

Reviw Article - Year 2014 - Volume 29 -
Functional nutrition in postoperative plastic surgery: focus on seroma and fibrosis prevention
Nutrição funcional no pós-operatório de cirurgia plástica: enfoque na prevenção de seroma e fibrose
ABSTRACT
INTRODUCTION: Based on the wide range of possible consequences and complications of plastic surgery and the role of nutrition in the recovery process of these, we see the need for therapeutic practices to ensure effectiveness of the final aesthetic result.
METHOD: This study is characterized as a non-systematic review of the possible associated nutrients, bioactive compounds and herbal medicines to prevent seroma and fibrosis. Thus, we used scientific journal articles from electronic media, national laws and textbooks published between 2002 and 2012.
RESULTS: We demonstrated that the main causes of seroma and fibrosis are changed healing, inflammation, oxidative stress and edema. Thus, we designed a practical guide with nutrients, bioactive compounds and herbal medicines that can be used to prevent, control or mitigate the complications after plastic surgery.
CONCLUSION: It was emphasized that for the treatment of covered conditions is essential that there is a multidisciplinary approach, emphasizing a medical, nutritional and therapeutic monitoring covering the known changes in the pathophysiology of seroma and fibrosis.
Keywords: Seroma; Fibrosis; Nutrition; Functional food; Phytotherapy.
RESUMO
INTRODUÇÃO: Com base na grande variedade de possíveis sequelas e intercorrências de cirurgias estéticas e no papel da nutrição no processo de recuperação dessas, percebe-se a necessidade de práticas terapêuticas que garantam eficácia do resultado estético final.
MÉTODO: O presente estudo caracteriza-se como uma revisão não sistemática que associou possíveis nutrientes, compostos bioativos e fitoterápicos que previnam ou amenizem seroma e fibrose no pós operatório. Para tanto, foram utilizados artigos de revistas científicas do meio eletrônico, legislações nacionais e livros didáticos, publicados entre os anos de 2002 e 2012.
RESULTADOS: Demonstrou-se que as principais causas de seroma e fibrose são cicatrização alterada, inflamação, estresse oxidativo e edema. Dessa forma, foi elaborado um guia prático com os nutrientes, compostos bioativos e fitoterápicos que podem ser utilizados a fim de prevenir, controlar ou amenizar as complicações no pós-cirúrgico de procedimentos estéticos.
CONCLUSÃO: Salientou-se que para o tratamento das condições abordadas é fundamental que haja um trabalho multiprofissional, enfatizando um acompanhamento médico, nutricional e fisioterápico que abranja as alterações conhecidas na fisiopatologia de seroma e fibrose.
Palavras-chave: Seroma; Fibrose; Nutrição; Alimento funcional; Fitoterapia.
The Brazilian Society of Plastic Surgery showed that a substantial number of plastic surgeries, both aesthetic and reparative, were performed in the last years. The number of interventions positions the country in the second place in plastic surgery, next only to the United States, where approximately 800,000 procedures are done every year1.
In Brazil, every 3 years, >1,000,000 aesthetic surgeries are performed. However, the efficiency of plastic surgery does not depend solely on the surgical planning. Both pre- and postoperative care have been highlighted as important factors in preventing complications and promoting a more satisfactory aesthetic result1,2.
Concerning complications, the local ones, such as edema, seroma, bruising, hematoma, fibrosis, and necrosis of the dermofat flap, are the most frequent; however, although rare, systemic complications such as deep vein thrombosis and pulmonary embolism also occur3.
Despite the popularity of aesthetic surgery, there is a need for technical improvements in order to enhance the postoperative physical aspect and reduce complications. A number of risk factors have been proposed, including previous abdominal surgery, smoking, hypertension, and obesity4.
The risk factors mentioned above could lead to nutritional deficits, which, in turn, could more easily be reverted to reduce the susceptibility to complications and impaired healing. Furthermore, the expectations from the patients may exceed the goals and experience of the surgeon, increasing the need to optimize aesthetic results with the help of practices alternative to surgery5.
For this reason and with the goal of improving the effects of the surgical intervention, it is essential to evaluate the nutritional status of the patient and plan the intake of macro- and micronutrients in the interval between the perioperative and postoperative periods6.
On the basis of the growing number of aesthetic surgeries in Brazil; the wide variety of possible consequences and complications from these operations; the role of nutrition in the healing process, inflammation, and immunity; the need for multidisciplinary work; and, primarily, the lack of studies addressing these topics, the present study is important in order to improve the recovery, quality of life, and self-esteem of patients, in addition to the effectiveness of the final aesthetic result1,3.
Therefore, in this study, we aimed to perform a literature review on the causes of seroma and fibrosis, including the possible nutrients, bioactive compounds, and phytotherapeutics that can prevent or mitigate these complications after plastic surgery, and to organize the knowledge acquired in a practical guide to assist in its clinical application.
METHODS
This study is a nonsystematic review of the literature with data from electronically available scientific journal articles, national legislation, and textbooks at the Universidade do Vale do Itajaí (UNIVALI) that have been published between 2002 and 2012.
In searching for scientific articles, the following databases were consulted: Google Scholar, Medline (United States National Library of Medicine), Latin American and Caribbean Health Sciences (LILACS), Science Direct, PubMed, and Scielo. The following Portuguese keywords and their English equivalents were searched: "complicações" (complications), "pós-operatório" (postoperative), "fibrose" (fibrosis), "seroma" (seroma), "cicatrização" (healing), "colágeno" (collagen), "inflamação" (inflammation), "sistema imune" (immune system), "estresse oxidativo" (oxidative stress), "edema" (edema), "alimentação" (food), "nutrientes" (nutrients), and "nutrição funcional" (functional nutrition).
Seroma and Fibrosis
Seroma has a prevalence of between 1% and 57%, with the most frequent values being between 10% and 15% after an abdominoplasty3,7. In Brazil, studies on this surgical procedure indicate highly variable percentages, ranging from 1.8% to 30%8-11.
Seroma is characterized by an accumulation of serous fluid due to leakage of plasma and lymph deep in the dermofat flap, displacement of the abdominal flap, section of blood and lymph vessels, disruption of lymphatic channels, an augmented inflammatory and healing process, and an increase in fibrinolytic activity3,7,12.
The primary physiopathology of seroma is poorly elucidated and remains controversial. Nevertheless, it is known to cause bulging and fluctuation of the site, leading to discomfort, dissatisfaction, predisposition to morbidities (necrosis, dehiscence, and sepsis), and delayed healing, recovery, adjuvant therapy, and hospital discharge. The treatment consists in puncture, drainage, and medication13,14.
The patient's age, arterial hypertension, use of heparin, high body mass index, low concentration of protein and albumin, and high serum concentration of interleukin (IL)-1-RA have been recently reported to be linked to a higher risk of postoperative seroma formation15. The treatment strategies are based on medication, including antibiotics, anti-inflammatory drugs, analgesics, and diuretics, as well as on aspiration, puncture, and drainage, and reconstructive surgery16.
On the other hand, fibrosis occurs in the lipoaspirated tissue as a repair mechanism; however, it is characterized by the formation or abnormal development of excess fibrous connective tissue composed of elastin and collagen, resulting in hard or nodular lesions17. In a retrospective analysis covering 25 years in the databases of 26,259 health-care services of four surgeons, the prevalence of fibrosis was of 2.3%18.
It should be emphasized that this complication, which may occur especially after liposuction, has a genetic cause induced by enzymatic deficiencies or pathological processes, and that the blood and lymph circulation efficiency is an important factor in the healing process, in acute trauma, or in chronic inflammation19. Furthermore, fibrosis formation is mediated by the interaction between growth factors and pro-fibrotic cytokines, as well as by the influence of these mechanisms on the extracellular matrix, mechanical tension, and oxidative stress20.
Treatment of the condition is important to avoid future deformities. Treatment needs to be initiated early in collagen synthesis, which increases between the 6th and 17th day; after the 42nd day, this process ceases and remodeling of the accumulated collagen occurs21.
In view of the possible causes of seroma and fibrosis, which include healing, collagen formation, inflammation, immune dysfunction, oxidative stress, and edema, it becomes necessary to understand the reactions involved in the above conditions and correlate them with functional nutrition. Figure 1 shows the metabolic network of the interrelation of the factors contributing to the formation of seroma and fibrosis.
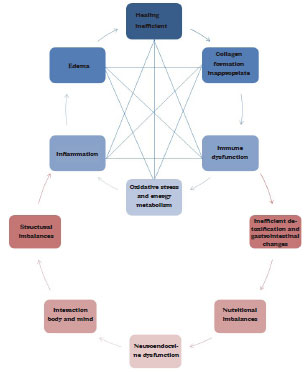
Figure 1 - Metabolic network of the interrelation between seroma and fibrosis. Adapted from Vasquez22.
FACTORS CONTRIBUTING TO SEROMA AND FIBROSIS FORMATION
Healing and collagen formation
Healing is a dynamic and immediate process of tissue repair in response to an injury, with the aim of restoring the anatomical, structural, and functional characteristics of the patient. This recovery comprises three phases that occur simultaneously. The inflammatory phase, which is dependent on vitamin K, has a duration of 4-6 days and includes hemostasis, phagocytosis, and cell migration. From the third day up to weeks after, the phase known as proliferative, granulation, or fibroplasia phase occurs, where the requirements for carbohydrates, protein, lipids, vitamins A, C, and B complex, iron, zinc, and magnesium increase to promote cell proliferation, collagen synthesis, and neovascularization. Finally, the maturation phase or remodeling can extend up to 2 years, which involves stabilization of collagen and an increase in the resistance of the scar23. Figure 2 shows the healing phases, predominant cell types, and vascular response24.
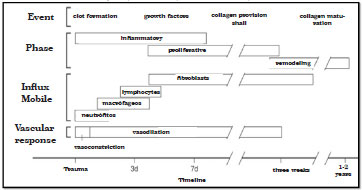
Figure 2 - Healing phases, predominant cell types, and vascular response.
Source: Mathes.24.
Under this system, nutrition is considered a major factor as many nutrients can influence the healing phases because of their involvement in the synthesis of new tissues, suppression of oxidation, healing optimization, and also in immunocompetence25.
Concerning the nutritional factors, it is observed that decreases in amino acids, nucleic acids, or any cofactors involved in the repair process is significantly harmful. Preoperative malnutrition and especially protein deficiency and DNA dysfunctions interfere with collagen synthesis, fibroblast proliferation, decrease in angiogenesis, and reduction in proteoglycans. Furthermore, carbohydrate deficiency leads to protein catabolism and, together with the deficit in vitamins and minerals, such as vitamin A, thiamine, vitamin C, and zinc, worsens the clinical condition26.
Inflammation and immune system
Inflammation is the first stage of the healing process, and is closely linked to oxidative stress and reduced antioxidant capacity. However, when inflammation becomes chronic, it is characterized by a prolonged inflammatory response and tissue destruction. Many inflammatory cells secrete cytokines such as TGF-1 and IL-13, which are fibrinogenic20.
The inflammatory phase comprises the influx of neutrophils, macrophages, and lymphocytes to the lesion site, as well as vasoconstriction, platelet aggregation, and increase in vascular permeability; the proliferative phase involves granulation, the influx of fibroblasts and keratinocytes, re-epithelialization, capillary formation, and extracellular matrix production; and the final stage (or remodeling) is dependent on the balance between the synthesis and degradation of collagen27.
Thus, age, nutritional status, chronic diseases, caloric intake, cortisol release, stress, pain, and anesthesia affect the body's immune function, and can negatively interfere in the defense mechanism of the patient. Deficiencies in nutrients such as zinc, selenium, and vitamin B6 alter the immunity and protection of the host, increasing the risk for infections in the postoperative period28.
In addition to those nutrients, Mitchell et al.29 cite vitamins A, C, and E as important owing to their antioxidant functions and their role in cellular metabolism; however, only vitamin E has been proven to strengthen the immune system, as the scientific proof is poor or inconsistent for the other nutrients. However, the authors emphasize that high doses of vitamins B1, B2, B6, folate, and niacin may impair immunology; moreover, supplementation of >800 mg/day of vitamin E seems to have no benefits, whereas that of >100 mg/day of zinc has an effect on lymphocyte proliferation.
Oxidative stress
The process of ischemia-reperfusion during plastic surgery creates a state of oxidative stress, which is characterized by an increase in reactive oxygen species (ROS) released by damaged tissues and inflammatory cells, and is interrelated with the formation of fibrosis. Excess ROS damages cellular components such as proteins, lipids, and nucleic acids, creating an oxidant-antioxidant imbalance, which is represented by an increase in NADPH oxidase, activation of cytochrome c and xanthine oxidase, dysfunctions in mitochondrial respiration, and suppression of superoxide dismutase20.
Anesthetic agents are a considerable source of oxidation, causing the formation of reactive oxygen, which inflicts damage to tissues and the healing process. The excess of free radicals, as mentioned, has several harmful effects, such as immunity suppression, altered cellular function, increase in lipid peroxidation, and inadequate interaction of the nutrients that form collagen, which, in turn, causes loss of tissue flexibility30.
In parallel, oxidative stress during and after the surgical procedure involves inflammatory, endocrine, and immunological activation, characterized by the massive production of cytokines (such as IL-1, IL-2, IL-6, and IL-8) that are responsible for the progression and amplification of the immune response, and the activation of macrophages, platelets, and mast cells, which form free radicals, becoming a vicious cycle31.
Top part of the form
According to Ratnam and colleagues32, the human antioxidant defense system is not complete without the dietary antioxidants, which confirms the importance of the daily intake of these compounds.
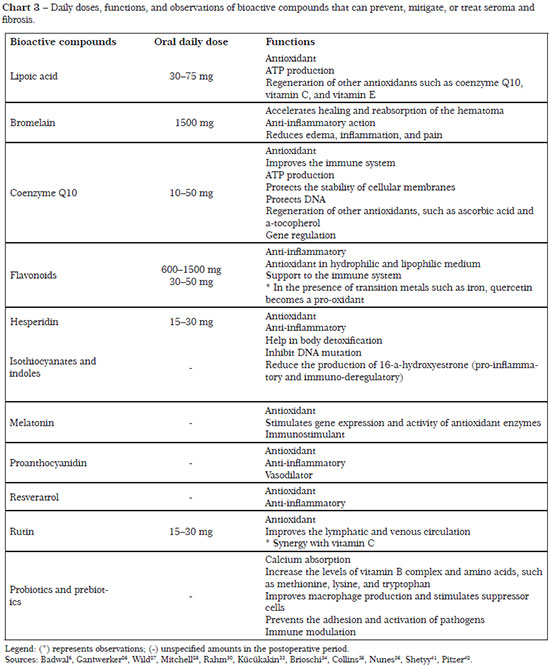
In this context, antioxidants (carotenoids, vitamins A and C, selenium, and phenolic compounds) can neutralize the free radicals resulting from the surgical procedure, playing an important role in the prevention of additional damage. Some enzymes also have an antioxidant function, such as catalase, superoxide dismutase, and glutathione peroxidase, although there are nutrients that act as cofactors for their proper functioning. Even though there is a clear need for supplementation of antioxidant nutrients and enzyme cofactors, this is not yet clearly elucidated in the literature. Commonly, a combination of nutrients in low doses is prescribed instead of mega-doses, solely to supplement the blood and tissue losses after the surgery28,33.
Edema
Edema is generated by inflammation through the increase in vascular permeability of a noninfectious exudate that leaks into the interstitial space, providing a humid layer that contains growth factors crucial for wound healing and facilitating the infiltration of inflammatory cells in the lesion; however, when the edema is excessive, it allows the development of fibrosis and seroma26.
Other contributing factors
On the basis of the metabolic network of interrelations shown in Figure 1, it can be stated that several organic imbalances contribute to the formation of seroma and/or fibrosis; this is based on the concept that functional nutrition has a multidisciplinary coverage focused on gene expression and biochemical individuality34. In this context, it is easy to understand that structural dysfunctions may contribute to inflammation and oxidative stress. Problems of the body/mind interaction, such as depression, anxiety, and psycho-emotional stress, lead to inflammation and a decline in immune system functions, in addition to intestinal hyperpermeability and urinary excretion of nutrients. Problems in detoxification, in turn, overload the body, initiating a pro-inflammatory process; finally, intestinal dysbiosis impairs food digestion and absorption, producing algogenic and inflammatory substances capable of activating the immune system22.
Role of functional nutrition
Functional nutrition has the goal of improving the quality of life, health, and well-being of patients, beyond the basic nutritional functions of food. For this purpose, initially the mediators of the symptoms presented by the patient and corresponding to each system of the metabolic interrelation network are identified. Then, strategies to inhibit or modulate the triggers are then chosen in order to reestablish the balance of the systems, allowing a multidimensional approach and a higher therapeutic efficacy34,35.
As there is an increase in nutritional needs in hypermetabolism and organic disorders, the nutrient requirements are often not satisfied with traditional food, and supplements and/or fortified food become necessary. Therefore, a wide variety of nutritional supplements are available to improve the triggers of seroma and fibrosis25.
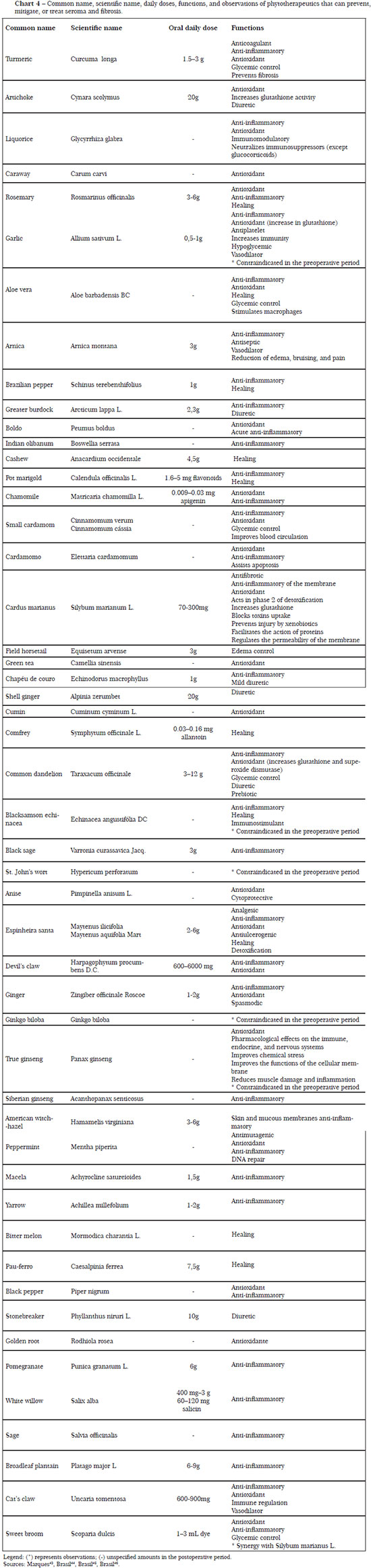
According to Rahm30, nutritional supplementation during the pre- and postoperative periods can have a significant impact on the surgical outcome, reducing hematomas, edema, and inflammation, and promoting proper wound healing, besides increasing immunity and reducing oxidative stress. In this way, when addressing the nutritional status and providing guidance focused on supplementation, the aesthetic surgeon or nutritionist can positively influence the prevention of postoperative complications.
However, supplementation should be specific to the nutritional deficiency, with the main source being food intake and avoiding administering a supplementation amount that surpasses pharmacological values and generates adverse effects. In this context, the use of supplements that cause prolonged bleeding, drug or anesthetic interaction, and cardiovascular disturbances should be suspended for 2 weeks before and 1 week after the surgical procedure27.
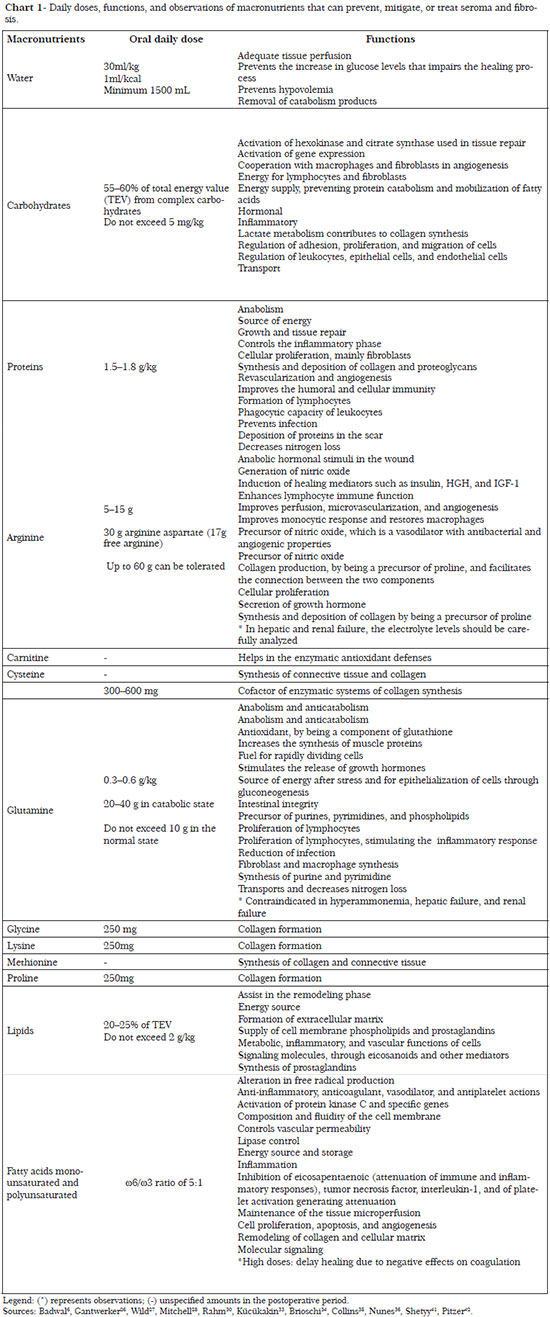
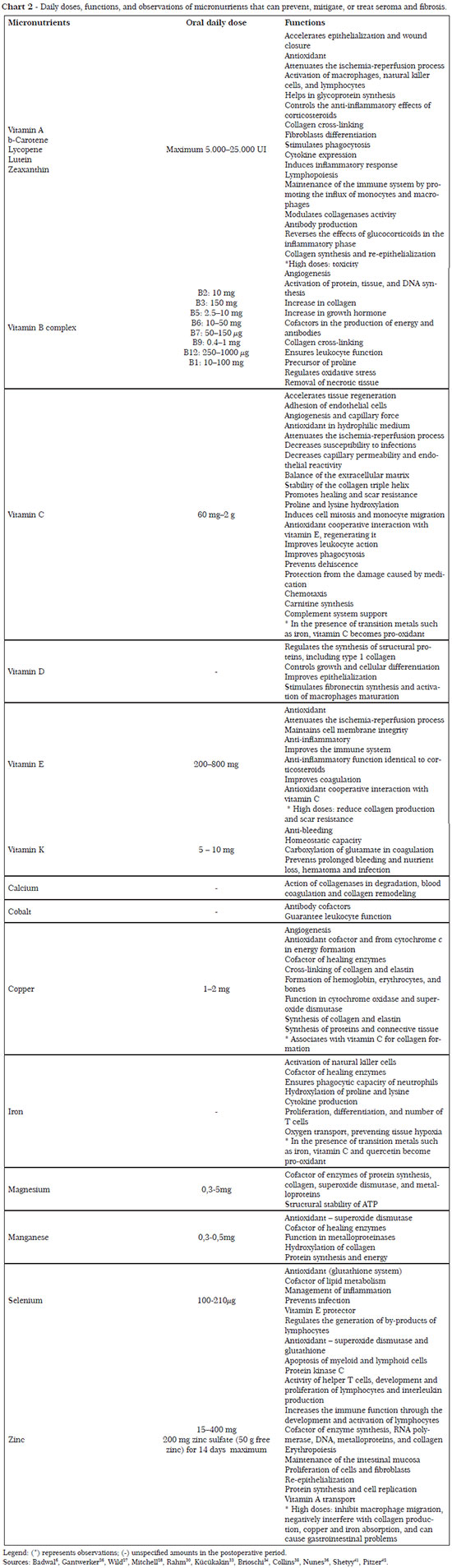
The necessary nutrients, as well as their daily doses and functions for the regulation of processes linked to prevention of seroma and fibrosis, are described in Charts 1 to 4.
CONCLUSION
The results of the research performed show that seroma and fibrosis are caused by exacerbated and interrelated physiological alterations, such as the healing process, collagen formation, inflammation, immune system changes, oxidative stress, and edema, and that there are several components of functional food, such as nutrient, bioactive, and phytotherapeutic compounds, that are well documented and can be used to prevent, control, or mitigate complications in the postoperative period after plastic surgery. In this sense, it is essential that the nutritional and metabolic imbalances are recognized and corrected by the professional, who is ethically committed to maintain the functional and aesthetic well-being of the patient. This correction is primarily based on food ingestion and secondarily on supplementation, taking into account, however, the contraindications and possible adverse effects. It is important to highlight that for the treatment seroma and fibrosis, a multidisciplinary approach is crucial, with emphasis on a clinical, nutritional, and physiotherapeutic monitoring that covers all changes known to be related to the physiopathology of these conditions.
In addition, taking into account the complexity of the topic, more studies are required concerning the influence of food consumption with the goal of improving the surgical results, quality of life, and aesthetic outcomes. We suggest the use of other scientifically valid methods for the assessment of the complications mentioned in this work, aiming at results that include all the variables involved in the diagnosis, prevention, and treatment of seroma and fibrosis.
REFERENCES
1. Ferreira FR. Cirurgias estéticas, discurso médico e saúde. Ciência & Saúde Coletiva. 2011;16(5):2373-82.
2. Flores A, Brum KO, Carvalho RM. Análise descritiva do encaminhamento médico a tratamentos fisioterapêuticos dermato-funcionais nos períodos pré e pós-operatório de cirurgias plásticas cosméticas. O mundo da saúde. 2011;35(4):408-14.
3. Di Martino M, Nahas FX, Novo NF, Kimura AK, Ferreira LM. Seroma em lipoabdominoplastia e abdominoplastia: estudo ultrassonográfico comparativo. Revista Brasileira de Cirurgia Plástica. 2010;25(4):679-87.
4. Momeni A, Heier M, Bannasch H, Stark B. Complications in abdominoplasty: a risk factor analysis. Journal of Plastic, Reconstructive & Aesthetic Surgery. 2009;62(10):1250-54.
5. Rahm D. Perioperative nutrition and nutritional supplements. Plastic SurgicalNursing. 2005;25(1):21-8.
6. Badwal RS, Bennett J. Nutritional considerations in the surgical patient. The Dental Clinics of North America. 2003;47(2):373-93.
7. Hafezi F, Nouhi AH. Abdominoplasty and seroma. Ann Plast Surg. 2002;48(1):109-10.
8. Arantes HL, Rosique RG, Rosique, MJF, Mélega JM. Há necessidade de drenos para prevenir seroma em abdominoplastias com pontos de adesão? Revista Brasileira de Cirurgia Plástica.2009;24(4):521-524.
9. Nurkim MV, Mendonça LB, Martins PAM, Silva JLB, Martins PDE. Incidência de Hematoma e Seroma em Abdominoplastia com e sem uso de Drenos. Rev. Soe. Bras. CiroPlást. 2002;17(l):69-74.
10. Silva RF, Arantes HL, Maciel PJ, Pinheiro AS. Comparação entre drenagem aspirativa e pontos de adesão na prevenção de seroma em pós-operatório de abdominoplastia associada à lipoaspiração. 2011;26(3):74-74
11. Baroudi R, Ferreira CA. Seroma: how to avoid it and how to treat it. Aesthet Surg J.1998;18(6):439-41.
12. Oliveira EA, Valera F, Monte ALR, López C. Prevenção do seroma nas abdominoplastias associadas à lipoaspiração e sem drenagem ativa. Revista da Sociedade Brasileira Cirurgia Plástica. 2008;23(1):41-47.
13. Hashemi E, Kaviani A, Najafi M, Ebrahimi M, Hooshman H, Montazeri A. Seroma formation after surgery for breast câncer. World Journal of Surgical Oncology. 2004;2(44):1-5.
14. Kuroi K, Shimozuma K, Taguchi T, Imai H, Yamashiro H, Ohsumi S et al. Evidence-Based Risk Factors for Seroma Formation in Breast Surgery.Japanese Journal of Clinical Oncology. 2006;36(4):197-206.
15. Al-Gaithy ZKA, Ayuob NN. Vascular and cellular events in post-mastectomy seroma: an immunohistochemical study. Cellular Immunology. 2012:272(2):130-6.
16. Shermak MA, Rotellini-Coltvet LA, Chang D. Seroma development following body contouring surgery for massive weight loss: patient risk factors and treatment strategies. Plastic and Reconstrutive Surgery.2008;122(1):280-8.
17. Guirro E, Guirro R. Fisioterapia em estética: fundamentos, recursos e patologias. São Paulo: Editora Manole; 2002.
18. Triana L, Triana C, Barbato C, Zambrano M. Liposuction: 25 Years of Experience in 26,259 Patients Using Different Devices. Aesthetic Surgery Journal. 2009;29(6):509-12.
19. Schwuchow LS, Souza VP, Pellini E, Caloy L, Resende TL. Estudo do uso da drenagem linfática manual no pós-operatório da lipoaspiração de tronco em mulheres. Revista da Graduação. 2008;1(1):1-9.
20. Mu X, Bellayr IH, Walters TJ, Li Y. Mediators leading to fibrosis - how to measure and control them in tissue engineering. Operative Techniques in Orthopaedics. 2010;20(2):110-18.
21. Altomare M, Machado B. Cirurgia plástica: terapêutica pré e pós. São Paulo: Fontes; 2006.
22. Vasquez A. Web-like interconnections of physiological factors. Integrative Medicine 2006;5(2):32-7.
23. Bottoni A, Bottoni A, Rodrigues RC, Celano RMG. Papel da nutrição na cicatrização. Revista Ciências em Saúde. 2011;1(1):1-5.
24. Mathes SJ. Plastic Surgery. In: Ono MCC. Influência da dieta imunomoduladora na cicatrização cutânea em ratos [Dissertação]. Curitiba:Universidade Federal do Paraná, Pós-Graduação em Clínica Cirúrgica;2009. 136 p.
25. Dias CA. Nutrição e Cicatrização de Feridas - Suplementação Nutricional? [Monografia]. Porto:Universidade do Porto, Faculdade de Ciências da Nutrição e Alimentação;2009. 61p.
26. Gantwerker EA, Hom DB. Skin: Histology and Physiology of Wound Healing. Facial Plast Surg Clin North Am.2011;19(3):441-53.
27. Wild T, Rahbarnia A, Kellner M, Sobotka L, Eberlein T. Basics in nutrition and wound healing. Nutrition. 2010;26(9):862-6.
28. Rahm D, Labovitz JM. Perioperative Nutrition and the Use of Nutritional Supplements. Clin Podiatr Med Surg. 2007;24(2):245-59.
29. Mitchell BL, Ulrich CM, McTiernan A. Supplementation with vitamins or minerals and immune function: can the elderly benefit? Nutrition Research. 2003;23(8):1117-39.
30. Rahm D. A Guide to Perioperative Nutrition. Aesthetic Surgery Journal. 2004;24(4):385-90.
31. Gitto E, Reiter RJ, Cordaro SP, et al. Oxidative and inflammatory parameters in respiratory distress syndrome of preterm newborns: Beneficial effects of melatonin. Am J Perinatol. 2004;21(4):209-16.
32. Ratnam D, Ankola D, Bhardwaj V, Sahana D, Kumar M. Role of antioxidants in prophylaxis and therapy: A pharmaceutical perspective. J. Control Release. 2006;113(2):189-207.
33. Kücükakin B, Gögenur L, Reiter RJ, Rosenberg J. Oxidative Stress in Relation to Surgery: Is There a Role for the Antioxidant Melatonin? Journal of Surgical Research. 2009;152(2):338-47.
34. Brioschi EF, Brioschi ML, Yeng LT, Teixeira MJ. Nutrição funcional no paciente com dor crônica. Rev Dor. 2009;10(3):276-85.
35. Henry CJ. Functional foods. Eur. J. Clin. Nutr. 2010;64(1):657-659.
36. Demling RH. Nutrition, Anabolism, and the Wound Healing Process: An Overview. Eplasty. 2009;9(1):65-94.
37. Rowe DJ, Baker AC. Perioperative Risks and Benefits of Herbal Supplements in Aesthetic Surgery. Aesthetic Surgery Journal. 2009;29(2):150-57.
38. Williams JZ, Barbul A. Nutrition and Wound Healing. Critical care nursing clinics of North America. 2012;24(2):179-200.
39. Collins CE, Kershaw J, Brockington S. Effect of nutritional supplements on wound healing in home-nursed elderly: A randomized Trial. Nutrition. 2005;21(2):147-55.
40. Nunes LAS, Silva FO, Ceglio AC, Venturini TS. Efeitos da Suplementação com Panax ginseng Sobre Parâmetros de Estresse Oxidativo e Lesão Muscular Induzidos por Exercício Exaustivo Agudo em Ratos. Revista Ciências em Saúde. 2011;1(1):1-8.
41. Shetyy V, Schwartz HC. Wound Healing and Perioperative Care. Oral Maxillofacial Surg Clin N Am. 2006;18(1):107-13.
42. Pitzer GB, Patel KP. Proper care of early wounds to optimize healing and prevent complications. Facial Plast Surg Clin N Am. 2011;19(3):491-504.
43. Marques N. Nutrição clínica funcional: Fitoterapia. São Paulo:VP Editora; 2011.
44. Brasil. Agência Nacional de Vigilância Sanitária. Formulário de Fitoterápicos da Farmacopéia Brasileira / Agência Nacional de Vigilância Sanitária. Brasília: Anvisa, 2011. 126p.
45. Brasil. Agência Nacional de Vigilância Sanitária. Instrução Normativa nº 5, de 11 de dezembro de 2008. Determina a publicação da "Lista de medicamentos fitoterápicos de registro simplificado". Diário Oficial da União, Poder Executivo, Brasília, DF, 12 dez. 2008.
46. Brasil. Agência Nacional de Vigilância Sanitária. Resolução de Diretoria Colegiada nº 10 de 10 de março de 2010. Dispõe sobre a notificação de drogas vegetais junto à Agência Nacional de Vigilância Sanitária (ANVISA) e dá outras providências. Diário Oficial da União, Poder Executivo, Brasília, DF, 10 mar. 2010.
1. Bachelor in Nutrition from UNIVALI - Postgraduate in Functional and Aesthetic Nutrition and in Advanced Clinical Nutrition - Nutritionist at Hospital Santo Antônio and Blumenau City Hall
2. Technologist in Aesthetics and Cosmetology at ULBRA - Professor of the Course Technology in Cosmetology and Aesthetics at UNIVALI - Master Student of the Master Program in Health and Management of Work at UNIVALI
Institution: Centro de Ciências Avançadas (CCA Cursos).
Corresponding author:
Camile Laís Rocha
Rua Luiz Altemburg Sênior, 90 - Escola Agrícola
Blumenau, Santa Catarina, Brazil Zip code: 89031-300
E-mail: camile.rocha@hotmail.com
Article received: November 24, 2012.
Article accepted: June 29, 2013.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter