

Original Article - Year 2015 - Volume 30 -
Reconstruction in Mohs micrographic surgery
Reconstrução em cirurgia micrográfica
ABSTRACT
INTRODUCTION: Mohs micrographic surgery is used for the excision of skin neoplasms, especially in locally aggressive histological subtypes of basal cell carcinoma, tumor recurrences, or tumors located in critical areas. This technique has a high cure rate and allows maximum preservation of tissues. In this study, we aimed to assess the effectiveness of Mohs micrographic surgery and reconstruction methods.
METHODS: Data from 50 patients who underwent Mohs micrographic surgery to excise skin tumors and reconstruct lost tissue were collected retrospectively. All patients were operated on between January 2005 and December 2013 at the Plastic Surgery Clinic of the Felício Rocho Hospital (Belo Horizonte, MG, Brazil). The patients' age, sex, tumor location, previous treatment, histological type, number of segments analyzed by micrographic surgery, reconstruction method used, and preservation were studied.
RESULTS: Thirty-one patients (62%) were women and 19 (38%) were men. The mean age was 63.8 years. All lesions were facial, with 66% of cases affecting the nasal area. Presurgery, there were 48 cases (96%) of basal cell carcinoma and 2 cases (4%) of microcystic adnexal carcinoma. Local flaps were the most used reconstruction method. The patients were followed-up for a mean of 48.4 months. We did not observe any cases of tumor recurrence.
CONCLUSION: Mohs micrographic surgery was shown to be effective in the treatment of 50 skin neoplasms. We recommend that surgical defects should be repaired by the plastic surgeon.
Keywords: Mohs surgery; Skin neoplasms; Surgical flaps; Head and neck neoplasia; Plastic surgery
RESUMO
INTRODUÇÃO: A cirurgia micrográfica de Mohs é empregada para exérese de neoplasias cutâneas, especialmente carcinomas basocelulares de subtipos histológicos localmente agressivos, tumores recidivados ou localizados em regiões nobres. Apresenta elevados índices de cura e permite preservação tecidual. O objetivo é analisar a eficácia da cirurgia micrografia de Mohs e os métodos de reconstrução utilizados.
MÉTODO: Foram coletados, retrospectivamente, dados de 50 pacientes submetidos à exérese de tumores cutâneos por meio da cirurgia micrográfica de Mohs e à reconstrução da perda de substância. Todos os pacientes foram operados no período entre janeiro de 2005 a dezembro de 2013 na Clínica de Cirurgia Plástica do Hospital Felício Rocho (Belo Horizonte, MG, Brasil). Os pacientes foram estudados com relação à idade, gênero, localização do tumor, tratamento prévio, tipo histológico, número de fragmentos analisados na cirurgia micrográfica, método de reconstrução empregado e proservação.
RESULTADOS: Trinta e um pacientes (62%) foram do gênero feminino e 19 (38%) do masculino. A média de idade foi de 63,8 anos. Todas as lesões encontravam-se na face, com 66% dos casos com acometimento da região nasal. Considerando o diagnóstico pré-operatório, 48 casos (96%) eram carcinoma basocelulares e dois casos (4%) correspondiam ao carcinoma microcístico anexial. Retalhos locais foram o tipo de reconstrução mais utilizado. Os pacientes foram acompanhados por média de 48,4 meses. Nenhum caso de recidiva tumoral foi observado.
CONCLUSÃO: A cirurgia micrográfica de Mohs se mostrou altamente eficaz no tratamento dos 50 casos de neoplasias cutâneas. Recomenda-se que os defeitos cirúrgicos sejam reparados pelo cirurgião plástico.
Palavras-chave: Cirurgia de Mohs; Neoplasias cutâneas; Retalhos cirúrgicos; Neoplasias de cabeça e pescoço; Cirurgia plástica.
Mohs micrographic surgery (MMS) is a technique named after the doctor, Frederic Edward Mohs1. In 1932, Mohs established a technique known as Mohs chemosurgery at the Wisconsin University, USA, to remove skin tumors. This technique involved applying dichloroacetic acid over the lesion in order to coagulate skin proteins and enable percutaneous absorption of the fixing solution, in particular, zinc chloride, which was applied subsequently. The fixing solution was used to preserve the histological structure of the tissues. Approximately 6 to 24 hours thereafter, the tumor was removed, and the margins were carefully identified and divided into segments for anatomopathological examination. In case the segments contained tumors, the corresponding area was subjected to dichloroacetic acid and zinc chloride treatment. This technique was shown to be very useful because high cure rates were observed even in aggressive neoplasia. However, there were drawbacks, such as pain due to the fixing solution, the long duration, and the delay in reconstruction1-3. For instance, surgical activities lasted several days.
In 1953, when performing an eyelid tumor resection, Mohs3 used less fixing solution in order to prevent eye tissue damage due to zinc chloride. The lesion's margins were analyzed using frozen sections of fresh tissue, thus replacing chemosurgery with MMS.
In 1969, Mohs3 described a series of 70 eyelid tumors that were treated using the fresh specimens' technique at the Congress of the American College of Chemosurgery. After 5 years, 100% of tumors had been cured.
In 1974, Stegman and Tromovitch4 published a report about the resection of 102 basal cell carcinomas (BCC) from different regions of the body; these were analyzed using the fresh margins technique and a 97% cure rate was obtained in 5 years.
Since then, MMS has been used to treat several histological types of skin neoplasia, in particular BCC, with high cure rates and tissue preservation.
OBJECTIVES
We aimed to analyze the efficacy of MMS and reconstruction methods in the treatment of skin neoplasia at a plastic surgery clinic in a general hospital.
METHOD
Fifty patients who had undergone excision of skin tumors through MMS between January 2005 and December 2013 were assessed retrospectively at the Plastic Surgery Clinic of the Felício Rocho Hospital (Belo Horizonte, MG, Brazil). Data concerning sex, age, tumor location, histological subtype, previous treatment, number of segments assessed, type of reconstruction used, preservation, and photographic records were collected from the medical records. The due date for keeping the data was August 1, 2014.
The data were entered in a Microsoft Office Excel spreadsheet, following which statistical and graphical analyses were performed.
All patients without a complete medical record were excluded from the study.
The inclusion of patients in the study was according to the general guidelines for micrographic surgery5-7 as follows:
a) recurring BCC;
b) BCC with inaccurate clinical margins;
c) locally aggressive BCC forms, such as sclerodermiform, infiltrative, and micronodular BCC;
d) microcystic adnexal carcinoma (MAC)8,9;
e) BCC in locations where preserving healthy tissue was important, such as the nose and eyelids.
Surgical Technique
With the patient under mild sedation using intravenous midazolam and local anesthesia, we marked the lesion including free margins of approximately 5 mm. Using these marks, we demarcated segments around the neoplasia at 3-mm intervals from each other. Subsequently, the visible tumor was removed using a lateral margin of 5 mm (Figure 1). We first removed the perilesional margins, divided them into segments, and carefully marked them using different colors of 5.0 thick surgical thread as follows: blue polypropylene (Prolene®), black nylon, and colorless poliglecaprone (Monocryl®) (Figure 2). The deep margin of the lesion was removed individually and its superficial and deep sides were demarcated using surgical thread. The request for an anatomopathological examination was accompanied by a drawing of the affected area and identification of the resected fragments. The specimens were sent to the anatomopathology department of the same institution, in separate flasks containing physiological saline. The frozen examination was conducted using cryostat sectioning followed by hematoxylin and eosin staining. In case of residual tumors, the location was identified accurately and further local excision was performed (Figure 3).
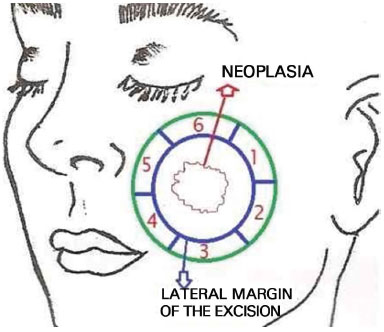
Figure 1. A skin lesion with a 0.5-cm lateral margin of excision and 6 marked segments of 3-mm width is shown. The margins were examined on frozen sections. Both sides of the deep margin were analyzed (segment 7).

Figure 2. The borders of segment 1 were identified using thread as follows: black thread marking the external border, blue thread marking the upper border, and colorless thread marking the lower border of the fragment.
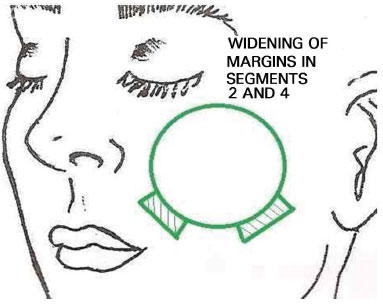
Figure 3. Because of positive margins, the margins of segments 2 and 4 were widened successively until the whole lesion was removed.
The excision of subsequent margins was repeated until the neoplasia was removed completely. In order to save time, the first resected margins were removed before the main lesion in some cases, and were sent for histopathological examination. The neoplastic lesion was removed only after this step. Thus, the margins were analyzed while the main lesion was removed and hemostasis was performed.
Whenever possible, the reconstruction of lost tissue was performed at the same time, using flaps and skin grafts.
The literature associated with this topic was analyzed using MEDLINE and PubMed.
RESULTS
Between January 2005 and November 2013, 50 patients underwent MMS. Thirty-one (62%) women and 19 (38%) men (i.e. a male:female ratio of 1.6:1) were included (Figure 4).
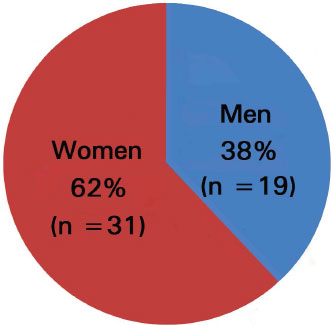
Figure 4. Sex distribution of patients.
The patients' age varied from 31 to 89 years, with a mean of 63.8 years. The highest prevalence was observed in patients in their 70s and 80s (Figure 5).
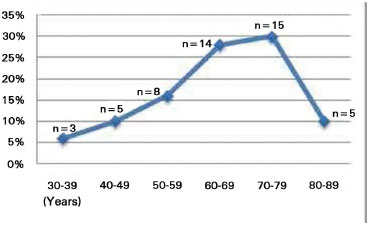
Figure 5. Age distribution of patients.
In the topographic assessment of the lesions, all lesions were found to be facial, and were predominantly in the nasal region (24 cases: 48%) or extended into surrounding regions from the nasal region (9 cases: 18%).
Other affected regions were as follows: nasogenian sulcus - 3 cases (6%), supercilii - 3 cases (6%), medial corner of the eye - 3 cases (6%), upper lip - 2 cases (4%), eyelids - 1 case (2%), frontal region - 1 case (2%), and zygomatic region - 1 case (2%). Locations that were affected simultaneously were as follows: frontal and temporal region - 1 case (2%), zygomatic and lower eyelid region - 1 case (2%), and lower eyelid and medial corner of the eye - 1 case (2%).
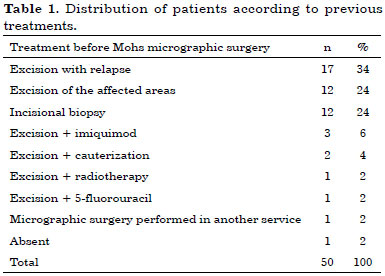
Before the introduction of MMS, 58% of patients had tumor recurrences or affected margins after undergoing conventional excision (Table 1).
The presurgery diagnosis was BCC in 48 (96%) cases, and its subtypes and incidence are presented in Figure 6. In 2 cases (4%), the preoperative diagnosis was MAC.
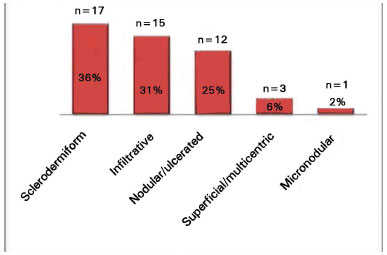
Figure 6. Subtypes of basal cell carcinoma identified in preoperative diagnosis (n = 48).
In 1 case of neoplasia at the nasal wing, which was treated with imiquimod at another location, the preoperative diagnosis of BCC was incorrect. The anatomopathological examination during MMS revealed squamous cell carcinoma.
In 44 (88%) out of 50 MMS cases, we were able to remove the whole tumor in just a single surgical attempt. In 6 cases (12%), more attempts were required to remove the residual tumor fragments. The number of fragments analyzed per patient varied from 3 to 15, with a mean of 4.2 fragments.
For reconstruction, local flaps were used in 24 cases (48%). We used total skin grafts separately in 14 cases (28%); the donor areas were the preauricular, retroauricular, and supraclavicular regions, and good aesthetic results were obtained. In 5 patients (10%), we used both flaps and grafts; 3 patients (6%) underwent primary closure, 2 (4%) underwent secondary intention healing, 1 (2%) underwent skin flap grafting and secondary intention healing, and 1 (2%) underwent skin grafting and secondary intention healing (Figure 7).
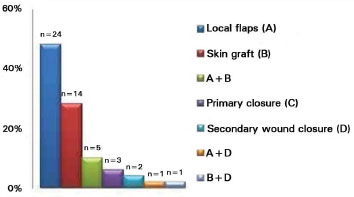
Figure 7. Surgical techniques used in 50 reconstructions.
A total of 31 local grafts were performed; 13 nasogenian grafts were used to correct nasal defects (Figure 8), 7 skin grafts were taken from the surroundings and selected randomly, 5 frontal oblique fasciocutaneous grafts were performed for nasal reconstruction, 4 Mc-Gregor skin flaps (Z-plasty) were employed to correct defects in the lower eyelids (Figure 9), and an Esser skin flap (bilobed) in the nasal dorsum and a myocutaneous flap in the upper eyelid were used to reconstruct the ipsilateral medial corner of the eye.
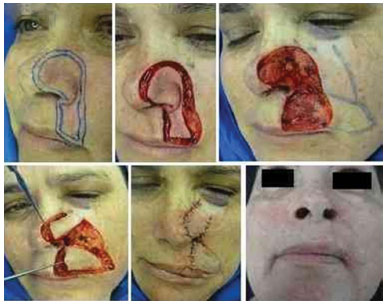
Figure 8. Multicentric basal cell carcinoma in the left alar lobule and upper lip, which was subjected to surgery twice and recurred. The lost tissue was repaired using a cheek flap and advanced nasogenian V-Y flap for the upper lip.
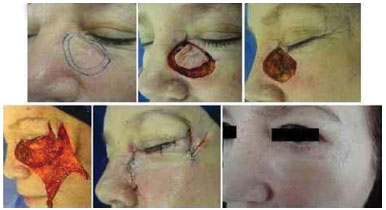
Figure 9. Infiltrative basal cell carcinoma showing non-defined margins on the left side of the nose and lower eyelid region. The tissue lost because of the micrographic surgery was repaired by the McGregor technique (lateral zetaplasty).
In 2 patients with nasal neoplasia, MMS was suspended owing to invasion of the maxillary periostitis by the tumor. In 1 case, we resected the frontal maxillary process after using local and topical anesthesia in the same surgical time. The wound healed by second intention, and we performed nasal reconstruction with a frontal flap subsequently (Figure 10). In another case with invasion of the periosteum of the alveolar process, we performed partial maxillectomy at a different time, with the patient under general anesthesia. As BCC presented with areas of squamous differentiation, we opted for radiotherapy followed by local composition using an external prosthesis. An elderly woman became restless as the surgical procedure was extended, and hence, required intubation and general anesthesia. In 1 case, even though the surgery was extended for 6 hours, the procedure could not be completed; therefore, the patient was hospitalized and MMS was continued on the following day, under general anesthesia. Almost the whole nose, part of the upper lip, and the nasogenian sulcus were excised. Reconstruction was subsequently performed with an expanded frontal flap.
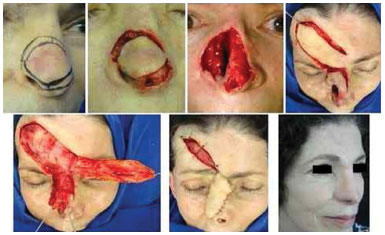
Figure 10. Recurrent infiltrative basal cell carcinoma in the nose. Micrographic surgery was interrupted because of invasion of the maxillary periosteum. Resection of the frontal process of the right maxilla at the same time as the surgery. Oblique forehead flap combined with a reversed flap from the root of the nose (lining). Result after retouching.
The patients showed satisfactory aesthetic results, even in those who underwent secondary intention healing.
The patients were followed-up for varying durations, from 6 to 114 months, with a mean of 48.4 months. As many patients were referred by dermatologists, follow-ups with dermatologists were recommended for all patients. During follow-up, no recurrences were observed.
DISCUSSION
MMS allows safe histological control of tumor margins10-12. As the procedure needs to be performed meticulously, it has a long surgical time and a higher initial cost; these drawbacks are largely overcome by the excellent results obtained.
The advent of the cryostat, which is a microtome with a freezer, has allowed thinner slicing of tissues than by freezing with CO2, and can provide an adequate number of slides for an accurate diagnosis13. The demarcation of borders using surgical thread of different colors is safe and greatly simplifies MMS. It is of utmost importance that the anatomopathologist and histopathology technician are familiar with the method, and are able to clarify any queries with the surgeon during the surgical procedure.
BCC was the most prevalent lesion in this study, representing 96% of cases. Previous studies have demonstrated that the 5-year relapse rate for primary and recurrent BCC treated with conventional surgery is 10% and 17%, respectively. In the tumors treated with MMS, this rate decreases to 1% and 5.6%, respectively14 .
Two cases of MAC were identified; they should be carefully evaluated because they are rare neoplasia of the eccrine sudoriferous glands with slow growth and locally aggressive behavior. It mainly affects the head and is included in the differential diagnosis, along with benign adnexal tumors, such as trichoepitheliomas, syringomas, and malignant skin tumors. Owing to its subclinical evolution and perineural invasion, it is diagnosed at an advanced stage and incorrect treatment results in a high recurrence rate. Many studies confirmed the efficacy of micrographic surgery for the treatment of MAC. Conventional excision showed relapse rates greater than 47%, generally in the first 3 years. On the other hand, MMS has a relapse rate of 0-22% in a follow-up time of 5 years8,9.
During the follow-up of the 50 cases herein, with a mean preservation time of 48.4 months, we did not identify any recurrences, which may be explained by the use of wider lateral and deep resected margins. Overall, the margins described in the method section are larger than those usually used by dermatologic surgeons.
The tumors analyzed were facial, in particularly in the H-zone, which includes the lateral frontal, temporal, and periocular regions, the nose and upper lips11. Previously, 58% of the treated patients showed tumor recurrence or affected margins after the first excision was conducted elsewhere. Interestingly, 1 patient relapsed 9 times following the conventional surgical procedure. Tumor relapses have a worse prognosis, with an even higher rate than other recurrences; therefore, we underline the need to search for a definitive treatment for the first intervention.
The nose was the most affected region (66% of cases); during nose reconstructions, we considered the characteristics of the surgical wound, such as the topography, extension, and depth. Whenever possible, the following aesthetic subunits of the nose were taken into consideration: the dorsum, tip, lateral sidewall, alar lobule, and columella12,14. The focus was to replace the lack of tissue in the affected area by surrounding skin. In cases where the tumor affected a lower extension of the aesthetic subunit, a total excision of the subunit was performed to leave the scars in the natural sulci of the skin.
In 5 patients with a nose defect that affected the total thickness, we used the frontal flap. In 3 of these cases, the flap was divided. One part comprised the nasal lining, whereas the other was used to reconstruct the external tegument. In 1 case where the surgical defect surpassed the nose borders, the frontal region was expanded, allowing the flap procedure to be completed in two parts, in additional time. In 2 patients, the nasal lining was reconstructed using local flaps with the epidermis side turned to the nasal cavity. In the expanded flap, the donor area was closed by direct suture. In 1 case, the margins were brought together with elastic sutures, and, in 3 cases, we performed partial synthesis of the donor area and waited for closure by secondary intention.
Among the techniques used for nasal reconstruction, the nasogenian flap was the most common (13 cases). We produced flaps from the upper pedicles to mainly repair the alar lobules, and the lower pedicled flaps were reserved for the reconstruction of lesions in the upper lip.
We used the bilobed flap procedure given by Esser15 in 1918 in 1 case that presented a surgical defect of small dimensions in the nasal dorsum.
In 3 cases (6%) of eyelid reconstruction wherein there was a loss of total thickness, we associated local flaps with an ear conchal cartilage graft with perichondrium to replace the posterior lamellae according to the method of Matsuo et al.16,17.
We used McGregor grafts18 in 3 cases for the reconstruction of defects in the lower eyelid through a lateral periorbital zetaplasty.
In 3 cases with great nasal tissue loss in elderly patients, we opted for an external prosthesis.
Respecting the anatomical units of the area subjected to surgery is of utmost importance. For a plastic surgeon who reconstructs affected areas, reconstruction of a larger defect is frequently easier and offers better results than the reconstruction of small defects.
CONCLUSION
MMS was shown to be highly efficient in the treatment of 48 cases of sclerodermiform, infiltrative, micronodular, or recurrent BCC, or BCC with inaccurate borders. Two cases of MAC were successfully treated. The efficacy of the method was 100% with a preservation mean of 48.4 months. Many methods were used in the reconstruction of the defects. We recommend that the defects should be repaired by a plastic surgeon more familiar with reconstruction techniques than a dermatologic surgeon who uses MMS more frequently.
REFERENCES
1. Mohs FE. Chemosurgery, a microscopically controlled method of cancer excision. Arch Surg. 1941;42(2):279-95. DOI:http://dx.doi.org/10.1001/archsurg.1941.01210080079004
2. Trost LB, Bailin PL. History of Mohs surgery. Dermatol Clin. 2011;29(2):135-9. DOI: http://dx.doi.org/10.1016/j.det.2011.01.010
3. Mohs FE. Cancer of eyelids. Bull Am Coll Chemosurg. 1970;3:10-1.
4. Tromovitch TA, Stegeman SJ. Microscopically controlled excision of skin tumors. Arch Dermatol. 1974;110(2):231-2. PMID: 4853214 DOI:http://dx.doi.org/10.1001/archderm.1974.01630080033008
5. Robins P. Mohs micrographic surgery. In: McCarthy JG, ed. Plastic surgery. Philadelphia: WB Saunders; 1990. p.3652-62.
6. Terzian LR. Cirurgia micrográfica de Mohs. Rev Bras Med. 2011;68(supl.2):4-12.
7. Chagas FS, Santana Silva Bd. Mohs micrographic surgery: a study of 83 cases. An Bras Dermatol. 2012;87(2):228-34. PMID: 22570026
8. Diamantis SA, Marks VJ. Mohs micrographic surgery in the treatment of microcystic adnexal carcinoma. Dermatol Clin. 2011;29(2):185-90. DOI:http://dx.doi.org/10.1016/j.det.2011.01.012
9. Thomas CJ, Wood GC, Marks VJ. Mohs micrographic surgery in the treatment of rare aggressive cutaneous tumors: the Geisinger experience. Dermatol Surg. 2007;33(3):333-9.
10. Kopke LFF, Gouvêa OS, Bastos JCF. Dez anos de experiência com cirurgia micrográfica pelo método de Munique: relato de 93 casos operados. An Bras Dermatol. 2005;80(6):583-59. DOI: http://dx.doi.org/10.1590/S0365-05962005000700003
11. Terzian LR, Nogueira VMA, Paschoal FM, Barros JC, Machado Filho CAS. Cirurgia micrográfica de Mohs para preservação tecidual nas cirurgias oncológicas da face. Surg Cosmet Dermatol. 2010;2(4):257-63.
12. Sclafani AP, Sclafani JA, Sclafani AM. Successes, revisions, and postoperative complications in 446 Mohs defect repairs. Facial Plast Surg. 2012;28(3):358-66. DOI: http://dx.doi.org/10.1055/s-0032-1312691
13. Técnica de biópsia - Anatpat - Unicamp [Acesso 28 Jul 2014]. Disponível em: http:www.anatpat.unicamp.br/mustecbiop.html
14. Reis NA, Azevedo LCM, Stolf HO, Nouri K, Kimyai-Asadi A, Goldberg LH. Cirurgia micrográfica de Mohs. Surg Cosmet Dermatol. 2011;3(3):227-31.
15. Esser JFS. Gestielte lokale nasenplastik mit zwerkipugen lappen, deckung des sekunderen defektes von ersten zipfel durch den zweiten. Dtsch Z Chir. 1918;143:385. DOI: http://dx.doi.org/10.1007/BF02793149
16. Matsuo K, Hirose T, Takahashi N, Iwasawa M, Satoh R. Lower eyelid reconstruction with a conchal cartilage graft. Plast Reconstr Surg. 1987;80(4):547-52. DOI: http://dx.doi.org/10.1097/00006534-198710000-00012
17. Alves JCR, Liu RT, Silva Filho AS, Pereira NA, Carvalho EES. Eyelid repair using an autologus conchal cartilage graft. Rev Bras Cir Plást. 2012;27(2):243-8. DOI: http://dx.doi.org/10.1590/S1983-51752012000200013
18. McGregor IA. Eyelid reconstruction following subtotal resection of upper or lower lid. Br J Plast Surg. 1973;26(4):346-54. PMID: 4586380
1. Hospital Felício Rocho, Belo Horizonte, MG, Brazil
2. Sociedade Brasileira de Cirurgia Plástica, Belo Horizonte, MG, Brazil
3. Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brazil
4. Universidade de Itaúna, Itaúna, MG, Brazil
5. Instituto de Cirurgia Plástica Avançada, Belo Horizonte, MG, Brazil
6. Faculdade de Ciências Médicas de Minas Gerais, Belo Horizonte, MG, Brazil
Institution: Clínica de Cirurgia Plástica do Hospital Felício Rocho, Belo Horizonte, MG, Brazil.
Corresponding author:
Augusto César de Melo Almeida
Rua Ouro Preto, 617/1904, Barro Preto
Belo Horizonte, MG, Brazil Zip code 30170-040
E-mail: almeidaugusto@hotmail.com
Article received: September 10, 2014.
Article accepted: March 15, 2015.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter