

Original Article - Year 2015 - Volume 30 -
Strategies for chest reconstruction following extensive resection of locally advanced breast tumors: an 11-case series
Estratégias em reconstruções de tórax pós-ressecções extensas de tumores de mama localmente avançados: uma série de 11 casos
ABSTRACT
INTRODUCTION: Breast cancer is a major cause of mortality among women in Brazil. Locally advanced breast tumors are classified as stage III because they compromise the breast tissue in all or almost all of its extensions and regional lymph nodes, without distant metastases. Surgical treatment of these advanced tumors includes modified radical mastectomy, which involves axillary dissection and large skin resection, leading to major defects in the chest cavity that require complex reconstruction.
METHOD: A retrospective clinical study including a series of 11 cases diagnosed with locally advanced breast tumors was performed from January 2006 to March 2014.
RESULTS: The chest cavity reconstruction methods performed after resection of advanced breast tumors included three fasciocutaneous flaps, two skin grafts, and six reconstructions with muscle flaps. These procedures resulted in effective coverage of extensive chest defects.
CONCLUSION: Aggressive surgical treatment is feasible in these tumors; although numerous reconstruction options are available, individual case assessment is necessary to determine the optimal technique to reduce surgical trauma and lower morbidity in order to avoid delays in adjuvant treatment in these patients. The reconstructions were safe, with satisfactory results similar to those reported in the literature.
Keywords: Chest wall; Surgical flaps; Mastectomy; Breast tumor/surgery.
RESUMO
INTRODUÇÃO: O câncer de mama representa um dos maiores responsáveis pela mortalidade feminina no Brasil. Os tumores de mama localmente avançados são classificados como estádio III, pois comprometem a mama em toda ou quase toda sua extensão e/ou linfonodos regionais sem apresentar metástase à distância. O tratamento cirúrgico desses tumores avançados consiste na realização de mastectomia radical modificada, que inclui esvaziamento axilar e grandes ressecções cutâneas, levando a grandes defeitos na caixa torácica, necessitando de complexas reconstruções.
MÉTODO: Foi realizado um estudo clínico retrospectivo, no período de janeiro de 2006 a março de 2014, de uma série de 11 casos diagnosticados com tumores de mama localmente avançados.
RESULTADOS: Foram realizados 3 retalhos fasciocutâneos, 2 enxertos de pele e 6 reconstruções com retalhos musculares para a reconstrução da caixa torácica após a resseção de tumores avançados da mama. Observouse uma cobertura eficaz dos extensos defeitos em tórax.
CONCLUSÃO: O tratamento cirúrgico agressivo é viável nestes tumores; diferentes técnicas para a reconstrução do tórax são possíveis; cada caso deve ser avaliado individualmente, devendo ser realizado o reparo que possibilite um menor trauma cirúrgico e menor morbidade, de forma a não retardar o tratamento adjuvante nestes pacientes. As reconstruções apresentadas foram consideradas seguras, os resultados obtidos foram satisfatórios e de acordo com a literatura.
Palavras-chave: Parede torácica; Retalhos cirúrgicos; Mastectomia; Neoplasias da mama/Cirurgia.
Breast cancer is a major cause of mortality among women in Brazil. In its most recent estimates, the National Cancer Institute (INCA) predicted 57,120 new cases of breast cancer in 2014, with an estimated risk of 56.09 cases per 100,000 women. Locally advanced breast tumors are classified as stage III because they compromise the breast in all or almost all of its extensions and regional lymph nodes, without distant metastases1-3.
The standard surgical treatment for these advanced tumors is modified radical mastectomy, which includes axillary dissection and large skin resections, which lead to major defects in the chest cavity that require complex reconstruction. Halsted mastectomies, with extended resections and removal of the pectoralis major and minor muscles, are reserved for cases with tumor involvement of the pectoral muscle. Radiotherapy, chemotherapy, and adjuvant hormonal therapy comprise the set of clinical treatment measures1,2,4.
Various forms of chest wall reconstruction have been used to repair defects after breast tumor resection, including autologous muscle flaps such as the vertical rectus abdominis myocutaneous (VRAM) and transverse rectus abdominis myocutaneous (TRAM) flaps, first described in 1977 by Drever; in 1979, Robbins was the first to use the latissimus dorsi muscle flap, described by Tansini in 1906, for breast reconstruction. In cases of advanced tumors in which radiotherapy and chemotherapy treatments cannot be delayed, the recommended reconstruction techniques include fasciocutaneous flaps such as the thoracoepigastric flap described by Bohmert and Cronin, which allows coverage of breast areas, sternal defects and inferior or middle thoracic regions, lateral thoracic flaps, and even skin grafting1,4-9.
OBJECTIVE
To present a case series of techniques and results of reconstruction of extensive chest defects after resection of locally advanced breast tumors.
METHODS
We performed a retrospective clinical study of a series of cases diagnosed with locally advanced breast tumors from January 2006 to March 2014. A total of 11 chest reconstructions were performed after resection of advanced breast tumors.
The inclusion criteria were patients with extensive thoracic defects after breast or chest tumor resection. The following parameters were studied: age, gender, etiology, lesion location, repair procedure, and complications. Reconstruction methods were selected based on the defect characteristics, location, and quantity and quality of tissues required.
RESULTS
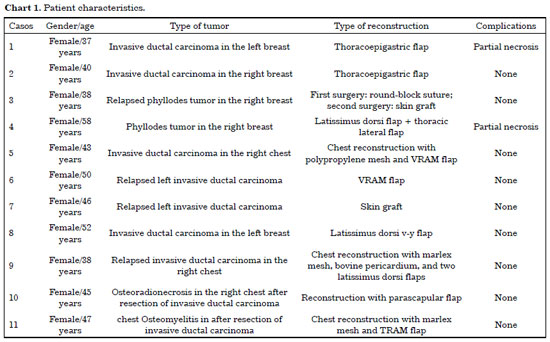
The patients in this study sample were female with a mean age of 44.9 years, ranging in age from 37 to 58 years; the left breast was more often affected (54.5% of cases), and invasive ductal carcinoma was the most prevalent type of breast tumor (81.8% of cases). The patient characteristics are presented in Chart 1.
The reconstructions included two thoracoepigastric fasciocutaneous flaps, one lateral thoracic fasciocutaneous flap, two skin grafts, and six muscle flap reconstructions for chest wall reconstruction, consisting of three latissimus dorsi muscle flaps, two vertical rectus abdominis myocutaneous (VRAM) flaps, and one transverse rectus abdominis myocutaneous (TRAM) flap.
These reconstructions resulted in effective coverage of extensive thoracic defects, and none of the patients presented with flap infection. Two cases (18.1%) developed slight partial flap necrosis, which were debrided, and a random flap was rotated for closure of the remaining defect. The recovery time after surgery ranged from 20 to 60 days.
DISCUSSION
Locally advanced breast tumors remain an extremely important issue in developing countries, since it is difficult for patients to seek and receive treatment. The increased incidence in Brazil has been accompanied by increased mortality, which can be attributed mainly to delayed diagnosis and appropriate therapy. Even so, locoregional treatment by means of wide surgical resection and reconstruction of the chest wall was a treatment option for the cases presented in this study1,4,5.
Chest wall reconstruction provides stability to the skeletal system of the thorax, preventing its collapse and permitting physiological respiratory exchange; reconstruction also repairs and synthesizes lost soft tissue covering the chest in order to preserve its contour. Resections may involve the entire thickness of the chest wall, and more extensive resection typically requires more complex repairs. Resection of the sternum, costal arches, or chondrocostal cartilage of the anterior or lateral walls of the chest require use of bones or synthetic prostheses combined with skin coverage for rehabilitation of the chest as well as stabilization of the chest cavity and adequate respiratory physiology. Polypropylene mesh is preferred, offering easy handling, permeability, durability, inertness, and economy; however, intense fibrotic reactions can occur when the mesh contacts the pleura and lung4,8,10-13.
Several reconstructive surgery techniques for the chest have been described, including skin grafts to fasciocutaneous, muscle, myocutaneous, and microsurgical flaps14,11-14. The choice of reconstructive technique is based on not only clinical criteria, but also the surgeon experience and patient preference; thus, similar case presentations may receive different types of reconstruction4,5,7.
Fasciocutaneous flaps are based on superficial vascularization of the subdermal plexuses of the superficial fascia of the tissues covering the chest wall. This region is richly vascularized, allowing the construction of long, thick, resilient, and safe flaps. They can be used with both medial and lateral pedicles. In our study, there were three fasciocutaneous flaps for coverage of chest abnormalities: two medial and one lateral pedicle. In this study, use of this flap was indicated for extensive of skin and subcutaneous tissue resections that had preserved the pectoralis major and minor muscles as well as the thoracic cavity (Figure 1).
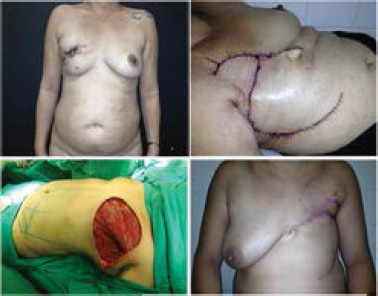
Figure 1. Two cases of invasive ductal carcinoma that received thoracoepigastric fasciocutaneous flap reconstruction.
In the thoracic and abdominal region, a series of muscles can be used for the most varied coverage of skin loss and exposure, including the pectoralis major, latissimus dorsi, and rectus abdominis, among others. When there are limiting factors for use of conventional flaps, it is necessary to apply microsurgical flaps or skin grafts4,6. In the current study, six muscle flaps were constructed for reconstruction of the chest after resection of skin, subcutaneous tissue, pectoral muscle with exposure of the rib cage, and after whole-thickness resections of the chest cavity (Figures 2 and 3). Three latissimus dorsi muscle flaps, two vertical rectus abdominis muscle flaps and one TRAM flap were constructed. In patients with whole-thickness resection of the chest wall, a marlex mesh was used for reconstruction of the ribcage, which was then covered with a myocutaneous flap (Figures 4 and 5).
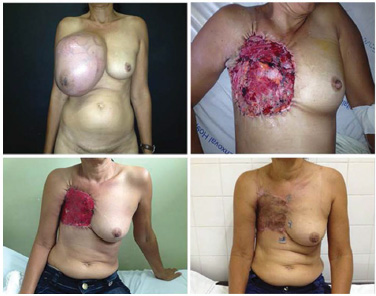
Figure 2. A 52-year-old patient with invasive ductal carcinoma in her left breast who underwent large v-y latissimus dorsi muscle flap reconstruction.
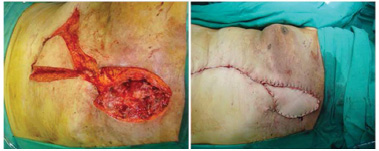
Figure 3. A 50-year-old patient with relapsed invasive ductal carcinoma who underwent VRAM flap reconstruction.
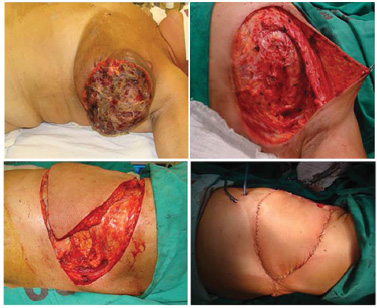
Figure 4. Osteomyelitis in the chest after resection following invasive ductal carcinoma; chest reconstruction with polypropylene mesh and TRAM flap.
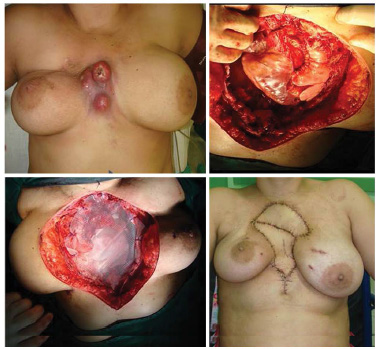
Figure 5. A 38-year-old patient with relapsed invasive ductal carcinoma in chest who underwent reconstruction with bovine pericardium, polypropylene mesh and two latissimus dorsi muscle flaps.
Chest reconstruction with skin graft is the treatment of choice for extensive skin defects in which the pectoralis major and minor muscles have been preserved, including resection of tumors with high relapse rates such as phyllodes tumors of the breast. In their malignant forms, these infrequent tumors (0.5-1% of cases) have high local aggressiveness and relapse rates, with poor prognosis in their metastatic and advanced forms. In this case, skin grafting is justified, since this repair typically presents poor aesthetic results4,5. In our sample, two patients with bulky phyllodes tumors underwent reconstruction with skin grafts. This repair was performed in two separate surgeries. The first was a double round-block suture with prolene 0 to decrease the chest defect; after 14 days, a skin graft was performed with granulation of the wound bed (Figure 6).
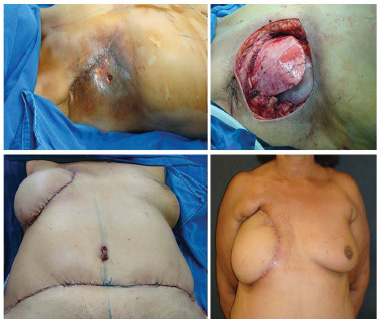
Figure 6. A 38-year-old patient with relapsed phyllodes tumor in her right breast; reconstruction with round-block suture and skin grafting were performed on the 24th postoperative day.
In this plastic surgery service, muscular flaps are not considered a first option for chest reconstruction in patients with stage III tumors who will undergo adjuvant radiotherapy. When possible, its use should be postponed due to risks of complications from ionizing radiation therapy. The reconstruction strategies used in our study resulted in suitable coverage of the defect and quicker initiation of adjuvant therapy.
CONCLUSION
Aggressive surgical treatment is feasible in patients with locally advanced breast tumors, and may be the only option for local control in these patients. While a variety of chest reconstruction techniques are available, each case should be assessed individually in order to select the optimal method for minimal surgical trauma, lower morbidity, and acceptable aesthetic results and to avoid delay in adjuvant treatment. The reconstruction methods presented in this study are safe, feasible, and offered satisfactory results, similar to other reports in the literature.
REFERENCES
1. Graziosi GB, Lucas FAS, Maximiano AMC, Caiado Neto BR, Prota Júnior MLC. Reconstrução de parede torácica em tumores de mama localmente avançados. Rev Bras Cir Plást. 2013;28(supl):64.
2. Hara RHA, Rodrigues HL, Bonomi D, Lage RR, Carneiro JL, Cruzeiro CNL. Reconstrução de parede torácica pós-osteoradionecrose: relato de caso. Rev Bras Cir Plást. 2011;26(supl):88.
3. Freitas RS, Ambrósio AMS, Filho Fagotti A, Oliveira e Cruz GA, Romano G. Reconstrução de grandes defeitos torácicos com retalho toracoabdominal de base medial. Anais do 42º Congresso Brasileiro de Cirurgia Plástica; 2005 Nov 11-14; Belo Horizonte, MG, Brasil. p.48.
4. Mélega JM, Viterbo F, Mendes FH. Cirurgia plástica: os princípios e a atualidade. Rio de Janeiro: Guanabara Koogan; 2011. p.573-4.
5. Tavares FMO, Menezes CMGG, Moscozo MVA, Xavier GRS, Oliveira GM, Amorim Júnior MAP, et al. Retalho de omento: uma alternativa em cirurgia reparadora da parede torácica. Rev Bras. Cir Plást. 2011;26(2):360-5.
6. Pinto EBS, Muniz AC, Erazo P, Cação EG, Abdalla PCSP. Reconstrução mamária: princípios geométricos dos retalhos cutâneos em duplo V. Rev Soc Bras Cir Plást. 1998;13(3):19-42.
7. Losken A, Thourani VH, Carlson GW, Jones GE, Culbertson JH, Miller JI, et al. A reconstructive algorithm for plastic surgery following extensive chest wall resection. Br J Plast Surg. 2004;57(4):295-302. PMID: 15145731 DOI: http://dx.doi.org/10.1016/j.bjps.2004.02.004
8. Costa PR, Melo JRC, Andrade Júnior JCCG, Bezerra MM, Neves LJVA, Araújo JMC. Reconstrução da parede torácica com metilmetacrilato: relato de caso. Rev Soc Bras Cir Plást. 2007;22(4):266-8.
9. Alvarado M, Ewing CA, Elyassnia D, Foster RD, Shelley Hwang E. Surgery for palliation and treatment of advanced breast cancer. Surg Oncol. 2007;16(4):249-57. DOI: http://dx.doi.org/10.1016/j.suronc.2007.08.007
10. Novoa N, Benito P, Jiménez MF, de Juan A, Luis Aranda J, Varela G. Reconstruction of chest wall defects after resection of large neoplasms: ten-year experience. Interact Cardiovasc Thorac Surg. 2005;4(3):250-5. DOI: http://dx.doi.org/10.1510/icvts.2004.103432
11. Chin PL, Andersen JS, Somlo G, Chu DZ, Schwarz RE, Ellenhorn JD. Esthetic reconstruction after mastectomy for inflammatory breast cancer: is it worthwhile? J Am Coll Surg. 2000;190(3):304-9. PMID: 10703855
12. Miller DL, Force SD, Pickens A, Fernandez FG, Luu T, Mansour KA. Chest wall reconstruction using biomaterials. Ann Thorac Surg. 2013;95(3):1050-6. PMID: 23333060 DOI: http://dx.doi.org/10.1016/j.athoracsur.2012.11.024
13. Weyant MJ, Bains MS, Venkatraman E, Downey RJ, Park BJ, Flores RM, et al. Results of chest wall resection and reconstruction with and without rigid prosthesis. Ann Thorac Surg. 2006;81(1):279-85. PMID: 16368380 DOI: http://dx.doi.org/10.1016/j.athoracsur.2005.07.001
14. Tukiainen E. Chest wall reconstruction after oncological resections. Scand J Surg. 2013;102(1):9-13. PMID: 23628630 DOI: http://dx.doi.org/10.1177/145749691310200103
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
2. Universidade Federal do Ceará, Fortaleza, CE, Brazil
Institution: Hospital Universitário Walter Cantídio da Universidade Federal do Ceará, Fortaleza, CE, Brazil.
Corresponding author:
Caio Alcobaça Marcondes
Rua Luiza Amélia Brandão, 916, São Cristóvão
Teresina, PI, Brazil Zip Code 64056-170
E-mail: caio_alcobaca@hotmail.com
Article received June 26, 2014.
Article accepted May 26, 2014.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter