

Original Article - Year 2013 - Volume 28 -
Bilateral buccinator myomucosal flap for the treatment of velopharyngeal insufficiency: preliminary results
Uso do retalho miomucoso do músculo bucinador bilateral para o tratamento de insuficiência velofaríngea: avaliação preliminar
ABSTRACT
BACKGROUND: Velopharyngeal insufficiency in patients with cleft lip and palate is challenging for plastic surgeons. One of the treatment options is palatoplasty using buccinator muscle myomucosal flaps to extend the nasal and oral mucosa. This prospective study presents the preliminary results of the use of buccinator muscle myomucosal flaps for the treatment of velopharyngeal insufficiency in patients with cleft lip and palate.
METHODS: This study was performed between January 2010 and July 2012 at the Craniofacial Institute of the Hospital of SOBRAPAR. All patients with cleft lip and palate who had undergone nasofibroscopy and had severe velopharyngeal insufficiency with scar tissue accumulation in the oral cavity as observed by oroscopy, previous palatoplasty with total dissection of the soft palate, or were referred by other services were submitted to a secondary or in some cases, tertiary palatoplasty using a bilateral buccinator myomucosal flap to extend the soft palate.
RESULTS: Among 20 patients undergoing treatment, 4 (20%) were excluded from the analysis, 1 went from a very large to a large circular gap (5%), 6 went from a large circular gap to no gap (5%), 2 went from a medium to a small coronal gap (10%), and 3 exhibited no postoperative improvement (15%).
CONCLUSIONS: Although longer postoperative follow-up is required, the results indicate THAT buccinator muscle flaps are reliable, reproducible, and lead to good final results.
Keywords: Cleft lip. Cleft palate. Velopharyngeal insufficiency. Palatoplasty. Flap.
RESUMO
INTRODUÇÃO: A insuficiência velofaríngea em pacientes com fissura labiopalatal é um desafio para o cirurgião plástico e pode ser resolvida, entre outras maneiras, pela palatoplastia, com a utilização de retalhos miomucosos do músculo bucinador para alongar as mucosas nasal e oral. Este estudo prospectivo teve por objetivo fazer a avaliação preliminar do uso de retalho miomucoso do músculo bucinador bilateral para o tratamento da insuficiência velofaríngea em pacientes com fissura labiopalatal.
MÉTODO: Entre janeiro de 2010 e julho de 2012, todos os pacientes do Hospital SOBRAPAR - Crânio e Face com fissura labiopalatal submetidos a nasofibroscopia e que apresentavam insuficiência velofaríngea grave, grande quantidade de tecido cicatricial na oroscopia, que já haviam sido submetidos a repalatoplastias com dissecção radical da musculatura do véu e/ou encaminhados de outros serviços foram submetidos a palatoplastia secundária, por vezes terciária, com retalho miomucoso bucinador bilateral para alongamento do véu palatino.
RESULTADOS: Dos 20 pacientes submetidos à técnica proposta, 4 pacientes foram excluídos do estudo (20%), 1 paciente passou de um gap circular muito grande para grande (5%), 6 passaram de circular grande para médio (30%), 2 de circular grande para pequeno (10%), 1 de circular grande para puntiforme (5%), 1 de circular grande para ausente (5%), 2 de coronal médio para coronal pequeno (10%), e 3 mantiveram o gap circular grande no pós-operatório (15%).
CONCLUSÕES: Embora o presente estudo necessite de maior tempo de seguimento pós-operatório, os retalhos do músculo bucinador são confiáveis e de fácil reprodução, obtendo-se bons resultados.
Palavras-chave: Fenda labial. Fissura palatina. Insuficiência velofaríngea. Palatoplastia. Retalho.
The use of the buccinator myomucosal flap in patients with cleft lip and palate was first described in the 1960s by Ecker1, who termed it the buccal flap. In 1975, Kaplan2 described the use of the unilateral buccal flap associated with the reconstruction of the elevator muscle of the soft palate for the reconstruction and extension of the nasal mucosa. In 1987, Maeda et al.3 described the use of the bilateral flap, in which one of the sides is used in the reconstruction of the nasal mucosa and the other is used in the reconstruction of the oral mucosa, with formation of a sandwich flap. In 1989, Bozola et al.4 first described the detailed anatomy of the buccal flap, naming it the buccinator muscle myomucosal flap.
Since then, both uni- and bilateral buccal flaps have been extensively used to treat patients with cleft lip and palate in primary palatoplasty for the prevention of velopharyngeal insufficiency as well as in secondary palatoplasty for the treatment of velopharyngeal insufficiency3-13. Freedlander et al.14 in 1989 and Jackson et al.15 in 2004 questioned the use of the buccal flap in primary palatoplasty; however, their results show that this flap is suitable for the extension of the nasal mucosa and prevention of subsequent velopharyngeal insufficiency during recovery.
Velopharyngeal insufficiency in patients with cleft lip and palate can be corrected by plastic surgeons in different ways. The surgical procedure to be used depends on the treatment protocol and type of defect detected by nasofibroscopy. In 2009, Raposo do Amaral et al.16 described palatoplasty with total dissection of the soft palate muscle as the procedure of choice when a conservative dissection had previously been performed as a primary surgery. Meanwhile, in cases presenting with extensive scar tissue accumulation, surgery alone is not enough to correct the insufficiency.
In this context, the buccinator myomucosal flap is underutilized, probably because of insufficient knowledge concerning its anatomy, versatility, and clinical applicability in specific conditions. Even though published studies indicate that this flap is an alternative treatment deserving consideration, additional controlled clinical studies with long follow-up are required to clarify the long-term benefits of this surgical procedure5-15.
Therefore, this work presents and discusses the preliminary results of the use of the bilateral buccinator myomucosal flap for the treatment of velopharyngeal insufficiency in patients with cleft lip and palate who have undergone previous palatoplasty with intravelar veloplasty.
METHODS
A prospective study starting in January 2010 was conducted in the Service of Plastic Surgery Prof. Dr. Cassio Menezes Raposo do Amaral/The Hospital of SOBRAPAR - Craniofacial Institute (Campinas, SP, Brazil). The inclusion criteria were as follows: patients with cleft lip and cleft palate who had been subjected to nasofibroscopy and exhibited severe velopharyngeal insufficiency; patients with scar tissue accumulation detected by oroscopy (Figure 1) and who had previously undergone palatoplasty with total dissection of the soft palate; and patients referred by other services.
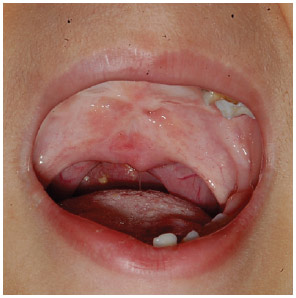
Figure 1 - Oroscopy with visible scars, which are stretched slightly vertically, in the palate transition area.
The study was conducted from January 2010 to July 2012 and included 20 patients: 9 women and 11 men. The following variables were analyzed: cleft type, determined according to the classification by Spina17; age at the time of surgery; and possible complications such as necrosis evolving to oronasal fistula. All patients underwent secondary or in some cases, tertiary palatoplasty in which a bilateral buccinator myomucosal flap was used to extend the soft palate. All procedures were performed by the author.
The exclusion criteria were as follows: patients who had fistulas corrected with the use of the buccinator flap and patients who had not yet undergone the first postoperative nasofibroscopy, meaning they were less than 3 months into recovery.
A nasofibroscopy protocol was established to evaluate the results every 3 months for 15 months postoperatively. Because of the short follow-up, the space between the soft palate and posterior wall of the pharynx (i.e., gap) was determined up to the third month, even though voice improvements have been reported to occur at least 10 months postoperatively16. Therefore, future studies will require longer follow-up.
Surgical Technique
Surgeries began with the patient in horizontal dorsal decubitus position, with orotracheal intubation and under general anesthesia. Topical anesthesia of the palate was performed using physiological 0.9% saline solution containing lidocaine and bupivacaine (2:1:1) and adrenaline (1:100,000). The topical anesthetic was administered in the palate transition areas only and never in the donor regions, thus preventing a possible decrease in flap blood supply due to edema or arterial lesion during infiltration.
An incision with a number 15 cold scalpel was made in the transition region between the hard and soft palates, and the total dissection of the soft palate including the hamulus was performed when required. The nasal mucosa was detached from the muscle, which was inserted posteriorly to the mucosa to facilitate the suture of the buccinator flap in the nasal mucosa (Figure 2).
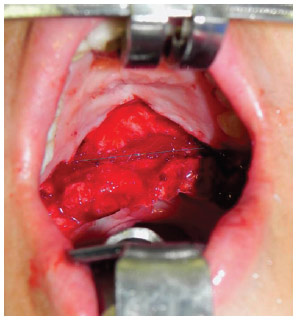
Figure 2 - Distance after total detachment of the soft palate. The gray line shows the incision in the nasal mucosa posterior to the oral mucosa.
After palate dissection, the buccinator myomucosal flaps were stained with methylene blue: the cranial end of the flap was located immediately under the termination of the Stenon's duct, whereas the caudal end was parallel to the cranial end. The distance between those 2 lines was determined by measuring the size of the defect created in the transition between the soft and hard palates. The ends of the flap incisions were V-shaped in the proximity of the lip. The flaps were dissected, and flipped towards the nasal mucosa on one side (Figure 3) and rotated towards the oral mucosa on the other side, thus forming a sandwich (Figure 4). Direct sutures were made in the mucosa of the donor regions (Figure 5).
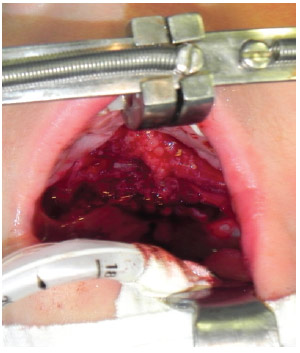
Figure 3 - Flip flap with total filling of the defect.
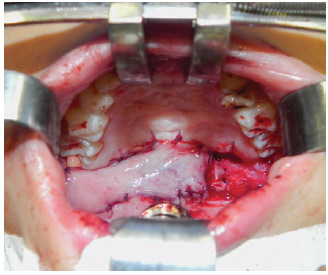
Figure 4 - Sandwich flap. The rotation flap can be observed on the left, whereas the flip flap is on the right with lateral bleeding.
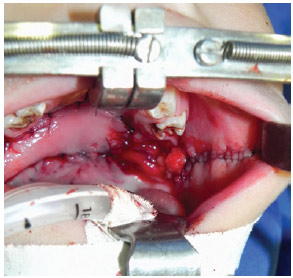
Figure 5 - Primary closure in the flap donor area without complications.
Nasofibroscopy
By allowing direct observation of the soft palate and gap between the soft palate and posterior wall of the pharynx, nasofibroscopy findings were used as an indication for surgery and oroscopy.
The patient was seated and under topical anesthesia (lidocaine spray) intraoperatively. The optical fiber was introduced in the right nostril and passed through the middle meatus to a posterior location and making an approximate right angle with the soft palate. Patients then repeated words and phrases according to the protocol established by the phonoaudiology team.
The linear distance between the soft palate and posterior wall of the pharynx was determined subjectively by 3 observers during the sustained phoneme /S/. The distance (gap) was classified by the observers as very large, large, medium, small, punctiform, or absent. The shape of the space between the soft palate and posterior wall of the pharynx was also subjectively classified as circular, coronal, or sagittal.
Size was determined according to a subjective formula that divided size during the resting of the soft palate and during the sustained /S/. The linear distance between the soft palate and posterior wall of the pharynx was divided into 5 equal parts: the decrease in the gap was classified as large, medium, and small when zero to two-fifths, two-fifths to four-fifths, and four-fifths or more without the soft palate touching the posterior wall of the pharynx, respectively. When the soft palate touched the posterior wall of the pharynx, the gap was classified as punctiform, even when air was allowed to escape. The gap was classified as very large only when the distance between the soft palate and posterior wall of the pharynx exceeded the circle observed with the optical fiber.
The team of observers included 1 member of the phonoaudiology team and 2 plastic surgeons with experience interpreting nasopharyngoscopy results as described previously16.
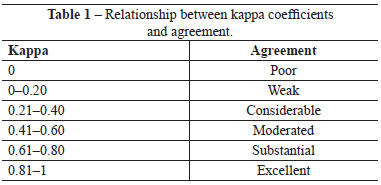
The kappa agreement coefficient was used to quantify agreement between the evaluators; it is a measure of agreement used in nominal scales that gives an idea of how much observations differ from random, indicating the legitimacy of interpretations. The relationship between kappa coefficients and agreement levels can be found in Table 1.
RESULTS
Twenty patients with cleft lip and cleft palate aged 5 to 60 years (average: 23.1 years) underwent palatoplasty with buccal flaps. According to the classification of Spina17, 7 (35%), 2 (10%), 6 (30%), and 5 (25%) patients had bilateral total foramen, right total foramen, left total foramen, and total post-foramen cleft lip and cleft palate, respectively.
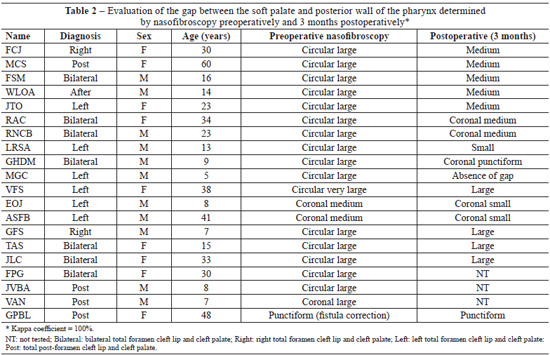
The results obtained for the gaps can be found in Table 2. In 13 cases (65%), there was improvement in the size of the gap between the soft palate and posterior wall of the pharynx; 3 patients (15%) exhibited no improvements in their large gaps. Four patients (20%) were excluded from the analysis: 1 was excluded because he underwent corrective surgery for a fistula in the palate transition region, whereas other 3 were excluded because they had not completed 3 months of postoperative follow-up at the time of this study.
In the 13 cases with gap improvement, the distance between the soft palate and posterior wall of the pharynx changed from large circular to medium circular (n = 5), circular large to coronal medium (n = 2), circular large to circular small (n = 1), circular large to circular punctiform (n = 1), circular large to absent (n = 1), coronal medium to coronal small (n = 2), and circular very large to circular large (n = 1). No sagittal gaps were observed.
The kappa coefficient between evaluators was 0.9 (90%), which is considered excellent.
Complications occurred in only 1 patient, who developed flap necrosis and a fistula in the palate transition region; they were successfully corrected with a tongue flap 15 months postoperatively. Another 3 patients reported pain associated with the oral mucosa flaps; however, the evolution was positive and no fistulas developed.
DISCUSSION
Successful palatoplasty with ideal speech results depends on the surgical closure of the soft palate without tension and with soft palate extension and muscle reconstruction. However, this is not always possible, particularly in developing countries such as Brazil where patients reach treatment centers past the ideal age for such procedures or following previous unsuccessful surgeries. Therefore, secondary and even tertiary procedures are often required.
The type of procedure chosen for the treatment of velopharyngeal insufficiency varies among centers where cleft lip and palate surgeries are performed. However, when patients have a large gap between the soft palate and posterior wall of the pharynx, repositioning the muscle of the soft palate or performing pharyngoplasty is insufficient for complete treatment. A tongue flap can be performed in some cases, but patients can consequently develop snoring and sleep apnea, which can even lead to sudden death. Because of the possible complications associated with this technique, which was first described in the 1960s18,19, it is used as a last resort in this plastic surgery service.
Palatoplasty performed with buccinator muscle myomucosal flaps for palate extension is an effective technique, particularly in cases with extensive scarring. Bozola et al.4 report that this procedure can extend the palate up to 2.5 cm in adults.
This study did not include the measurement of soft palate extension. However, sufficient extension was achieved in most cases (Figure 6), with a concomitant decrease in the gap as shown by nasofibroscopy. A better final result concerning soft palate extension can usually be observed from the 10th month postoperatively16. Therefore, future studies should include longer follow-up. Complementary studies should also include voice examinations, because articulation errors that occur in patients with cleft palate and velopharyngeal insufficiency play a role in the final results concerning the voice of the patients.
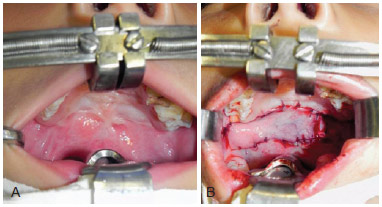
Figure 6 - In A, preoperatively. In B, postoperatively with extension of the soft palate and scar tissue clearance.
There were no improvements 3 months postoperatively in 2 cases, probably because of the large amount of scar tissue, which led to total immobilization of the soft palate as shown by nasofibroscopy. In contrast, the best results were obtained for patients with greater mobility of the soft palate as detected by preoperative nasofibroscopy.
The shape of the gap between the soft palate and posterior wall of the pharynx can vary: it can be circular when the contraction is similar between the soft palate, and lateral and posterior walls of the pharynx (i.e., when the Passavant ring is present); coronal when the soft palate has greater mobility than the walls; sagittal when the lateral walls have greater mobility than the other walls; and punctiform when the soft palate leans on the posterior wall of the pharynx, but air can still escape during the sustained /S/. Therefore, not only a decrease in the gap, but also changes in the shape from circular to coronal in 2 cases indicate that the soft palate became more mobile postoperatively.
Although the results obtained by nasofibroscopy are subjective, they can be considered valid and valuable in the context of the literature16,20. The present study employed 3 experienced and independent evaluators to diminish the subjectivity associated with the examination. The gaps were determined during the sustained /S/, as this is the phoneme that brings the soft palate closer to the posterior wall of the pharynx; if the gap decreases or becomes absent following surgery, then the insufficiency can be regarded as a lack of ability, and speech therapy will be essential for voice improvements.
When possible, the treatment of velopharyngeal insufficiency has always been one of the major aims of plastic surgery offered to patients with cleft palate. Successful treatment allows the possibility of voice and speech improvements and consequently improvements in communication, which is one of the most important factors associated with the psychosocial integration of an individual.
CONCLUSIONS
The treatment of velopharyngeal insufficiency in patients with cleft lip and palate is challenging for plastic surgeons. Multiple procedures are required in most cases. However, in cases in which there is a large gap between the soft palate and posterior wall of the pharynx, all surgical procedures that allow palate extension can be performed, including the use of buccinator myomucosal flaps, thus allowing the best possible results.
Even though this prospective study may have benefited from longer follow-up, the results indicate that buccinator flaps are reliable, reproducible, and lead to good results.
REFERENCES
1. Ecker HA. The use of the buccal flap in cleft palate repair. Plast Reconstr Surg Transplant Bull. 1960;25:235-9.
2. Kaplan EN. Soft palate repair by levator muscle reconstruction and a buccal mucosal flap. Plast Reconstr Surg. 1975;56(2):129-36.
3. Maeda K, Ojimi H, Utsugi R, Ando S. A T-shaped musculomucosal buccal flap method for cleft palate surgery. Plast Reconstr Surg. 1987;79(6):888-96.
4. Bozola AR, Gasques JA, Carriquiry CE, Cardoso de Oliveira M. The buccinator musculomucosal flap: anatomic study and clinical application. Plast Reconstr Surg. 1989;84(2):250-7.
5. Jackson IT, McLennan G, Scheker LR. Primary veloplasty or primary palatoplasty: some preliminary findings. Plast Reconstr Surg. 1983;72(2):153-7.
6. Zöller J, Maier H. The primary reconstruction of soft palate by intraoral buccal transposition flap. Dtsch Stomatol. 1991;41(8):287-90.
7. Nakakita N, Maeda K, Ojimi H, Utsugi R, Maekawa J. The modified buccal musculomucosal flap method for cleft palate surgery. Plast Reconstr Surg. 1991;88(3):421-6.
8. Mann RJ, Fisher DM. Bilateral buccal flaps with double opposing Z-plasty for wider palatal clefts. Plast Reconstr Surg. 1997;100(5):1139-43.
9. Chen GF, Zhong LP. A bilateral musculomucosal buccal flap method for cleft palate surgery. J Oral Maxillofac Surg. 2003;61(12):1399-404.
10. De Riu G, Meloni SM, Bozzo C, Meloni F, Tullio A. A double buccal fat pad flap for middle palate defect closure: a new technique for palate closure. Int J Oral Maxillofac Surg. 2006;35(11):1057-9.
11. Robertson AG, McKeown DJ, Bello-Rojas G, Chang YJ, Rogers A, Beal BJ, et al. Use of buccal myomucosal flap in secondary cleft palate repair. Plast Reconstr Surg. 2008;122(3):910-7.
12. Ilango N. Use of the buccal fat pad flap for congenital cleft palate repair. Plast Reconstr Surg. 2009;124(2):668.
13. Mann RJ, Neaman NK, Armstrong SD, Ebner B, Bajnrauh R, Naum S. The double-opposing buccal flap procedure for palatal lengthening. Plast Reconstr Surg. 2011;127(6):2413-8.
14. Freedlander E, Jackson IT. The fate of buccal mucosal flaps in primary palatal repair. Cleft Palate J. 1989;26(2):110-2.
15. Jackson IT, Moreira-Gonzalez AA, Rogers A, Beal BJ. The buccal flap: a useful technique in cleft palate repair? Cleft Palate Craniofac J. 2004;41(2):144-51.
16. Raposo do Amaral CA, Sabbag A, Ferreira LA, Almeida AB, Buzzo CL, Raposo do Amaral CE. Dissecção radical da musculatura do véu palatino em casos secundários de pacientes fissurados. Rev Bras Cir Plást. 2009;24(4):432-6.
17. Spina V. A proposed modification for the classification of cleft lip and cleft palate. Cleft Palate J. 1973;10:251-2.
18. Nylén B, Wáhlin A. Post-operative complications in pharyngeal flap surgery. Cleft Palate J. 1966;3:347-56.
19. Graham WP 3rd, Hamilton R, Randall P, Winchester R, Stool S. Complications following posterior pharyngeal flap surgery. Cleft Palate J. 1973;10:176-80.
20. Ngim RC, Chua E, Lee ST. Speech and nasendoscopic evaluation of velopharyngeal incompetence (VPI) in cleft palate patients. Ann Acad Med Singapore. 1988;17(3):380-3.
Plastic surgeon, Chief of Residents of the Service of Plastic Surgery Prof. Dr. Cassio Menezes Raposo do Amaral/The Hospital of SOBRAPAR - Craniofacial Institute, Member of the Brazilian Society of Plastic Surgery, Campinas, SP, Brazil
Correspondence to:
Cesar Augusto Raposo-do-Amaral
Alameda das Palmeiras, 25 - Gramado
Campinas, SP, Brazil - CEP 13101-676
E-mail: cesaraugusto@sobrapar.org.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Article received: 26/7/2012
Articleaccepted: 15/12/2013
Work performed at the Service of Plastic Surgery Prof. Dr. Cassio Menezes Raposo do Amaral/The Hospital of SOBRAPAR - Craniofacial Institute, Campinas, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter