

Original Article - Year 2015 - Volume 30 -
Reduction mammoplasty with the use of breast implants
Mamoplastia redutora com utilização de implantes de mamários
ABSTRACT
INTRODUCTION: Advances in reduction mammoplasty techniques have contributed to make surgery safer and more acceptable to the medical community. However, in some patients, the aesthetic outcome remains unsatisfactory, mainly because of flaccid breasts and lack of projection. To achieve optimal results, the use of breast implants in conjunction with reduction mammoplasty was initiated. By combining the best of both techniques, it is possible to obtain good results and patient satisfaction. The objective of this study is to evaluate the results of reduction mammoplasty with insertion of silicone implants.
METHODS: For this study, 15 female patients aged 26 to 62 years were selected. All underwent reduction mammoplasty with immediate insertion of breast implants. Parameters such as asymmetry of the breast base and resection volume were evaluated. After the sixth postoperative month, patients reported their degree of satisfaction with the final surgery result, as dissatisfied, satisfied, or very satisfied.
RESULTS: Although presenting a paradox-removing breast tissue to place a silicone implant-breast reduction surgery with the use of implants can successfully achieve the aim of proper positioning of breast structures and providing higher durability of results, both in breast consistency and in the projection of the breast cone.
CONCLUSIONS: The technique achieved good results by increasing the durability of the surgical result and patient satisfaction.
Keywords: Mammoplasty; Breast Implants; Patient satisfaction.
RESUMO
INTRODUÇÃO: A evolução das técnicas de mamoplastia redutora contribuiu para tornar a cirurgia mais segura e, portanto, mais aceita pela comunidade médica. Contudo, em algumas pacientes, a estética final permanecia insatisfatória, principalmente, por resultar em mamas flácidas e sem projeção. Para alcançar melhores resultados, iniciou-se o uso do implante de mama em conjunto com a mamoplastia redutora. Ao agregar o melhor das duas técnicas, é possível obter bons resultados e a satisfação do paciente. O objetivo deste estudo é avaliar o resultado das cirurgias de mamoplastia redutora com a inclusão de implantes de silicone.
MÉTODOS: Para o desenvolvimento deste trabalho, foram selecionadas 15 pacientes, do sexo feminino, com idade entre 26 a 62 anos. Todas foram submetidas à mamoplastia redutora com inclusão imediata de implante mamário. Parâmetros, como assimetria da base mamária e volume da ressecção, foram avaliados. Após o sexto mês de pós-operatório, as pacientes responderam um questionário sobre o grau de satisfação do resultado final da cirurgia, que variou de insatisfeita, satisfeita e muito satisfeita.
RESULTADOS: Apesar de apresentar um paradoxo - retirar tecido mamário para colocar um implante de silicone -, a cirurgia para redução mamária com utilização implante consegue alcançar, com êxito, o objetivo de posicionar, adequadamente, as estruturas mamárias e de proporcionar maior durabilidade do resultado tanto na consistência quanto na projeção do cone mamário.
CONCLUSÃO: Pode-se concluir que a técnica apresenta bons resultados na medida em que aumenta a durabilidade do resultado cirúrgico e a satisfação das pacientes.
Palavras-chave: Mamoplastia; Implantes mamários; Satisfação do paciente.
Several reduction mammoplasty and mastopexy techniques have been described throughout the history of plastic surgery. At the beginning of the 20th century, Lexer (1912)1 and Kraske (1923)1 initiated the practice of breast reduction by means of resection en bloc. In 1925, Passot1 introduced the method of large detachment and transposition of the nipple-areola complex (NAC). This procedure was adopted for smaller breasts by Gilles and McIndoe (1939)1. A major advance, however, occurred when Schwartzmann reported a new concept of complete reduction mammoplasty in 19302, to preserve the periareolar dermis, maintain the vascularization of the areola, and thus provide higher safety in surgeries with larger detachments. Since the 1950s, reduction mammoplasty has gone through a golden era, with publications by Arie (1956)3, Strömbeck (1960)4, Pitanguy (1961)5, Dufoumentel- -Mouly (1961)1, and Skoog (1963)6. In 1975, Ribeiro et al.7,8 introduced mammoplasties using the inferior-based pedicle technique, which contributed much to the advancement of mastopexy. Since then, other procedures have been perfected for breast reduction and mastopexy.
With the development of breast implants, augmentation has accounted for the growth in the number of plastic surgeries in Brazil and worldwide. With recent developments in the implant technology, different uses are emerging. The first satisfactory silicone gel implants were employed by Cronin and Gerow (1963)1. In 1970, Ashley proposed the use of a silicone implant coated with a polyurethane foam, and a textured silicone implant was introduced in the 1980s. These developments have reduced the number of cases of capsular contracture and mammary asymmetry.
Nevertheless, some patients who underwent reduction mammoplasty complained of loss of consistency and volume of the breasts. Consequently, techniques used individually, such as reduction mammoplasty and breast implantation, have begun to be used in conjunction to achieve higher patient satisfaction.
OBJECTIVE
To assess the result of reduction mammoplasty combined with immediate insertion of silicone implants.
METHOD
Fifteen female patients aged 26 to 62 years were operated on from June 2010 to June 2012 for breasts with first and second degree ptosis (Chart 1) and mild or moderate hypertrophy (Chart 2); the exclusion criteria were smokers, prior mammary surgery, body mass index (BMI) above 30 kg/cm2, or refusal to use implants.


Routine preoperative evaluations were conducted (complete blood count and biochemical evaluations, EAS, chest radiography, and discussion of surgical risk), in addition to mammography.
All implants were placed in a retroglandular position. The chosen brand was Silimed, advanced type (conical) with a high-profile polyurethane coating. Volumes varied from 185 cc to 235 cc.
The technique chosen for the procedure was tower mammoplasty, demonstrated by Prof. Ronaldo Pontes during the academic training of the author in the Plastic Surgery Service of the Hospital Universitário Pedro Ernesto, where two patients were operated on and subsequently followed-up by the service. All surgeries were performed with general anesthesia in a hospital environment with closed unit support.
Marking
In preoperative assessment, the bases of the breasts were measured, and the differences were defined (Figure 1).
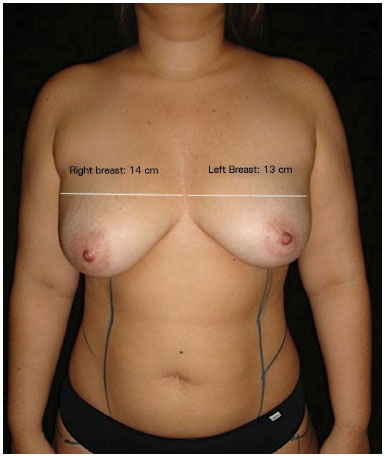
Figure 1. Marking of the breast base diameter, showing asymmetry.
The patient was positioned on the surgical table, with the torso elevated at 45° and arms abducted at 90°. Marking was initiated with a median line passing through the suprasternal notch and xiphoid process. The midclavicular point and inframammary crease were delimited (Figure 2). A line was drawn that divided the breast into two meridians, departing from the midclavicular point.
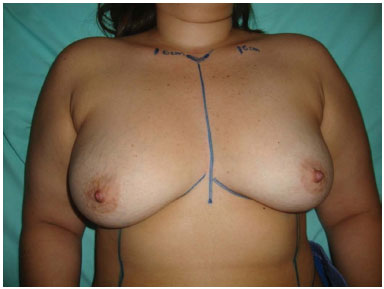
Figure 2. The median line passing through the suprasternal notch and xiphoid process. Inframammary fold delimited, and midclavicular point marked.
The projection of the inframammary crease (Point A) was marked, maintaining a distance of 18-20 cm from the suprasternal notch, followed by a point in the inframammary crease 9-10 cm from the median line (Point B) (Figure 3).
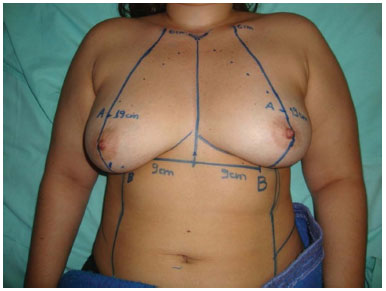
Figure 3. The division of the breast into two meridians. Marking of points A and B.
In the inframammary crease, points C and D, equidistant 2 cm from point B, were marked (Figure 4). In the case of breast asymmetry, the size difference of the base (previously measured) to the distance CD was added to the larger diameter of the breast.
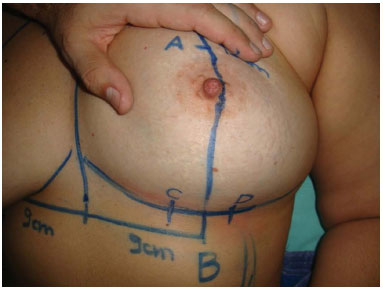
Figure 4. Marking of points C and D. Initially, this marking should not exceed 4 cm, thus avoiding large resections and difficulty in closure of the breast pillars.
A curved line was drawn, extending from point A to points C and D (Figure 5).
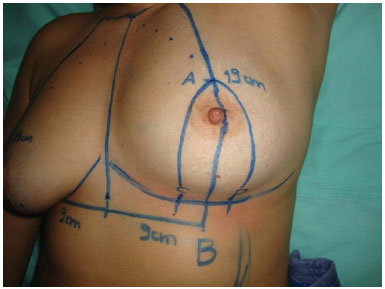
Figure 5. Marking of lines AC and AD, presenting a discrete curve similar to a tower.
Point E was marked on the midline, 1 cm from the inferior areola. The distance AE was transferred to the contralateral breast, delimiting superiorly the resection of the parenchyma (Figure 6).
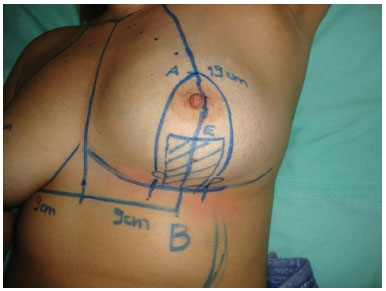
Figure 6. Point E is marked 1 cm below the inferior nipple-areola, delimiting the upper resection of the breast parenchyma. The distance AE is transferred to the other breast.
Surgery
Surgery began by applying the Schwartzmann maneuver in the periareolar region and making an incision on the previously marked lines. Using a hook, the breast was suspended from point A. The breast parenchyma was then incised (0.5 cm from the skin) perpendicular to the fascia of the greater pectoral muscle. The excess gland was removed in the shape of a keel (Figure 7). The implant pocket was then detached in the subglandular plane to receive the implant (Figure 8). After careful hemostasis, the selected implant was inserted, the areola repositioned, and the pillars sutured in three planes with nylon 3-0 and 4-0 (Figures 9 and 10). When properly sutured, the base of the resection showed two ears that were incorporated in a horizontal scar (Figure 11). The new position of the areola should be within 5.5 and 6.5 cm of the inframammary crease, as marked with an areolotome, positioned with nylon 5-0 cardinal sutures, and subsequently with intradermal Monocryl 3-0 (Figure 12).
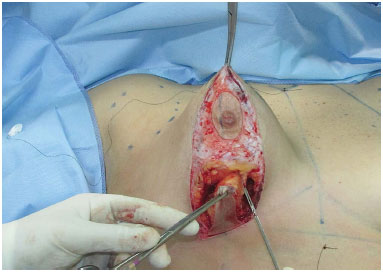
Figure 7. After the Schwartzmann maneuver, the breast tissue is incised perpendicular to the fascia of the pectoral muscle, and is removed in the shape of a keel. Note that the incision is 0.5 cm from the skin.
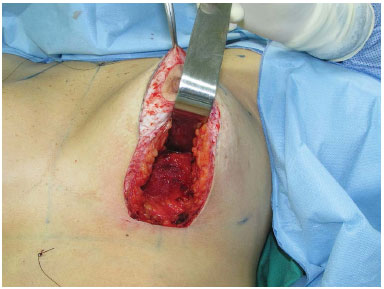
Figure 8. Overview of the inside of the dissected subglandular pocket.
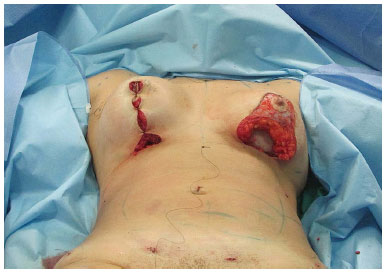
Figure 9. Breast implant positioned inside the subglandular pocket.
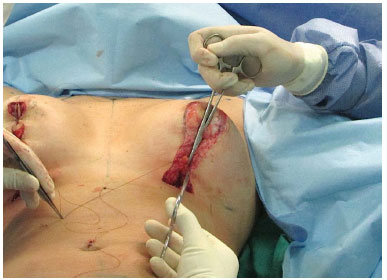
Figure 10. Suture of the breast pillars with 3-0 nylon after repositioning of the areola.
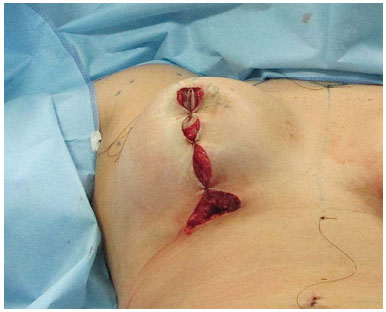
Figure 11. After the approximation of the pillars, two ears are formed at the base of the resection and should be accommodated within a horizontal scar.
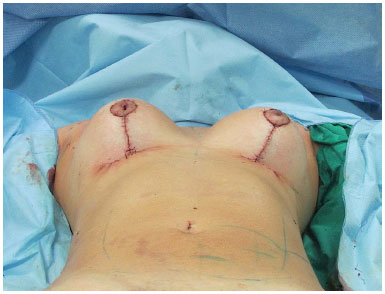
Figure 12. After intradermal suturing, the final result presents a vertical scar that varies from 5.5 cm to 6.5 cm, with a projected and well-defined breast cone.
A suction drain was used as indicated, but was not routine.
Dressing was performed with strips of sterile Micropore tape, and an occlusive dressing was applied.
The resected material was sent for pathological examination.
Postoperative care
All patients were discharged within 24 h, and any drains were removed. The patients were advised to use a surgical bra for 30 days.
Outpatient monitoring was done weekly for the first 2 weeks, and monthly for the following 3 months.
A return to work activities was permitted after 21 postoperative days, and physical exercises, such as gym and contact sports, were permitted only after 45 days.
In the sixth postoperative month, the patients were asked to rate the degree of satisfaction with the surgery, as dissatisfied, satisfied, or very satisfied.
RESULTS
The technique yielded good results with regard to the projection of the breast cone and consistency of the breasts, and increased the durability of the surgical results and patient satisfaction.
Case 1
Patient A.G.M., 33 years, G1P2, nurse, breastfed for 13 months, reported falling breasts with significant differences between the breasts. She underwent reduction mammoplasty with high-profile conical polyurethane breast implants (Advanced) (volume 185 cc) (Figure 13: A, B and C: preoperative, D, E and F: postoperative).
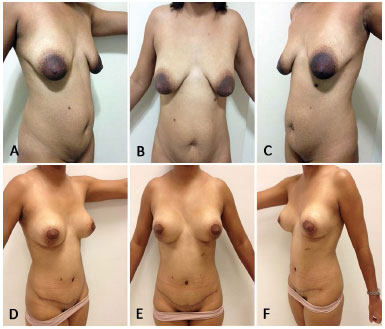
Figure 13. A, B and C preoperative - D, E e F 8 months postoperatively.
Case 2
Patient E.S.F., 35 years, G1P1, housewife, breastfed for 10 months, reported dissatisfaction with breast appearance, and complained of falling breasts and a difference in their shape. Reduction mammoplasty was performed with high-profile conical polyurethane breast implants (Advanced) (volume 235 cc) (Figure 14: A, B and C: preoperative, D, E and F: postoperative).
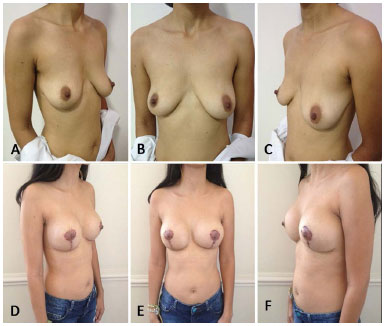
Figure 14. A, B and C preoperative - D, E and F 6 months postoperatively.
Case 3
Patient L.M.F., 43 years, G1P2, professor, reported dissatisfaction with breast appearance, and considered them fallen and voluminous. Reduction mammoplasty was performed with high-profile conical polyurethane breast implants (Advanced) (volume 185 cc) (Figure 15: A, B and C: preoperative, D, E and F: postoperative).
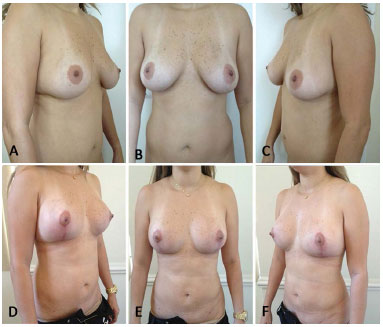
Figure 15. A, B and C preoperative - D, E and F 11 months postoperatively.
Case 4
Patient S.A.A., 28 years, G1P1, nutritionist, reported dissatisfaction with breast appearance and considered them sagging and large. Reduction mammoplasty was performed with high-profile conical polyurethane breast implants (Advanced) (volume 235 cc) (Figure 16: A, B and C: preoperative, D, E and F: postoperative).
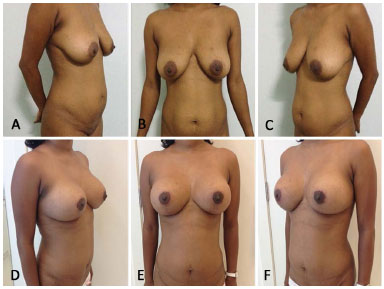
Figure 16. A, B and C preoperative - D, E and F 9 months postoperatively.
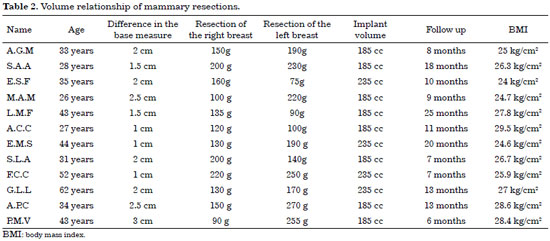
A total of 15 surgeries were performed in this study, with a variation in breast base measurements from 1 cm to 3 cm, which resulted in asymmetric resections ranging from 75 g to 270 g. The largest difference in the same surgery was 165 g, with 90 g in the right breast and 255 g in the left (Table 1).

Of the 15 patients, only one was dissatisfied with the procedure, reporting that the difference in breasts remained; two were satisfied, and 12, very satisfied.
During the postoperative follow-up, three complications were seen: one hypertrophic scar, one permanent breast asymmetry (complaint by the patient), and one dehiscence (Table 2).
DISCUSSION
The appropriate selection of patients, combined with proper technique, yielded good results with regard to the positioning of breast structures and greater durability of the result, both in consistency and the projection of the breast cone9,10.
Despite presenting a paradox-removal of breast tissue to place a silicone implant-breast reduction with the use of implants can combine the advantages of both techniques (reduction mammoplasty and breast augmentation). The results were pleasing for both patients and surgeons, mainly in terms of breasts with accentuated skin flaccidity, reduction in volume, and loss of breast consistency.
The physical examination was restricted to the indication for the technique; the skin type (flaccid or not, with or without splines) and breast parenchyma (glandular, fatty, or mixed) also must be evaluated. These variables and the assessment of breast asymmetry will directly indicate the surgical technique to be employed.
Since the use of implants in this surgery was not for augmentation, but for parenchymal replacement, implants with high projection and reduced volume were needed. Thus, the choice of conical implants was justified, as this model presents a narrow base and high projection, which allowed a reduced volume in the final result. Another positive point was that the consistency of the prosthesis did not decrease with time, in contrast to flaps, which result in a lower projection of the upper pole over time.
A more projected breast results in a longer vertical and lower horizontal scar11.
Another advantage of the technique was to facilitate breast symmetry through the unequal marking of excess skin, which allowed a larger tissue resection in breasts with larger bases and more conservative resection in smaller breasts.
The main disadvantages were as follows: the high cost of implants and the risk of complications inherent to their use, such as infection, rotation, and capsular contracture.
Since breast reduction without the use of implants is possible, this factor must be discussed with the patient, always stating the positive and negative points previously described.
CONCLUSION
Mammoplasty seeks to match the breast content and breast pocket, while maintaining a good relationship between the nipple-areola complex and inframammary crease. Several techniques exist, based on variables such as breast volume, skin flaccidity, and type of parenchyma, among others, always with the intent of seeking patient satisfaction.
The safety offered by the new generation of breast implants allows us to progress in breast surgery by proposing a mixed technique in a single surgical encounter, in which the replacement of parenchyma is associated with the placement of breast implants to allow more projected and firmer breasts.
REFERENCES
1. Mélega JM. Cirurgia estética, cirurgia plástica: fundamentos e arte. Rio de Janeiro: Medsi; 2003. p.435-545.
2. Schartzman E. Die technik der mammaplastik. Der Chirurg. 1930;2:932.
3. Ariê G. Nova técnica em mamoplastia. Rev Latinoam Cir Plast. 1957;3:22-8.
4. Strombeck JO. Reduction mammaplasty. In: Grabb WC, Smith JW, eds. Plastic surgery. Boston: Little, Brown and Co;1968.
5. Pitanguy I. Hipertrofias mamárias: estudo crítico da técnica pessoal. Rev Bras Cir. 1966;56:263.
6. Skoog T. Plastic surgery: new methods and refinements. Philadelphia: Saunders; 1974.
7. Ribeiro L, Accorsi A Jr, Buss A, Marcal-Pessoa M. Creation and evolution of 30 years of the inferior pedicle in reduction mammaplasties. Plast Reconstr Surg. 2002;110(3):960-70. PMID: 12172167 DOI: http://dx.doi.org/10.1097/00006534-200209010-00038
8. Ribeiro L, Backer E. Mastoplastia com pedìculo de seguridad. Rev Esp Cir Plast. 1973;16:223-34.
9. Saldanha OR. Uso de prótese em mamaplastia redutora. Arq Catarin Med. 2000;29(supl. 1):261.
10. Saldanha OR, Maloof RG, Dutra RT, Luz OAL, Saldanha Filho O, Saldanha CB. Mamaplastia redutora com implante de silicone. Rev Bras Cir Plást. 2010;25(2):317-24.
11. Peixoto G. Reduction mammaplasty: a personal technique. Plast Reconstr Surg. 1980;65(2):217-26. DOI: http://dx.doi.org/10.1097/00006534-198002000-00015
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
2. Universidade do Estado do Rio de Janeiro, Rio de Janeiro, RJ, Brazil
3. Faculdade de Medicina de Campos, Campos dos Goytacazes, RJ, Brazil
Institution: Hospital Universitário Pedro Ernesto, Rio de Janeiro, RJ, Brazil.
Corresponding author:
Leonardo Araujo Guimarães
Rua Barao de Icarai, 16, Flamengo
Rio de Janeiro, RJ, Brazil Zip Code 22250-110
E-mail: drleonardoguimaraes@gmail.com
Article received: July 21, 2013.
Article accepted: February 4, 2014.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter