demonstrating the previous tissue loss area due to previous tumor resections, as well as presence of relapsed tumor. In C, Post-operative view.
Whenever possible, the selected flap was dissected simultaneously to the oncologic resection or preparation of the receptor site. This way, the operative time was shortened. In delayed reconstructions, we waited for the dissection, cervicotomy, and identification of receiver vessels were performed, in order to determine if they existed and whether they were viable for the anastomosis. If there were no receiver vessels, three alternatives were possible: contralateral dissection; vein graft to prepare an arteriovenous loop and reconstruct a pedicle; termination of the microsurgical procedure. The dissection of the flap vessels was usually performed using a 2.5x magnifying glass.
The dissection of free flaps can be performed in more than one way. We opted to first identify the pedicle, followed by preparation of the flap area. This procedure enables the identification of the pedicle viability and to avoid unnecessary incisions and handling in case the microsurgical flap is cancelled.
The selection of receiver vessels was performed according to caliber similarity with the donors. Generally, the selection procedure consisted in choosing between the external carotid, facial or lingual arteries. However, when selecting the vein, usually a branch of the thyrolinguofacial trunk or the trunk itself was used. Whenever possible, an end-to-end anastomosis was conducted with interrupted suture. The arterial anastomosis was performed with an 8-0 nylon thread and the venous anastomosis with a 9-0 nylon thread.
Approximately 15 to 20 minutes before the ischemia period, the patient was administered with 3,000 IU of intravenous heparin sodium. When ischemia started, the flap was washed with approximately 200 ml of a ringer lactate solution with 5,000 IU of heparin sodium, by cannulation of the artery from the flap with an Abocath/Gelco number 22.
The vascular stumps were prepared for the anastomosis with the aid of microscopic visualization. The position of the vessels was established in order to prevent the occurrence of a kink, rotations, excessive traction, or compression by the flap itself. In the cases where a risk of torsion in the anastomosis region was present, a plicature was performed with a 4-0 or 5-0 nylon thread in a portion of the pedicle, to prevent the torsion. When the distortion remained, the anastomosis was remade.
In case of mandibular reconstructions, the assembling of the fibula was performed before the ischemia period started, which means in situ. Therefore, the plate or template was shaped, when possible, with the patient in the occlusion position and before bone resection. Subsequently, the number of necessary osteotomies was determined, and the periosteum of the osteotomies region was dissected. After performing the osteotomy, a monocortical fixation was performed with the screws from the same plate system, i.e. 2.4 mm or 2.7 mm. Generally, two screws per osteotomized segment were used. In the native bone of the mandible, bicortical fixation was performed. The care given to the ischemia period was not so rigorous in the cases where skin was not present since the bone is more tolerant to the lack of blood circulation. Thus, we agreed to perform the osteosynthesis during the ischemia period.
The condylar reconstruction was performed in more than one way. In some cases, a cartilage graft and shaping of the fibula extremity was performed, whereas in other cases, it was left floating, enclosing the mandible with the maximum of muscle layers. The reconstruction of the temporomandibular joint is controversial, complex and has variable results, and is not part of the scope of this work.
After finishing the anastomoses, we waited for 5 minutes to check for possible leaks, maintaining a moist gauze over it. In case a leakage occurred during that period, new sutures would be performed, without clamping. After this step, a patency test was performed using two microsurgical tweezers. If an alteration was observed, the anastomosis would be remade, as many times as necessary according to the surgeon. If the problem remained, the receptor vase would be replaced. If there was still no success, the microsurgical reconstruction would be terminated and replaced by another made from pediculated flaps or deferred for the next operative time, depending on the conditions of the patient and on the integrity of the deep structures that had to be covered.
The mean blood pressure was maintained, whenever possible, above 70 mmHg. Therefore, vasopressors, considered harmful to the microvasculature of the flap, were not used. The use of colloids may also be harmful since they direct fluid into the third-space, increasing the risk of congestion and flap loss, as a consequence of venous return decrease and thrombosis. According to many anesthesiologists, an early beginning of the transfusion is an adequate measure for the surgeries involving microvascular anastomoses.
The patients were maintained in an intensive care unit or in rehabilitation rooms under semi-intensive support, with a doctor on duty (anesthetist or intensivist), in order to monitor the hemodynamic parameters.
The evaluation of the flaps was performed approximately in every three hours, through the clinical parameters color and venous return to digital pressure, bleeding to a 25 Gauge needle puncture and measurement of capillary glycemia of that blood. When available, the evaluation by doppler was used. An alteration in those parameters would indicate reintervention.
All the patients signed a written informed consent before the procedures. The patients' identities were kept anonymous. The photographs of this work demonstrate only the anatomical region (or part of it) strictly necessary to present the tissue gap and the reconstruction that was performed.
RESULTS
Between March 2010 and March 2012, 61 microsurgical reconstructions were performed. One patient was excluded because she was submitted to mammary microsurgical reconstruction, making up 60 free flaps performed in the head and neck. From these, two patients were submitted to salvage reconstruction with microsurgical flaps, and one female patient was reconstructed with two simultaneous microsurgical flaps.
Thirty one (52.7%) patients were women. The average age was 52.0 ± 15.7 years, ranging from 13 to 81 years old. Regarding ethnic background, 65% of patients were Caucasians and the remaining patients were Afro-descendants. Twenty one (35%) patients were submitted to delayed reconstructions. Two patients were submitted to salvage microsurgical reconstructions (a second free flap), and one female patient was reconstructed with two free flaps in the same operative time.
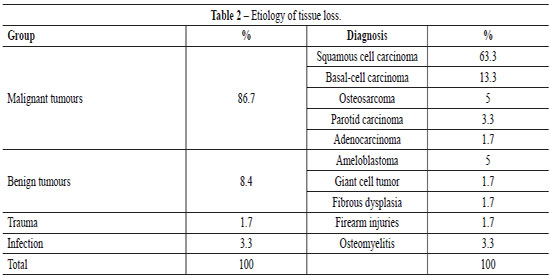
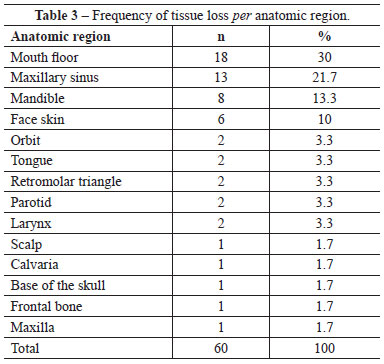
The etiology of the cases is presented in Table 2. Malignant tumours were the most frequent etiologic group with 86.7% of the cases. Squamous cell carcinoma was the most common diagnosis, representing 63.3% of the cases. The anatomic regions of the neoplasia are shown in Table 3. On the other hand, Table 4 shows the microsurgical flaps that were performed. In the reconstructions with fibula, a skin island was used in 13 of the 22 cases (59.1%).
The average operative time was 8.6 ± 2.1 hours, and the ischemia period for the anastomoses was 107.5 ± 27.6 minutes. Regarding the presence of complications, no difference between the times of surgery and ischemia was observed. The arterial anastomosis was performed in the external carotid in 76.7% of the cases, in the facial artery in 21.7%, and in the superior thyroid artery in 1.7%. The venous anastomosis was conducted in the thyrolinguofacial trunk in 78.3% of the cases, in the lingual vein in 18.3%, in the internal jugular vein in 1.7%, and in the inferior thyroid vein in 1.7%. The arterial anastomosis had to be remade in three (5%) patients, and the venous anastomosis in two (3.3%) cases.
Twenty-seven (45%) patients presented complications, but reintervention was not necessary in all the cases. Table 5 shows the incidence of those complications; some patients had more than one complication. No statistical difference was observed between type of flap and the presence of complications (chi-square: 1.2; P = 0.876). However, patients with malignant tumors that were reconstructed with free fibula flap presented more complications than those who had a diagnosis other from cancer (P = 0.012). The patients whose fibulas were reconstructed with skin islands (osteocutaneous flap) presented a higher incidence of complications than those with no associated skin flap (P = 0.026), but there was no significant increase in flap loss (P = 0.076). The gaps at the mandibular central arch did not represent significant risk of complications (P = 0.454) nor microsurgical flap loss (P = 0.620).
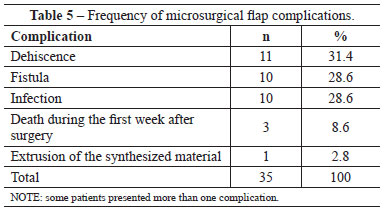
From the 60 flaps that were performed, 20 (33.3%) were subjected to reinterventions and 13 (21.7%) were lost. Tables 6 and 7 show the reasons why reinterventions were necessary and why flaps were lost, respectively. Venous thrombosis was the major cause of reintervention (40%) and flap loss (46.1%). Three deaths were registered in the immediate post-operative period (until 7 days) among the cases included in this study. All complications occurred in patients with malignant tumors. The success of reintervention was 60% (12 among 20 reoperated flaps). A statistical difference was observed between subjecting or not a flap in distress to surgery to save it (chi-square = 5.9; P = 0.022).
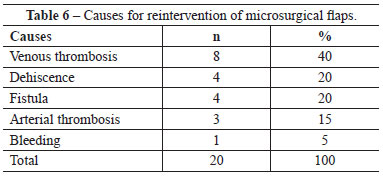

Table 8 shows the distribution of complications per microsurgical flaps, and Table 9 shows the microsurgical flaps loss.

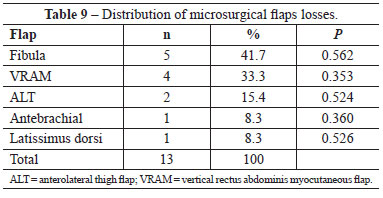
Tables 10 and 11 illustrate the evaluation of risk factors for complications and flap loss. The diagnosis of malignant tumor and the anatomic region were significant for the complications; on the other hand, the arterial reanastomosis, presence of complications, and the need for surgery reintervention presented a statistically significant correlation with the loss of free flaps.

In the cases presented here, six salvage flaps were performed, and among these three were pectoralis major muscle flaps (Figure 10) and three latissimus dorsi muscle flaps. From the latter group, two were microsurgical. All the salvage flaps survived.
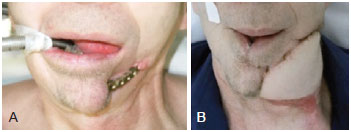
Figure 10 - Patient subjected to a pectoralis major myocutaneous flap, as a rescue procedure to the loss of a microsurgical flap of fibula with skin island. In A, Pre-operative view of microsurgical reconstruction, recommended due extrusion of previous reconstruction, performed only with titanium plate, while we were waiting for the liberation of the oncological teams. In B, View of the pectoralis major flap during the first month post-operative.
DISCUSSION
Microsurgical reconstruction is a set of required techniques in of complex surgical reconstructions centers2. Undoubtedly, microsurgery is mostly needed in the reconstruction applied to the oncological surgery of the head and neck. Microsurgery decreases morbidity and rehabilitation treatment costs. However, microsurgery requires a rigorous screening of patients, a trained team for the procedure and post-operative period, and a hospital with the appropriate resources for its accomplishment. Lack of availability of a surgery room for a prompt reintervention is one of the obstacles found among our centers; it is responsible for losses due to the increasing ischemia periods of the flaps. It should also be taken into account that the patients were subjected to surgery during the learning curve, which determined a markedly higher incidence of complications. The success shown in these cases (78.3%) remains lower than that of the great reference centers, which have success rates above 91%. The reintervention rate was also higher in our sample (33.3%), when compared to that described in the literature, which varies from 6% to 14%3. The reintervention rate in a study performed with 1193 flaps at the Memorial Sloan-Kettering Cancer Center was 6% due to arterial or venous thrombosis in 53% of the cases, whereas in this study thrombosis was responsible for 47.9% of the reoperations4. According to that previous study, venous thrombosis was the most common (74% of all the thrombosis cases), but showed a higher rescue rate (71%)4. In the present study, it was only possible to save two (25%) of the eight venous thrombosis cases. There was no success in the reintervention of the arterial thrombosis cases (n = 3). Approximately 80% of the thrombosis cases occur in the first two days post-surgery, and 90% of arterial thromboses occur one day after the procedure5. Therefore, evaluations should be performed very frequently during that period and reintervention should be recommended at the slightest suspicion.
The recommendations for the use of each flap vary between institutions but some flaps are established for certain situations. Immediate reconstructions are preferable whenever possible. This way, cicatricial complications occur less, and the surgical technique is easier. However, in cases where there are doubts regarding the tumor resection margins, which may require reintervention, it is preferable to wait and perform a delayed reconstruction.
The antebrachial flap was described by Yang, in 1981, and has been widely used in cervicofacial reconstructions6. One of the advantages of this flap is its small thickness, thus, it does not implicate excessive volumes. It can be used in different situations, i. e. tissue gaps and can be molded for complex gaps, even when it is used folded over itself ("folded flap"). The vascular pedicle is stable and the tolerance to radiation therapy in the post-surgery period is high. It is always important to consider the cephalic vein since it presents significantly higher caliber and length than the concomitant radial veins. According to our experience, we have been using the cephalic vein very frequently, which has become our selected vein from the donor area, whenever viable. Although the antebrachial flap is not used often anymore, it can be produced as an osteo-fasciocutaneous flap. In the mandibular reconstruction, that technique is seldom used since the fibula is presently the gold-standard, and the subsequent flap to be chosen would be the one from the iliac crest (except for the mandibular central arch). In addition, it can be elevated in conjunction with the tumor resection, decreasing the operative time. Aki et al.6 performed that flap among a group of 11 patients that were not older than seven years old. The donor area should be tested with the Allen test, which is negative when the test is normal, i. e. when there is vascular sufficiency of the ulnar artery for the blood flow in the hand. When the sensitive reinnervation of the receiving area is planned, the forearm medial or lateral cutaneous nerves may be dissected, taken to the flap and anastomosed with a sensory nerve. The antebrachial flap is routinely used in our institution for reconstruction of the oropharynx and the tongue, and used as second microsurgical flap for the reconstruction of total thickness in the mandibular region. It can be used for the reconstruction of the gingival arch, together with bone grafts. However, if the receptive area was subjected to radiation therapy, the flap survival might be compromised. Probably, the previous and post-surgery implementation of hyperbaric oxygen therapy might result in some modification of the prognosis of such type of reconstruction. When radiation therapy is not a concern, we rather use a corticomedullary flap of the iliac crest, which gives satisfactory thickness and bone regeneration characteristics for the stomatognathic rehabilitation, including in implants or dental prosthesis.
The disvantage of the antebrachial flap is the usage of a hand artery and leaving a visible scar, since the donor area will always require a skin graft for its production. The fibrosis of that area will probably compromise the tendinous movement of the flexor region in particular. The early care of patients subjected to those procedures by a physiotherapist is suggested.
Reconstruction with two free flaps was selected only for one of the cases, but it is the selected procedure in many reference centers1. This method is useful in particular in gaps of the oral cavity, and also in mandibulectomy with loss of total thickness ("through-and-through") cases. Groth & Silva2 documented reconstructions with two flaps in six patients, and showed a preference for the association of fibula and the anterolateral area of the thigh. However, these types of reconstruction require at least two microsurgeons and a large team, as a result of the significant increase of the operative time. In our case series, we performed only one case like that. The association between a free flap and a pediculated flap is an alternative solution that is not as perfect, but offers similar efficiency.
The mandibular reconstruction comprising autologous transfer of the vascularized bone by microsurgical technique (free flap) is the gold-standard for gaps above 5 cm, in particular in the central arch losses (space between canine teeth)7,8. The fibula was the chosen flap for the mandibular reconstruction. The iliac crest is not so often used in our institution due to morbidity of the donor area, and to the difficulty or even impossibility to reconstruct the mandibular central arch with it. In addition, the skin flap is usually large, as a consequence of the underlying layers. The fibula flap causes little morbidity to the donor area9. The patients that required skin island have higher complication rates , probably due to the morbidity of the resection and to the exposure of the flap to the oral cavity. In this study, the central arch resection did not increase the complication rate; however, this finding contrasts with a previous study by the same authors that included 85 cases of mandibular reconstruction10.
In the middle face reconstructions, the vertical rectus abdominis myocutaneous (VRAM) flap is usually the preferred. This flap allows the closure of not only the skin but also the mucosae, as well as the obliteration of the dead spaces of the paranasal sinuses regions.
The free flap from the latissimus dorsi muscle is an alternative to the VRAM, since it has similar characteristics but with a higher muscle volume than skin volume. This flap is usually used for reinterventions in our working environment. It requires the change from the decubitus position during the surgery period, which is a disadvantage of this technique since it extends the operative time.
The anterolateral thigh (ALT) flap is increasingly gaining space in the microsurgical reconstruction possibilities. It allows the reconstruction of less extensive areas, more than the VRAM and the latissimus dorsi, but is becoming increasingly recommended for the reconstruction of the cervical esophagus because it can be segmented into three parts due to its three perforators. When all are viable, the central portion is used - with the main perforator - to produce the esophagus tube, and the remaining portions for the skin coverage and the tracheostoma.
The reconstruction of the cervical esophagus is performed mainly with microsurgical technique. The ALT flap has a lower dysmotility incidence than the jejunum, presents less morbidity since it doesn't require laparotomy, and has a lower incidence of fistulas than the pediculated reconstructions or the antebrachial flap11-13. The morbidity of the donor area is low because primary closure can be performed; there is no sectioning of the muscles, and even if part of the vastus lateralis muscle is removed no significant gait dysfunction is observed. The VRAM flap, together with the transverse rectus abdominis myocutaneous (TRAM) flap, lead to alterations in the abdominal wall, making it vulnerable to bulging and hernias; these do not occur with the ALT flap.
The microsurgical flaps present vascular complications as major disadvantages . The venous thrombosis is the main disadvantage and the highest cause of flap loss. Unlike pediculated flaps, they generally function under the law of "all or nothing", i.e., ischemia and necrosis are usually total. Their emergence is more likely in the first two to three days. After this period, the risk declines substantially. The arterial thrombosis has different characteristics. It can occur even after the first week and indicate a condition in the vascular wall, which decreases the chance of success of the reintervention. When receiver vessels are not adequate or when we have a large distance to a higher quality vessel, we may choose to use a arteriovenous loop anastomosis (AV loop), which consists in producing an arteriovenous fistula with a vein flap, usually saphenous or cephalic, leaving the definitive anastomosis for the second procedure; this will be performed after the vein graft section, with one part being used for the venous anastomosis and the other for the arterial anastomosis. In our group of cases this technique was not conducted.
Salivary fistula comprises another important complication11. It increases the risk of infection, thrombophlebitis, anastomosis thrombosis, and flap loss. It also significantly increases hospitalization time, which may cause other complications such as nosocomial infections, malnutrition, and depression8.
A rescue routine should also be included in the services that perform microsurgery reconstructions. When the free flap is the first option, other options such as pediculated or microsurgical flaps could be present. In the cases when reconstructions are performed in the lower facial third, we prefer to use the pectoralis major flap (Figure 6). However, the situation becomes more difficult when there are no adequate pediculated flaps, or when the recipient vascular bed for the anastomoses is damaged.
CONCLUSIONS
The microsurgical flaps are of utmost importance in the reconstruction of complex and extensive cases in the head and neck (usually oncological), and are the first recommendation for many cases. These are superior to pediculated flaps in the majority of cases, and when successful determine a decrease of morbidity, promotion of rehabilitation, and decrease in costs. In order to diminish vascular complications a selection, recommendations and management routine of those cases are important. The loss of a free flap severely increases morbidity, since another flap is many times required, thus creating at least two donor areas and exposing the patient to multiple procedures. Thus, a good standard of the technique should be attained in the first surgery, besides urgent exploring measures, and an aggressive approach with the aim of saving the flap in case it is required.
REFERENCES
1. Wei FC, Demirkan F, Chen HC, Chen IH. Double free flaps in reconstruction of extensive composite mandibular defects in head and neck cancer. Plast Reconstr Surg. 1999;103(1):39-47.
2. Groth AN, Silva ABD. Reconstrução oromandibular complexa com dois retalhos microcirúrgicos. Rev Bras Cir Plást. 2009;24(1):11-21.
3. Kubo T, Yano K, Hosokawa K. Management of flaps with compromised venous outflow in head and neck microsurgical reconstruction. Microsurgery. 2002;22(8):391-5.
4. Bui DT, Cordeiro PG, Hu QY, Disa JJ, Pusic A, Mehrara BJ. Free flap reexploration: indications, treatment, and outcomes in 1193 free flaps. Plast Reconstr Surg. 2007;119(7):2092-100.
5. Kroll SS, Schustermann MA, Reece G, Miller MJ, Evans GR, Robb GL, et al. Timing of pedicle thrombosis and flap loss after free-tissue transfer. Plast Reconstr Surg. 1996;98(7):1230-3.
6. Yang G, Chen B, Gao Y. Forearm free skin flap transplantation. Nat Med J China. 1981;61:139-44.
7. Aki FE, Besteiro JM, Pinto FR, Durazzo MD, Cunha AS, Filho GBS, et al. Emprego do retalho microcirúrgico antebraquial na reconstrução em cabeça e pescoço: experiência de 11 casos. Rev Assoc Med Bras. 2000;46(2):182-5.
8. Takushima A, Harri K, Asato H, Nakatsuka T, Kimata Y. Mandibular reconstruction using microvascular free flaps: a statistical analysis of 178 cases. Plast Reconstr Surg. 2001;108(6):1555-63.
9. Portinho CP, Sbalchiero JC, Souza THS, Ohana BM, Cardoso MM, Leal PRA. Manejo das fístulas salivares em reconstruções mandibulares microcirúrgicas. Rev Bras Cir Craniomaxilofac. 2010;13(4):236-40.
10. Portinho CP, Russano M, Sbalchiero JC, Leal PRA. Complicações da área doadora pós-fibulectomia para reconstrução microcirúrgica da mandíbula. Rev Bras Cir Craniomaxilofac. 2010;13(3):153-5.
11. Portinho CP, Ohana BMB, Sbalchiero JC, Silva e Souza TH, Leal PR, Galvão MSL, et al. Ressecção e reconstrução mandibular: análise de 85 casos. Rev Bras Cir Cabeça Pescoço. 2010;39(2):113-6.
12. Reece GP, Schusterman MA, Miller MJ, Kroll SS, Robb GL, Baldwin BJ, et al. Morbidity and functional outcome of free jejunal transfer reconstruction for circumferential defects of the pharynx and cervical esophagus. Plast Reconstr Surg. 1995;96(6):1307-16.
13. Scharpf J, Esclamado RM. Reconstruction with radial forearm flaps after ablative surgery for hypopharyngeal cancer. Head Neck. 2003;25(4):261-6.
14. Yu P, Hanasono MM, Skoracki RJ, Baumann DP, Lewin JS, Weber RS, et al. Pharyngoesophageal reconstruction with the anterolateral thigh flap after total laryngopharyngectomy. Cancer. 2010;116(7):1718-24.
1. Plastic Surgeon and Craniomaxillofacial Surgeon, Master in Medicine, Postgraduate in Reconstructive Microsurgery in Oncology, Member of the Hospital de Clínicas de Porto Alegre (HCPA), Microsurgery Specialist of the Hospital Santa Rita de Câncer/Complexo Hospitalar Santa Casa de Porto Alegre (CHSCPA), Full Member of the Associação Brasileira de Cirurgia Craniomaxilofacial (Brazilian Association of Craniomaxillofacial Surgery - ABCCMF) and of the Sociedade Brasileira de Cirurgia Plástica (Brazilian Society of Plastic Surgery - SBCP), Porto Alegre, RS, Brazil
2. Plastic Surgeon, Master in Medicine, Member of the Hospital Universitário/Universidade Federal do Rio de Janeiro (UFRJ), Preceptor of the Program of Medical Residency in Plastic Surgery at Instituto Nacional do Câncer (INCa), Full Member of the SBCP, Rio de Janeiro, RJ, Brazil
3. Plastic Surgeon, Member of the INCa, Member and Preceptor of the Program of Medical Residency at Hospital dos Servidores do Estado do Rio de Janeiro, Full Member of the SBCP, Rio de Janeiro, RJ, Brazil
4. Hand Surgeon and Orthopedist, Master in Medicine, Member and Head of the Hand Surgery Service at Hospital Cristo Redentor, Member of the Sociedade Brasileira de Ortopedia e Traumatologia (Brazilian Society of Orthopaedics and Traumatology) and Sociedade Brasileira de Cirurgia da Mão (Brazilian Society of Hand Surgery), Porto Alegre, RS, Brazil
5.Plastic Surgeon, Postgraduate in Reconstructive Microsurgery in Oncology, Associate Member of the SBCP, Rio de Janeiro, RJ, Brazil
6.Plastic Surgeon and Craniomaxillofacial Surgeon, Professor of the Department of Surgery/Universidade Federal do Rio Grande do Sul School of Medicine, Head of the Plastic Surgery Service at HCPA, Craniomaxillofacial Surgeon at Hospital São José de Neurocirurgia and CHSCPA, Full Member of ABCCMF and SBCP, Porto Alegre, RS, Brazil
Correspondence to:
Ciro Paz Portinho
Rua Mostardeiro, 5 - 1009 - Moinhos de Vento
Porto Alegre, RS, Brazil - CEP 90430-001
Email: cportinho@hotmail.com
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: 23/6/2012
Article accepted: 1/10/2012
Work performed at the Hospital das Clinicas of Porto Alegre, Porto Alegre, RS, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter