

Reviw Article - Year 2015 - Volume 30 -
Current management for the prevention of postoperative scar hypertrophy
Condutas atuais na prevenção da hipertrofia cicatricial pós-operatória
ABSTRACT
INTRODUCTION: Considering that nearly 51 million surgeries are performed annually just in the USA, we can state that scar hypertrophy is a relevant problem, since a thin, good quality scar can be the dividing line between a good outcome and an unsuccessful surgery. The objective is to perform a bibliographic review of the noninvasive methods currently available to prevent postoperative hypertrophic scars and discuss their evidence-based effectiveness.
METHOD: A search was performed in PubMed, LILACS, and SciELO databases, using the terms "scar prevention" and "hypertrophic scars," for clinical trials, meta-analyses, and review articles published since 2004 in English or Portuguese language.
RESULTS AND CONCLUSIONS: Several studies using silicone were found, providing some evidence on its effectiveness; only 3 prospective clinical trials using Contractubex® were found; 2 controlled, randomized prospective clinical trials using 5% imiquimod were found, but only one was double-blind; one well-designed clinical trial using a micropore adhesive tape was found; a similar clinical trial using vitamin E did not show good results. Clinical trials on the use of massage and local pressure were not found. Despite the limitations of the studies, silicone is considered the first treatment option for the prevention of postoperative hypertrophic scars. There is no evidence proving the effectiveness of micropore adhesive tape, massage, local pressure, Contractubex, 5% imiquimod, or vitamin E.
Keywords: Hypertrophic scar; Hypertrophic scar/Prevention & Control; Cicatrization; Silicone; Vitamin E.
RESUMO
INTRODUÇÃO: Considerando um número estimado de cerca de 51 milhões de cirurgias a cada ano apenas nos EUA, podemos dizer que a hipertrofia cicatricial é um problema relevante, já que uma cicatriz fina, de boa qualidade, pode ser a linha divisória entre um bom resultado e uma cirurgia malsucedida. O objetivo é fazer uma revisão bibliográfica acerca dos métodos de tratamento não invasivos atualmente disponíveis para a prevenção da hipertrofia cicatricial pós-cirúrgica e discutir a sua eficácia baseada em evidências.
MÉTODO: Foi realizada uma pesquisa nas bases de dados Pubmed, Lilacs e SciELO, utilizando os termos "scar prevention" and "hypertrophic scars", por ensaios clínicos, meta-análises e artigos de revisão publicados a partir de 2004, em inglês ou português.
RESULTADOS E CONCLUSÕES: Foram encontrados vários trabalhos utilizando o silicone, proporcionando alguma evidência acerca da sua eficácia; foram encontrados apenas três ensaios clínicos prospectivos relacionados ao uso do Contractubex®; dois ensaios clínicos prospectivos, controlados, randomizados, sendo apenas um deles duplo-cego, com o imiquimode a 5%; foi encontrado apenas um ensaio clínico bem desenhado utilizando o esparadrapo microporoso e outro trabalho relacionado ao uso da vitamina E, que não mostrou bons resultados; não foram encontrados ensaios clínicos sobre o uso da massagem e da pressão local. Apesar das deficiências dos estudos, o silicone é considerado a primeira opção na prevenção da hipertrofia cicatricial pós-cirúrgica. Não há evidências que comprovem a eficácia do esparadrapo microporoso, da massagem, da pressão local, do Contractubex, do imiquimode a 5% e da vitamina E.
Palavras-chave: Cicatriz hipertrófica; Cicatriz hipertrófica/Prevenção & Controle; Cicatrização; Silicones; Vitamina E.
A hypertrophic scar is defined as "a reddish, elevated, sometimes pruritic scar, confined within the margins of the original incision"1. Scars may be painful, limit joint movement, cause psychological stress, and can be detrimental to social life. Considering that nearly 51 million surgeries are performed annually in the USA alone2, we can state that postoperative scar hypertrophy is a relevant problem, since a thin, good quality scar can be the dividing line between a good outcome or an unsuccessful surgery (Figure 1). Considering its importance, several noninvasive methods are available for prevention of postoperative hypertrophic linear scars: massage, local pressure, micropore adhesive tape, silicone, formulations containing onion extract (Allium cepa), vitamin E, and 5% imiquimod. Thus, the aim of this study was to perform a bibliographic review of the noninvasive treatments currently available for prevention of postoperative hypertrophic scars and to discuss their evidence-based effectiveness.
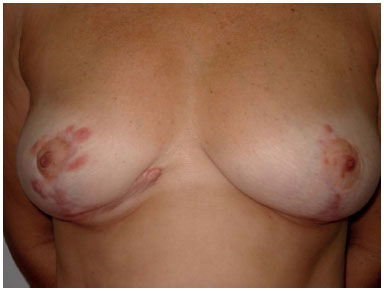
Figure 1. Scar hypertrophy after breast reduction surgery.
METHOD
A search of the PubMed, LILACS, and SciELO databases was performed, using the terms "scar prevention" and "hypertrophic scars," for studies published since 2004 in English and Portuguese. The following inclusion criteria were used for selecting the studies: clinical trials, meta-analyses, and review articles on noninvasive topical therapies. The most relevant prospective clinical trials cited in the review articles, but unrelated to the initial search because they were published before 2004, were also included. Studies published in other languages, experimental studies, studies on treatment of burn scars or scar hypertrophy, or those using infiltration treatments or laser were excluded.
RESULTS
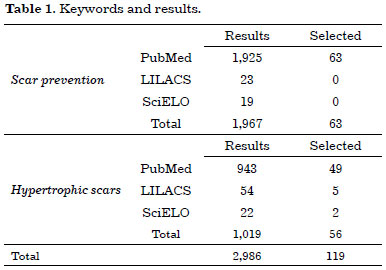
A total of 2,986 references were obtained, from which 119 were selected for the review (Table 1).
The following noninvasive methods were used to prevent postoperative hypertrophic scars found in the selected studies: scar massage, local pressure, micropore adhesive tape, silicone, A. cepa, vitamin E, and 5% imiquimod.
Massage
A study published in 2010 that aimed to validate the POSAS® scale (Patient and Observer Scar Assessment Scale) for facial scars described division of 30 patients into 2 groups, the first group being treated with silicone gel and the second group with local massage. After 2 months, the patients were evaluated using the POSAS® scale, and similar good scores were obtained in both groups3.
In 2012, a review article was published on the role of massage in the management of scars; of the 10 original articles reviewed in that study, 8 were prospective and 2 were case studies. Of the total 147 patients who received massage therapy in those studies, only 30 had postoperative scars. The authors concluded that additional, better-designed clinical trials, using objective evaluation criteria were required in order to establish evidence-based recommendations4.
Local pressure
Studies relating to the use of pressure devices for the prevention of postoperative hypertrophic scars were not found.
Micropore adhesive tape
In 1995, Reiffel5 published a prospective study with 64 patients who were treated with micropore adhesive tape applied in the longitudinal direction of scars and kept in place for 2 months or more, with replacement as and when necessary. The author concluded that "the majority of patients demonstrated complete satisfaction."
A randomized clinical trial on a control group was published in 2005, in which 70 patients who underwent a cesarean section had micropore adhesive tape applied to the scar soon after the removal of sutures. The study concluded that the micropore adhesive tape significantly reduced the volume of the scar, as assessed by ultrasonography, by an average 0.16 cm3 (95% confidence interval [CI], 0.05-0.36). The effect of treatment was greater when considering only those women who agreed to use the adhesive tape during 12 weeks of follow-up6. Four patients (12%) had an adverse reaction (cutaneous rash) at the site of contact with the tape within the first 6 weeks of use, and they were excluded from the study. The use of adhesive tape was discontinued and the local reactions spontaneously resolved. None of the 39 patients, who completed 12 weeks of treatment and 6 months of follow-up, developed a hypertrophic scar compared with 12 patients (41%) in the control group.
Silicone
In 1991, Ahn et al.7 published a prospective study wherein a curative product containing silicone gel was applied for 12 hours daily to 21 surgical scars and observed that the treated scars gained less volume after 1 and 2 months of treatment when compared with control scars.
In 1996, a prospective clinical study was published, evaluating 20 patients who underwent reduction mammoplasty and who used a silicone strip on the scars on only one of the breasts for 12 hours daily for 60 days. Of the untreated scars, 60% had hypertrophy, whereas only 25% of the treated scars had hypertrophy8.
In 2001, Gold et al.9 published a controlled, randomized clinical trial, in which 96 patients submitted to a dermatologic surgery were stratified into 2 groups, with one group having a normal healing history (low-risk) and the other group with a cicatrization history. Within each group, patients were randomly divided into 2 groups: the first had a silicone strip placed on the scar 48 hours after surgery; this was used 24 hours a day for 6 months. In the second group, the usual postoperative follow-up was performed. Of a total of 66 patients, 31 in the low-risk group and 35 in the high-risk group completed 6 months of study. In the low-risk group, no hypertrophic or keloid scar was observed in patients who used the silicone strip and only one patient who received routine postoperative care developed a hypertrophic scar. In the high-risk group of 35 patients, 17 used the silicone strip and 18 had routine care. Of those who used the silicone, 71% (12) did not have a hypertrophic or keloid scar, but 29% (5) had a hypertrophic scar; of the 18 patients who received routine care after surgery, 39% (7) did not have a hypertrophic scar, 17% (3) developed keloids, and 44% (8) developed a hypertrophic scar.
In 2005, a randomized, double-blind, prospective clinical trial with a control group was performed with 50 patients who underwent sternotomy. The patients received 2 envelopes, one containing gel silicone and the other containing placebo, and they were directed to apply the respective contents to either half of the scar. After 3 months of follow-up, a better score was observed using the Vancouver Scale in the half of the scar that received the silicone application, when compared to the control group, with a statistically significant improvement in the evaluated parameters, including pigmentation, vascularization, malleability, elevation, pain, and redness10.
In 2007, Signorini et al.11 published a study with 160 patients assessing the effectiveness of a new, clear, self-drying, silicone gel (Dermatix®). Patients who had undergone surgery 10 days to 3 weeks prior for removal of skin lesions were divided into 2 groups; in the first group, the silicone gel was applied twice daily for 4 months, with additional applications recommended after a shower and physical activities. Conventional treatments were prescribed for the control group such as local pressure, corticosteroid injection, or conventional silicone gel. All patients were monitored monthly for 4 months, and the final evaluation was performed after 6 months. Of the 80 patients in the group treated with silicone, 72 completed the study and 76 of 80 patients in the control group completed 6 months of observation. The authors observed that 67% of the treated scars could be classified as grade 1 (normal: flat, soft, normal color) compared with 28% of the scars treated by other methods; 26% were grade 2 (mild hypertrophic: slightly elevated, moderately hard, light pink color) compared with 46% of the control group; and 7% were grades 3 and 4 (hypertrophic and keloidal) compared with 26% of the control group.
In 2009, De Giorgi et al.12 published a randomized clinical trial, in which 110 patients who underwent skin injury removal were divided into treatment and control groups, and were followed for 8 months with continuous use of silicone gel (twice a day for 60 day). At the end of the follow-up period, of the 65 patients who used silicone gel only 15% (10) had an extended scar, 9% (6) had a hypertrophic scar, and none developed a keloid scar. Of the 45 patients in the control group, 18% (8) had an extended scar, 22% (10) had a hypertrophic scar, and 11% (5) developed a keloid scar.
In 2010, Radwanski et al.13 published a prospective, noncontrolled study of 128 patients with recent scars in the remodeling phase (between the removal of sutures and the third month after surgery) in whom silicone gel was applied (Kelocote®) over the entire length of the scar twice a day for 3 to 6 months. Of the total patients, only 6 completed the 3 steps of evaluation and the 6 months of the study. Using the Vancouver Scale, the authors observed a significant improvement in the treated scars (p < 0.0001) during the treatment period, in particular between the initial and intermediate point (in the first 3 months).
In 2010, Rhee et al.14 published a prospective study with a control group, in which 40 patients who underwent minor surgeries in exposed areas of the body were assessed; a silicone strip was used on the scar in 20 of these patients 12 hours daily for 3 months. At the end of the study period, the authors observed a statistically significant improvement in the evaluated parameters, including pigmentation (p = 0.0002), vascularization (p = 0.0002), and elevation of the scars (p < 0.0001).
In 2011, Sakuraba et al.15 published a study of 9 patients who underwent sternotomy, in which the silicone gel was used directly on the surgical incision from 2 weeks after the surgery onwards 24 hours a day. At the end of 24 weeks, none of the patients had keloid scars.
In 2013, Medhi et al.16 published a prospective study of 33 patients with recent postoperative scars; the patients underwent surgery 10 days to 3 weeks prior and silicone gel was applied twice a day for 3 months, with evaluation using the Vancouver Scale. After 3 months, a decrease in elevation was observed in 57.6% (19/33) of scars, with significant improvements in pigmentation, flexibility, and vascularization.
In 2013, Riedel et al.17 published a study of 20 patients who underwent removal of a costal cartilage segment; half of the scar was treated with silicone gel for 3 months and the other half was used as a control. The scars were evaluated visually; of 19 patients, only 8 showed improvement, and 10 of 19 showed worsening on visual evaluation. The authors concluded that there were no benefits from using silicone gel according to the parameters used.
In 2013, a meta-analysis was published, evaluating 20 clinical trials on the use of silicone in the prevention and treatment of hypertrophic scars. The study demonstrated that silicone reduced the incidence of hypertrophic scars in high-risk patients (relative risk [RR] 0.46, 95% CI) when compared with absence of scar treatment; however, the authors emphasized that these studies were highly susceptible to bias and concluded that evidence for benefit was poor18.
Onion extract (A. cepa)
Onion extract (A. cepa) is a common ingredient in some formulations used for the topical therapy of scars. Contractubex® (Merz Pharma, Frankfurt, Germany) is a gel containing 10% onion extract (A. cepa: Cepalina®), 50 IU of heparin, and 1% allantoin.
In 1994, Willital and Heine19 published a study of 45 patients who underwent thoracic surgery and who were randomly divided into two groups, with one group using Contractubex® daily for one year. The authors reported a reduction in the width of the scars and fewer hypertrophic and keloid scars in the treated group than in the control group. The authors did not observe a difference in the length or elevation of the scars. After one year of observation, the scars were classified as "very good" or "good" in more than 90% of patients who used the formulation and as "good" or "moderate or poor" among nearly 40 and 60% of untreated patients, respectively.
In 1995, Maragakis et al.,20 published a clinical trial with 65 children after thoracic surgeries who were randomly divided into 2 groups; one group used the onion extract formulation on the scar for 6 months. The authors reported that 84% of the treated scars were classified as "good" or "very good" compared to 59% of the scars in the control group and that size increase of the treated scars was "distinctly" less than in those who were not treated; in addition, "very good" tolerability to the product was reported in 37 of the 38 patients treated.
In 2003, De Vita Júnior et al.21 published a randomized, double-blind clinical trial with a control group, with 60 patients who underwent reduction mammoplasty or abdominoplasty and who applied the onion extract product or placebo on the scars from the 21st day after surgery onwards. Pictures of the scars were evaluated by 3 surgeons who were not part of the study and through clinical examinations performed by the researchers themselves. The authors concluded that topical use for 6 months significantly improved the quality of the postoperative scars.
Another study published in 2006, this time retrospective and multicentric, with 771 patients (555 treated with Contractubex® and 216 with intralesional corticosteroid injection), showed that Contractubex® was significantly more effective than the corticosteroid in reducing the erythema, pruritus, and consistency of the hypertrophic scar22.
In 2013, Willital and Simon23 published a noncontrolled, observational study, in which 1,268 patients were treated with Contractubex® by several different physicians, and described a significant improvement in color, flexibility, and size of scars, with an average reduction of 47.8% in elevation and 32.5% in width. The authors concluded that Contractubex® could be effective in the early treatment of scars and could help in the prevention of hypertrophic scars; however, a well-designed study would be required to prove effectiveness.
A review article published in 2014 by Rabello et al.24 concluded that the data were not sufficient to demonstrate the effectiveness of Contractubex, and no significant improvement in hypertrophic scars was observed in those patients.
5% Imiquimod
Imiquimod is an immunomodulator recommended for the treatment of genital warts, actinic keratosis, and superficial basal cell carcinoma25. Its mechanism of action is not fully known, but imiquimod seems to induce the production of proinflammatory cytokines, such as interferon-alpha (INF-alpha), tumor necrosis factor alpha (TNF-alpha), and interleukins, thereby increasing collagen degradation. Based on this, Berman and Kaufman26 published a prospective clinical trial in 2002, wherein they evaluated the effect of imiquimod in the local recurrence of keloid scars after surgical excision. Patients were instructed to apply 5% imiquimod daily to the scar for 2 months. Of the 11 keloid scars that completed 24 weeks of observation, no local recurrence was observed26.
In 2005, Prado et al.27 published a controlled, double-blind, randomized prospective clinical study of 15 patients who underwent reduction mammoplasty and were divided into 3 groups; in the first group, 5% imiquimod (Aldara®, 3M) was applied to the left breast and the right breast was used as the control; in the second group, the right breast was treated and the left one received petrolatum; a double-blind treatment was performed in the breasts of patients in the third group using Aldara® and petrolatum. This study concluded that almost all scars treated with Aldara® were less elevated, had coloration similar to skin around the scar, and scored better on the evaluation scale.
In 2009, Berman et al.28 published a clinical trial of 20 patients whose keloid scars were excised by shaving; the patients were randomly divided into 2 groups and instructed to apply 5% imiquimod daily for 2 weeks beginning on the evening after surgery, and then only 3 times per week as a curative. Only 8 patients completed the 6 months of the study. The authors reported that the local recurrence rate of keloids was 37.5% in the treated group and 75% in the control group (p = 0.54)28.
Vitamin E
Vitamin E is a fat-soluble vitamin comprising a group of 8 structurally-related compounds: tocopherols and tocotrienols. In 1999, a randomized, double blind clinical trial was published wherein 15 patients were submitted to Mohs micrographic surgery and were treated with an ointment containing 320 IU/g of d-α tocopherol (vitamin E), twice a day for 4 weeks. The authors concluded that 90% of patients showed no improvement in scar quality and 33% had contact dermatitis29.
In 2010, a single blind, prospective study with a control group was published, with 428 patients who initiated treatment at the incision site with topical vitamin E 15 days before surgery, and then applied it twice a day for 30 days in the postoperative period30. After 6 months, the family members were asked to respond to a questionnaire based on the Vancouver Scale. As early as 10 days after the treatment, the questionnaire identified a statistically significant difference (p = 0.004) in the aesthetic result between the treated groups and the control. At the end of 6 months, the questionnaire showed that 96% of patients in the treated group showed a "very good aesthetic result" and 4% showed a "good" result, whereas the control group showed that only 78% of family members considered the result to be "very good," and 15% considered the result to be "poor"30.
Another double-blind, randomized prospective study with a control group was published in 2010, with 122 patients with postoperative scars who were treated for 6 weeks with topical 5% tocotrienol; no significant difference was observed when compared with the control group using the POSAS® scale via pictures and a laser Doppler imaging device that accurately demonstrates skin blood perfusion31.
DISCUSSION
In order to recommend a therapeutic method, scientific evidence for the effectiveness of the method is required. Therefore, the use of evidence-based medicine has become more common in scientific medical practice. According to the Centre for Evidence-Based Medicine in Oxford, UK32, published studies are classified according to levels of evidence, and treatments may only be recommended to patients if classified at least in category B, with grades of recommendation that range from A to D32. To be classified in category B (recommendation), the treatment methods should find support in studies classified in evidence levels II, III, or IV. In order to attain this classification, the studies should be systematic reviews, controlled and random clinical trials with a narrow confidence Interval, therapeutic results of the type "All or Nothing", cohort studies, observation of therapeutic results, or case-control studies32. Based on these factors, the authors conducted this discussion.
Massage
The massage of scars is routinely applied at various specialized burn treatment centers4. The few available studies were performed on burn scars and failed to demonstrate benefit in vascularization, malleability, and elevation of hypertrophic scars. Nevertheless, several plastic surgeons recommend beginning massage as soon as the sutures are removed and the re-epithelialization of the wound occurs, generally with the aid of a moisturizing substance. For example, Foo and Tristani-Firouzi33, in their review article, acknowledged that it is not feasible to recommend massage as a treatment method for scars; nevertheless, they recommend that their patients begin massage 4 weeks after surgery, 2 to 3 times a day for 3 to 5 minutes with the use of a moisturizing cream, for 3 to 4 months. In their personal experience, this simple measure resulted in the improvement of the borders and thickness of scars.
Local pressure
Studies regarding the use of local pressure for preventing postoperative hypertrophic scars were not found. Currently, pressure-stretch vests are used for the prevention and control of hypertrophic burn scars34. Even when applied on burn scars, data on effectiveness are controversial and offer little scientific evidence34-37.
Micropore adhesive tape
Despite the fact that the use of micropore adhesive tape is a common practice among plastic surgeons to treat surgical scars in order to prevent scar hypertrophy, only 2 prospective clinical trials were found that aimed to assess its effectiveness: a study by Reiffel5, published in 1995, and a study performed on cesarean scars in 20056. The former was simply an observational case series, with a final subjective evaluation being reported by the author, with low-level evidence (evidence level IV: case series, with no control group). In contrast, the study published in 2005 was a randomized clinical trial with a control group and a blinded examiner, and thus had a greater evidence level (Evidence Level 1b: randomized clinical trial, with a small confidence interval). However, only 39 patients, a very small number of cases, completed the 6 months of follow-up6. In the past, the micropore adhesive tape was believed to provide "support" to the scar, without defining what "support" could be. Currently, a mechanical effect is believed to exist, equivalent to pressure; an occlusive effect, mimicking the stratum corneum, improves skin hydration, similar to the mechanism provided by the silicone. Lacking a larger number of randomized, double-blind clinical trials with control groups (Evidence Level 1b or 2b), stating whether the use of hypoallergenic micropore adhesive tape is effective is not possible. The tape is not invasive, is cheap and easy to use, and has few associated complications, which can be interrupted by removal6; it is only indicated in patients at a low risk for scar hypertrophy development or even before the beginning of silicone therapy38. It should be kept in place for at least 12 weeks, since a loss of effectiveness was observed if used for a shorter duration in the only available randomized clinical trial with a control group6.
Silicone
The mechanism of action of silicone is not fully understood, but it is believed that the benefits are associated with occlusion and hydration of scars. Approximately one or two weeks after tissue injury, an increase in transepidermal water loss (TEWL) occurs, leading to stratum corneum dehydration. Consequently, activation of interleukin 1 (IL-1) occurs, stimulating proliferation of keratinocytes, with the aim of restoring barrier function and preventing dehydration. In contrast, IL-1 signals the fibroblasts in the dermis to increase the production of collagen. Excessive production of collagen is responsible for abnormal cicatrization. The use of silicone over scars restores the barrier function of the stratum corneum, reducing the TEWL, interrupting the stimulation of keratinocytes, and terminating the stimulation of fibroblasts in dermis, thus normalizing the production of collagen (Figures 2 and 5)39-42.
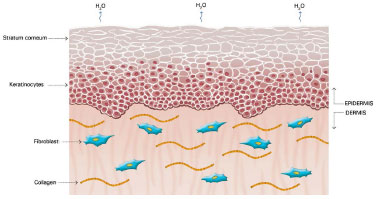
Figure 2. Normal Skin. Modified from: Mustoe TA. Evolution of silicone therapy and mechanism of action in scar management.
Source: Aesth Plast Surg. 2008;32(1):82-9242. Author of illustration: Luciane dos Santos Mori.
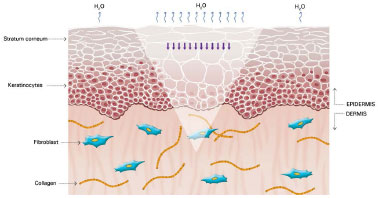
Figure 3. Cells from the stratum corneum signaling keratinocytes and the increase water loss in the form of vapor. Modified from: Mustoe TA. Evolution of silicone therapy and mechanism of action in scar management.
Source: Aesth Plast Surg. 2008;32(1):82-9242. Author of illustration: Luciane dos Santos Mori.
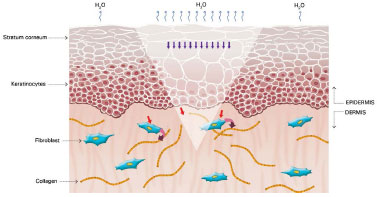
Figure 4. Keratinocytes stimulating fibroblasts to produce collagen. Modified from: Mustoe TA. Evolution of silicone therapy and mechanism of action in scar management.
Source: Aesth Plast Surg. 2008;32(1):82-9242. Author of illustration: Luciane dos Santos Mori.
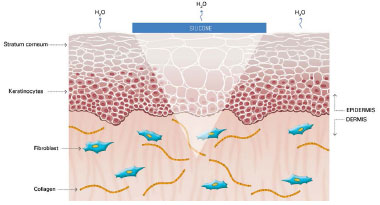
Figure 5. Occlusion by silicone restores water vapor loss by the scar to normal and interrupts the stimulus to the production of collagen by fibroblasts. Modified by: Mustoe TA. Evolution of silicone therapy and mechanism of action in scar management.
Source: Aesth Plast Surg. 2008;32(1):82-9242. Author of illustration: Luciane dos Santos Mori.
The beneficial effects of silicone were initially described for burn scars in 198343. A few years later, the first noncontrolled studies were published documenting successful use of silicone strips in hypertrophic scar treatment and keloids44-46. Since 1991, several other studies have been published providing evidence that silicone is effective in both the prevention7-10,12-16 and treatment of postoperative hypertrophic or traumatic scars47,48. Nevertheless, a majority of these studies have limitations: small sample size7-9,13-16,48-50, lack of objectivity in the evaluation criteria10-16,48-50, lack of control group13,15,16, and lack of standardization in the selection of patients7,8. Indeed, 2 studies concluded that silicone was not able to prevent hypertrophy in postoperative scars17,51. Niessen et al.51 stated that they started the treatment too early, 2 days after surgery, while still in the inflammatory phase.
Therefore, considering the existing evidence and advantages over other prevention methods, such as low cost and absence of severe adverse reactions, silicone is considered by several authors to be the first option for postoperative scar management and prevention of hypertrophic scars33,36-38,52-55.
The most frequently used polymer in the composition of the products is polydimethylsiloxane. Depending on the length of its chain and crosslinking level, silicone may be liquid, gel, or rubber. The choice of the form of application depends mainly on acceptance by the patient, since all forms have been shown to be effective; the gel may be preferable for exposed regions, such as the face, or for the sites, such as joints, where maintaining a rubber strip in contact with the scar is not possible without the support of adhesive tape. It is recommended for use soon after surgical closure when the incision is fully epithelialized38, which occurs after approximately 2 weeks, and can be maintained for up to 12 weeks54,56. It should be used for a minimum of 12 hours daily and, if possible, 24 hours a day38. At the end of 12 weeks, if the scar shows any signs of hypertrophy, the product should be used for another 12 weeks together with local pressure therapy for a total of 6 months54. The same is not recommended for 20% silicone cream, since it has only shown satisfactory results when used under occlusion in noncontrolled studies in a small number of patients57,58.
Onion extract (Allium cepa)
The first studies on the use of Contractubex® were published in 1994-95 by Willital (author and coauthor)19,20 and Maragakis20. These were controlled and randomized clinical trials, which could have been classified as evidence level 1b (randomized clinical trials with small confidence interval) because they had a greater sample size, and were submitted to statistical tests. In addition, they used subjective criteria to evaluate the results. Willital published a prospective multicenter study with a great number of cases in 2013, but without a control group, and the assessment of the results was subjective and performed by different evaluators23. Notably, the latter study23 was supported by Merz Pharma, which was the manufacturer of the product, and the author states in the study that he received financial support from the company for research and consultancy23. The authors from the other studies did not declare any conflict of interest21,22.
In order to recommend a therapy or the use of a product, more studies of evidence level II, III, or IV are required. Therefore, recommending the use of Contractubex® for the prevention of postoperative hypertrophic scars is still not possible.
5% Imiquimod
Studies conducted by Berman and Kaufman26, Prado et al.27, and Berman et al.28 included a very small number of cases, and one study did not even include a control group26. Furthermore, Prado et al., concluded that "other studies are required, using greater sample of cases and for longer periods of follow-up"27. It is worth mentioning that the study published in 2002 by Berman & Kaufman was supported by an educational grant of 3M Pharmaceuticals, the manufacturer of Aldara, and that Berman reported being a researcher and consultant for the same company28 in 2009. Therefore, new studies with higher evidence levels and with more significant findings are required in order to determine the role of topical 5% imiquimod in the management of postoperative hypertrophic scars.
Vitamin E
Evidence supporting the use of topical vitamin E for the healing of skin wounds is scarce. Studies of evidence level II or III were not found that would support recommendation to this therapy. The first study evaluating its use reported that it did not improve the appearance of scars and high rates of adverse effects were observed29 The two studies published in 2010, despite having a larger case series, were lacking in the method of evaluation: in the study conducted with 428 cases, the evaluators were family members of the patients, whereas in the study conducted with 128 patients, the results were evaluated with the use of the POSAS® scale and through the use of pictures, which are considered subjective methods of evaluation, with great variability depending on the examiner. Therefore, more studies that can provide significant results are required before recommending vitamin E for the prevention of hypertrophic scars.
CONCLUSION
Despite the shortcomings of existing studies, silicone is considered the first option for the prevention of postoperative scar hypertrophy and is recommended as soon as the scar is fully epithelialized, which occurs approximately 2 weeks after surgery.
Although its effectiveness has not been validated, micropore adhesive tape may be used in low-risk patients for the prevention of postoperative scar hypertrophy.
With the lack of evidence, we are not able to recommend massage, local pressure, onion extract (A. cepa), 5% imiquimod, or vitamin E for the prevention of postoperative hypertrophic linear scars.
REFERENCES
1. Peacock EE Jr, Madden JW, Trier WC. Biologic basis for the treatment of keloids and hypertrophic scars. South Med J. 1970;63(7):755-60. PMID: 5427162
2. Centers For Disease Control and Prevention. FastStats - Inpatient Surgery [Citado 28 Out 2015]. Disponível em: http://www.cdc.gov/nchs/fastats/inpatient-surgery.htm
3. Bianchi FA, Roccia F, Fiorini P, Berrone S. Use of Patient and Observer Scar Assessment Scale for evaluation of facial scars treated with self-drying silicone gel. J Craniofac Surg. 2010;21(3):719-23. DOI: http://dx.doi.org/10.1097/SCS.0b013e3181d841af
4. Shin TM, Bordeaux JS. The role of massage in scar management: a literature review. Dermatol Surg. 2012;38(3):414-23. DOI: http://dx.doi.org/10.1111/j.1524-4725.2011.02201.x
5. Reiffel RS. Prevention of hypertrophic scars by long-term paper tape application. Plast Reconstr Surg. 1995;96(7):1715-8. DOI: http://dx.doi.org/10.1097/00006534-199512000-00031
6. Atkinson JA, McKenna KT, Barnett AG, McGrath DJ, Rudd M. A randomized, controlled trial to determine the efficacy of paper tape in preventing hypertrophic scar formation in surgical incisions that traverse Langer's skin tension lines. Plast Reconstr Surg. 2005;116(6):1648-56. DOI: http://dx.doi.org/10.1097/01.prs.0000187147.73963.a5
7. Ahn ST, Monafo WW, Mustoe TA. Topical silicone gel for the prevention and treatment of hypertrophic scar. Arch Surg. 1991;126(4):499-504. PMID: 2009067 DOI: http://dx.doi.org/10.1001/archsurg.1991.01410280103016
8. Cruz-Korchin NI. Effectiveness of silicone sheets in the prevention of hypertrophic breast scars. Ann Plast Surg. 1996;37(4):345-8. DOI: http://dx.doi.org/10.1097/00000637-199610000-00001
9. Gold MH, Foster TD, Adair MA, Burlison K, Lewis T. Prevention of hypertrophic scars and keloids by the prophylactic use of topical silicone gel sheets following a surgical procedure in an office setting. Dermatol Surg. 2001;27(7):641-4.
10. Chan KY, Lau CL, Adeeb SM, Somasundaram S, Nasir-Zahari M. A randomized, placebo-controlled, double-blind, prospective clinical trial of silicone gel in prevention of hypertrophic scar development in median sternotomy wound. Plast Reconstr Surg. 2005;116(4):1013-20. DOI: http://dx.doi.org/10.1097/01.prs.0000178397.05852.ce
11. Signorini M, Clementoni MT. Clinical evaluation of a new self-drying silicone gel in the treatment of scars: a preliminary report. Aesthetic Plast Surg. 2007;31(2):183-7. DOI: http://dx.doi.org/10.1007/s00266-005-0122-0
12. de Giorgi V, Sestini S, Mannone F, Papi F, Alfaioli B, Gori A, et al. The use of silicone gel in the treatment of fresh surgical scars: a randomized study. Clin Exp Dermatol. 2009;34(6):688-93. DOI: http://dx.doi.org/10.1111/j.1365-2230.2008.03096.x
13. Radwanski HN, Correa WEMY, Refosco TJ, Farrapeira Júnior A. Pitanguy I. Silicone gel em cicatrizes de cirurgia plástica: estudo clínico prospectivo. Rev Bras Cir Plást. 2010;25(3):428-33. DOI: http://dx.doi.org/10.1590/S1983-51752010000300005
14. Rhee SH, Koh SH, Lee DW, Park BY, Kim YO. Aesthetic effect of silicone gel on surgical scars in Asians. J Craniofac Surg. 2010;21(3):706-10. DOI: http://dx.doi.org/10.1097/SCS.0b013e3181d83fec
15. Sakuraba M, Takahashi N, Akahoshi T, Miyasaka Y, Suzuki K. Use of silicone gel sheets for prevention of keloid scars after median sternotomy. Surg Today. 2011;41(4):496-9. DOI: http://dx.doi.org/10.1007/s00595-010-4265-5
16. Medhi B, Sewal RK, Kaman L, Kadhe G, Mane A. Efficacy and safety of an advanced formula silicone gel for prevention of post-operative scars. Dermatol Ther (Heidelb). 2013;3(2):157-67. DOI: http://dx.doi.org/10.1007/s13555-013-0036-8
17. Riedel M, Brinckmann J, Steffen A, Nitsch S, Wollenberg B, Frenzel H. Influence of silicone gel on standardized postoperative scars. J Dtsch Dermatol Ges. 2013;11(5):412-9. DOI: http://dx.doi.org/10.1111/ddg.12011
18. O'Brien L, Jones DJ. Silicone gel sheeting for preventing and treating hypertrophic and keloid scars. Cochrane Database Syst Rev. 2013;9:CD003826.
19. Willital GH, Heine H. Efficacy of Contractubex gel in the treatment of fresh scars after thoracic surgery in children and adolescents. Int J Clin Pharmacol Res. 1994;14(5-6):193-202.
20. Maragakis M, Willital GH, Michel G, Görtelmeyer R. Possibilities of scar treatment after thoracic surgery. Drugs Exp Clin Res. 1995;21(5):199-206.
21. De Vita Júnior J, Cezaretti JA, Tavarez PH, Vieira SAT, Oliveira MSL, Ávila DEP, et al. Avaliação da ação da associação de cepalin®, alantoína e heparina sódica* na evolução do processo de cicatrização de incisões cirúrgicas. Rev Bras Med. 2007;64(1):42-9.
22. Beuth J, Hunzelmann N, Van Leendert R, Basten R, Noehle M, Schneider B. Safety and efficacy of local administration of contractubex to hypertrophic scars in comparison to corticosteroid treatment. Results of a multicenter, comparative epidemiological cohort study in Germany. In Vivo. 2006;20(2):277-83.
23. Willital GH, Simon J. Efficacy of early initiation of a gel containing extractum cepae, heparin, and allantoin for scar treatment: an observational, noninterventional study of daily practice. J Drugs Dermatol. 2013;12(1):38-42.
24. Rabello FB, Souza CD, Farina Júnior JA. Update on hypertrophic scar treatment. Clinics (Sao Paulo). 2014;69(8):565-73. DOI: http://dx.doi.org/10.6061/clinics/2014(08)11
25. Berman B. Imiquimod: a new immune response modifier for the treatment of external genital warts and other diseases in dermatology. Int J Dermatol. 2002;41 Suppl 1:7-11. DOI: http://dx.doi.org/10.1111/j.1365-4632.2002.00018.x
26. Berman B, Kaufman J. Pilot study of the effect of postoperative imiquimod 5% cream on the recurrence rate of excised keloids. J Am Acad Dermatol. 2002;47(4 Suppl):S209-11. PMID: 12271279 DOI: http://dx.doi.org/10.1067/mjd.2002.126585
27. Prado A, Andrades P, Benitez S, Umaña M. Scar management after breast surgery: preliminary results of a prospective, randomized, and double-blind clinical study with aldara cream 5% (imiquimod). Plast Reconstr Surg. 2005;115(3):966-72. PMID: 15731724 DOI: http://dx.doi.org/10.1097/01.PRS.0000153823.52784.7E
28. Berman B, Harrison-Balestra C, Perez OA, Viera M, Villa A, Zell D, et al. Treatment of keloid scars post-shave excision with imiquimod 5% cream: A prospective, double-blind, placebo-controlled pilot study. J Drugs Dermatol. 2009;8(5):455-8.
29. Baumann LS, Spencer J. The effects of topical vitamin E on the cosmetic appearance of scars. Dermatol Surg. 1999;25(4):311-5. DOI: http://dx.doi.org/10.1046/j.1524-4725.1999.08223.x
30. Zampieri N, Zuin V, Burro R, Ottolenghi A, Camoglio FS. A prospective study in children: Pre- and post-surgery use of vitamin E in surgical incisions. J Plast Reconstr Aesthet Surg. 2010;63(9):1474-8. DOI: http://dx.doi.org/10.1016/j.bjps.2009.08.018
30. Zampieri N, Zuin V, Burro R, Ottolenghi A, Camoglio FS. A prospective study in children: Pre- and post-surgery use of vitamin E in surgical incisions. J Plast Reconstr Aesthet Surg. 2010;63(9):1474-8. DOI: http://dx.doi.org/10.1016/j.bjps.2009.08.018
31. Khoo TL, Halim AS, Zakaria Z, Mat Saad AZ, Wu LY, Lau HY. A prospective, randomised, double-blinded trial to study the efficacy of topical tocotrienol in the prevention of hypertrophic scars. J Plast Reconstr Aesthet Surg. 2011;64(6):e137-45. PMID: 20869928 DOI: http://dx.doi.org/10.1016/j.bjps.2010.08.029
32. Oxford Centre for Evidence-based Medicine - Levels of Evidence (March 2009) [Citado 28 Out 2015]. Disponível em: http://www.cebm.net/oxford-centre-evidence-based-medicine-levels-evidence-march-2009/
33. Foo CW, Tristani-Firouzi P. Topical modalities for treatment and prevention of postsurgical hypertrophic scars. Facial Plast Surg Clin North Am. 2011;19(3):551-7. DOI: http://dx.doi.org/10.1016/j.fsc.2011.06.008
34. Puzey G. The use of pressure garments on hypertrophic scars. J Tissue Viability. 2002;12(1):11-5. DOI: http://dx.doi.org/10.1016/S0965-206X(02)80004-3
35. Anzarut A, Olson J, Singh P, Rowe BH, Tredget EE. The effectiveness of pressure garment therapy for the prevention of abnormal scarring after burn injury: a meta-analysis. J Plast Reconstr Aesthet Surg. 2009;62(1):77-84. PMID: 18249046 DOI: http://dx.doi.org/10.1016/j.bjps.2007.10.052
36. Atiyeh BS. Nonsurgical management of hypertrophic scars: evidence-based therapies, standard practices, and emerging methods. Aesthetic Plast Surg. 2007;31(5):468-92. DOI: http://dx.doi.org/10.1007/s00266-006-0253-y
37. Shih R, Waltzman J, Evans GR; Plastic Surgery Educational Foundation Technology Assessment Committee. Review of over-the-counter topical scar treatment products. Plast Reconstr Surg. 2007;119(3):1091-5. PMID: 17312518 DOI: http://dx.doi.org/10.1097/01.prs.0000255814.75012.35
38. Mustoe TA, Cooter RD, Gold MH, Hobbs FD, Ramelet AA, Shakespeare PG, et al.; International Advisory Panel on Scar Management. International clinical recommendations on scar management. Plast Reconstr Surg. 2002;110(2):560-71. PMID: 12142678 DOI: http://dx.doi.org/10.1097/00006534-200208000-00031
39. Sawada Y, Sone K. Hydration and occlusion treatment for hypertrophic scars and keloids. Br J Plast Surg. 1992;45(8):599-603. PMID: 1493533 DOI: http://dx.doi.org/10.1016/0007-1226(92)90027-U
40. Tandara AA, Mustoe TA. The role of the epidermis in the control of scarring: evidence for mechanism of action for silicone gel. J Plast Reconstr Aesthet Surg. 2008;61(10):1219-25. PMID: 18653391 DOI: http://dx.doi.org/10.1016/j.bjps.2008.03.022
41. Mustoe TA, Gurjala A. The role of the epidermis and the mechanism of action of occlusive dressings in scarring. Wound Repair Regen. 2011;19 Suppl 1:s16-21. DOI: http://dx.doi.org/10.1111/j.1524-475X.2011.00709.x
42. Mustoe TA. Evolution of silicone therapy and mechanism of action in scar management. Aesthetic Plast Surg. 2008;32(1):82-92. PMID: 17968615 DOI: http://dx.doi.org/10.1007/s00266-007-9030-9
43. Perkins K, Davey RB, Wallis KA. Silicone gel: a new treatment for burn scars and contractures. Burns Incl Therm Inj. 1983;9(3):201-4.
44. Quinn KJ, Evans JH, Courtney JM, Gaylor JD, Reid WH. Non-pressure treatment of hypertrophic scars. Burns Incl Therm Inj. 1985;12(2):102-8. PMID: 4092154 DOI: http://dx.doi.org/10.1016/0305-4179(85)90035-X
45. Mercer NS. Silicone gel in the treatment of keloid scars. Br J Plast Surg. 1989;42(1):83-7. PMID: 2917222
46. Ohmori S. Effectiveness of silastic sheet coverage in the treatment of scar keloid (hypertrophic scar). Aesthetic Plast Surg. 1988;12(2):95-9. DOI: http://dx.doi.org/10.1007/BF01576921
47. de Oliveira GV, Nunes TA, Magna LA, Cintra ML, Kitten GT, Zarpellon S, et al. Silicone versus nonsilicone gel dressings: a controlled trial. Dermatol Surg. 2001;27(8):721-6. DOI: http://dx.doi.org/10.1097/00042728-200108000-00005
48. Li-Tsang CW, Lau JC, Choi J, Chan CC, Jianan L. A prospective randomized clinical trial to investigate the effect of silicone gel sheeting (Cica-Care) on post-traumatic hypertrophic scar among the Chinese population. Burns. 2006;32(6):678-83. DOI: http://dx.doi.org/10.1016/j.burns.2006.01.016
49. Lee SM, Ngim CK, Chan YY, Ho MJ. A comparison of Sil-K and Epiderm in scar management. Burns. 1996;22(6):483-7. PMID: 8884011 DOI: http://dx.doi.org/10.1016/0305-4179(95)00165-4
50. Maján JI. Evaluation of a self-adherent soft silicone dressing for the treatment of hypertrophic postoperative scars. J Wound Care. 2006;15(5):193-6. DOI: http://dx.doi.org/10.12968/jowc.2006.15.5.26913
51. Niessen FB, Spauwen PH, Robinson PH, Fidler V, Kon M. The use of silicone occlusive sheeting (Sil-K) and silicone occlusive gel (Epiderm) in the prevention of hypertrophic scar formation. Plast Reconstr Surg. 1998;102(6):1962-72. PMID: 9810992 DOI: http://dx.doi.org/10.1097/00006534-199811000-00023
52. Widgerow AD, Chait LA, Stals PJ, Stals R, Candy G. Multimodality scar management program. Aesthetic Plast Surg. 2009;33(4):533-43. PMID: 19048338 DOI: http://dx.doi.org/10.1007/s00266-008-9276-x
53. Tziotzios C, Profyris C, Sterling J. Cutaneous scarring: Pathophysiology, molecular mechanisms, and scar reduction therapeutics Part II. Strategies to reduce scar formation after dermatologic procedures. J Am Acad Dermatol. 2012;66(1):13-24. PMID: 22177632 DOI: http://dx.doi.org/10.1016/j.jaad.2011.08.035
54. Monstrey S, Middelkoop E, Vranckx JJ, Bassetto F, Ziegler UE, Meaume S, et al. Updated scar management practical guidelines: non-invasive and invasive measures. J Plast Reconstr Aesthet Surg. 2014;67(8):1017-25. DOI: http://dx.doi.org/10.1016/j.bjps.2014.04.011
55. Son D, Harijan A. Overview of surgical scar prevention and management. J Korean Med Sci. 2014;29(6):751-7. DOI: http://dx.doi.org/10.3346/jkms.2014.29.6.751
56. Kim S, Choi TH, Liu W, Ogawa R, Suh JS, Mustoe TA. Update on scar management: guidelines for treating Asian patients. Plast Reconstr Surg. 2013;132(6):1580-9. PMID: 24281584 DOI: http://dx.doi.org/10.1097/PRS.0b013e3182a8070c
57. Sawada Y, Sone K. Treatment of scars and keloids with a cream containing silicone oil. Br J Plast Surg. 1990;43(6):683-8.
58. Wong TW, Chiu HC, Chang CH, Lin LJ, Liu CC, Chen JS. Silicone cream occlusive dressing-a novel noninvasive regimen in the treatment of keloid. Dermatology. 1996;192(4):329-33. PMID: 8864367 DOI: http://dx.doi.org/10.1159/000246405
1. Universidade Federal Fluminense, Niterói, RJ, Brazil
2. Sociedade Brasileira de Cirurgia Plástica, Rio de Janeiro, RJ, Brazil
Institution: Hospital Universitário Antônio Pedro. Departamento de Cirurgia Geral de Especializada, Niterói, RJ, Brazil.
Corresponding author:
Pedro Leonardo Sanches Faveret
Rua Marquês de Paraná, 303
Niterói, RJ, Brazil Zip Code 24033-900
E-mail: pedrofaveret@id.uff.br
Article received: December 14, 2014.
Article accepted: May 26, 2015.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter