

Case Report - Year 2016 - Volume 31 -
Chest wall reconstruction using abdominal rectus and latissimus dorsi muscle flaps: case report
Reconstrução torácica com retalhos do músculo reto abdominal e grande dorsal: relato de casos
ABSTRACT
INTRODUCTION: Chest wall reconstruction remains one of the great plastic surgery repair challenges. The present work aims to report on cases of chest wall reconstruction using myocutaneous flaps after wall resection due to tumor/osteomyelitis.
METHODS: Four patients were included, among which three presented with osteomyelitis and the other presented with sarcoma; both of these conditions affected the chest wall. Each patient underwent wall resection and reconstruction using myocutaneous flaps from the latissimus dorsi (two cases) and abdominal rectus (two cases).
RESULTS: The flaps used were sufficient for skin-muscle covering and maintained good vitality. Partial flap injury occurred in one case. Chest cavity stability was preserved.
CONCLUSIONS: The use of the above flaps to repair chest defects was satisfactory with the aim of covering the skin and providing well-vascularized muscles, the latter of which was fundamental to preventing local infection.
Keywords: Reconstructive surgical procedures; Surgical flaps; Chest wall/surgery; Osteomyelitis; Sarcoma; Abdominal rectus; Superficial dorsal muscles.
RESUMO
INTRODUÇÃO: A reconstrução da parede torácica ainda permanece como um dos grandes desafios da cirurgia plástica reparadora. O presente estudo tem como objetivo apresentar o relato de pacientes submetidos à reconstrução de parede torácica com a utilização de retalhos miocutâneos, após ressecções de parede por tumores/osteomielite.
MÉTODOS: Foram incluídos quatro pacientes, três deles portadores de osteomielite e o último apresentando-se com sarcoma, todas as afecções acometendo a parede torácica. Foram submetidos à ressecção de parede e reconstrução com retalhos miocutâneos do grande dorsal (dois casos) e reto abdominal (dois casos).
RESULTADOS: Os retalhos utilizados foram suficientes para cobertura cutâneo-muscular e mantiveram boa vitalidade. Em apenas um caso, houve sofrimento parcial do retalho. Não houve recidiva precoce da doença. A estabilidade da caixa torácica foi preservada.
CONCLUSÕES: A utilização dos retalhos citados no reparo dos defeitos torácicos mostrou-se satisfatória na intenção de prover revestimento cutâneo e músculo bem vascularizado, este fundamental no combate aos quadros infecciosos locais.
Palavras-chave: Procedimentos cirúrgicos reconstrutivos; Retalhos cirúrgicos; Parede torácica/cirurgia; Osteomielite; Sarcoma; Reto do abdome; Músculos superficiais do dorso.
Chest wall reconstruction remains one of the greatest plastic surgery challenges despite notable advances in anesthesia and intensive therapy and the development of synthetic biomaterials that support the chest cavity. Chest wall conditions that eventually require resection/reconstruction - tumor, infections, radionecrosis1 and trauma - are better managed through multidisciplinary approaches, which require close interaction between chest and plastic surgeons2. Resection marking and distance range should be planned to enable adequate margins for treating the disease as well as the use of flaps, generally myocutaneous, to repair the defects.
OBJECTIVE
The present work aimed to report four cases of patients who underwent chest wall reconstruction using myocutaneous flaps from the latissimus dorsi muscle (LDM) or vertical rectus abdominis muscle (VRAM).
METHODS
Four patients who underwent surgery between 2011 and 2012 at the Plastic Surgery Service of the Hospital Universitário Walter Cantídio, Fortaleza, CE, were included in this study. All of the patients underwent resection of part of the chest wall and immediate reconstruction of defects using myocutaneous flaps. The resection step was performed by the hospital's chest surgery team.
Case 1
A 76-year-old woman presented with a long-standing history of radionecrosis with overlapping osteomyelitis in the sternal region after radiotherapy treatment for breast cancer in the left breast (previous mastectomy) (Figure 1A). Broad resection of all of the infected areas and a partial sternotomy were performed. There was no need to fix the mesh. The reconstruction was performed with a VRAM myocutaneous flap (Figure 1B).
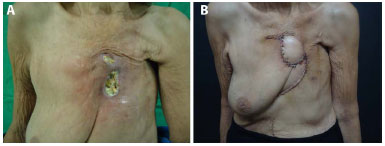
Figure 1. A: Preoperative image of sternal radionecrosis and osteomyelitis. B: Image taken 15 days after reconstruction with a vertical rectus abdominis muscle (VRAM) flap.
Case 2
A 72-year-old man with chronic renal disease and a history of osteomyelitis with projection in the eighth, ninth, and tenth costal arches that affected the total thickness of the chest wall (Figure 2A). The patient was subjected to broad resection of the infected area with resection of the two costal arches and reconstruction of the chest wall with polypropylene mesh (double) and a VRAM myocutaneous flap (Figure 2B).
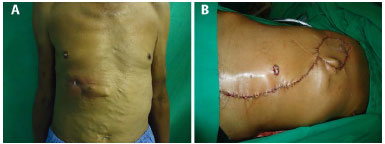
Figure 2. A: Preoperative image of osteomyelitis in the right hemithorax (RHT ). B: Image taken immediately after reconstruction with a vertical rectus abdominis muscle (VRAM) flap.
Case 3
A 58-year-old man presented with large masses in the left chest region (nearly 16 cm at its largest diameter) that showed a rapid evolution, and a biopsy revealed fibrosarcoma (Figures 3A and 3B). The broad resection of the tumor included three costal arches and the adjacent pleura. The chest wall reconstruction was performed with an LDM myocutaneous flap (Figure 3C) and double polypropylene mesh (Figure 3D).
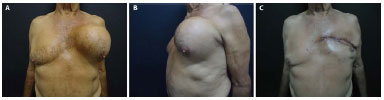
Figure 3. A: Preoperative image of sarcoma in the left chest region. B: Preoperative image of sarcoma in the left chest region. C: Image taken 15 days after reconstruction with a latissimus dorsi muscle (LDM) flap and mesh.
Case 4
A 63-year-old woman presented with a history of radionecrosis and overlapping osteomyelitis in the anterior chest wall and projection into the seventh and eighth costal arches. The patient underwent right mastectomy and adjuvant radiotherapy 5 years prior (Figure 4A). The patient underwent partial resection of the chest wall and removal of the two costal arches; however, there was no need to fix the mesh. The reconstruction was performed exclusively with an LDM flap (Figure 4B).
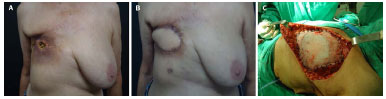
Figure 4. A: Preoperative image of radionecrosis and osteomyelitis in the right hemithorax (RHT). B: Image taken 30 days after reconstruction with a latissimus dorsi muscle (LDM) flap. C: Image taken in the intraoperative period during which the non-absorbable double-layer mesh was fixed.
RESULTS
The flaps used sufficiently covered the skin and muscle and maintained good vitality, probably due to the good blood irrigation provided by reliable pedicles, associated with the small dimension of the flaps used compared to the use of these flaps in reconstructions that require the use of a greater amount of tissue (for example, mammary reconstruction). In just one case (Case 1), a small skin portion of the VRAM flap was injured, and the rotation of a local fasciocutaneous flap was required to repair the defect. The remaining flaps showed no signs of injury or dehiscence.
DISCUSSION
Overall, there are four clinical situations that may require chest wall resection and consequent reconstruction: tumors (primary or recurring)3, infections, complications due to radiotherapy, and, less commonly, trauma2. These etiologies often overlap. A variety of myocutaneous flaps are used to correct the defects, including the latissimus dorsi, transverse rectus abdominis muscle (TRAM) or VRAM, pectoralis major, and trapezius. Other muscles are used less frequently. Omental transposition is a second interesting option when the previous methods fail or, in cases of recurrent infection, due to its rich vascularization.
The latissimus dorsi muscle flap, pedicled in the thoracodorsal artery, is a good option for covering the anterior and anterolateral defects of the hemithorax, although it may eventually be rotated until it reaches the sternal region. It has the advantage of offering a good rotation arch and can be designed in different forms and sizes according to defect type4.
The abdominal rectus muscle flap, pedicled in the superior epigastric artery, is used to cover anterior or central defects of the hemithorax. This flap may be designed in a transverse (TRAM) or vertical (VRAM) form according to the orientation of the area requiring repair. It does not present a pedicle as reliable as the LDM or pectoralis major, and the amount of muscle in the flap is lower than in the other above-mentioned flaps, but it remains a good option, mainly when there are concerns regarding pedicle viability in the other flaps5.
The major pectoral muscle flap, which is based on the thoracoacromial artery, is an excellent option for repairing the central and superior portions of the chest. The presence of a robust pedicle and muscle is important to the choice of this flap.
The omentum majus flap is an interesting option in complex situations wherein there are important local infections and/or a loss of myocutaneous flaps in the case of infection since this is a highly vascularized flap that well supports bone or skin grafts2.
Flaps from the serratus anterior external oblique muscles may be less commonly used in restricted situations only.
Structural wall reconstruction is generally performed with polypropylene mesh (double) added or not to methacrylate (the sandwich technique). This material provides adequate support and maintenance of the respiratory dynamic when costal or sternal resections are involved.
CONCLUSION
The use of the above-mentioned flaps in the repair of chest defects satisfactorily covered skin and vascularized muscles, and the latter is fundamental to the prevention of local infections. The use of a double-mesh polypropylene, when required, provided adequate stability and good respiratory function.
REFERENCES
1. Rouanet P, Fabre JM, Tica V, Anaf V, Jozwick M, Pujol H. Chest wall reconstruction for radionecrosis after breast carcinoma therapy. Ann Plast Surg. 1995;34(5):465-70. PMID: 7639482 DOI: http://dx.doi.org/10.1097/00000637-199505000-00003
2. Arnold PG, Pairolero PC. Chest-wall reconstruction: an account of 500 consecutive patients. Plast Reconstr Surg. 1996;98(5):804-10. DOI: http://dx.doi.org/10.1097/00006534-199610000-00008
3. Crosby MA, Chike-Obi CJ, Baumann DP, Sacks JM, Villa MT, Garvey PB, et al. Reconstructive outcomes in patients with sarcoma of the breast. Plast Reconstr Surg. 2010;126(6):1805-14. DOI: http://dx.doi.org/10.1097/PRS.0b013e3181f5276f
4. Maia M, Oni G, Wong C, Saint-Cyr M. Anterior chest wall reconstruction with a low skin paddle pedicled latissimus dorsi flap: a novel flap design. Plast Reconstr Surg. 2011;127(3):1206-11. PMID: 21088645 DOI: http://dx.doi.org/10.1097/PRS.0b013e318205f2f7
5. Chang RR, Mehrara BJ, Hu QY, Disa JJ, Cordeiro PG. Reconstruction of complex oncologic chest wall defects: a 10-year experience. Ann Plast Surg. 2004;52(5):471-9. DOI: http://dx.doi.org/10.1097/01.sap.0000122653.09641.f8
1. Universidade Federal do Ceará, Fortaleza, CE, Brazil
2. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
3. Hospital Universitário Walter Cantídio, Fortaleza, CE, Brazil
Institution: Universidade Federal do Ceará (UFC) Hospital Universitário Walter Cantídio (HUWC).
Corresponding author:
Vitor de Vasconcellos Muniz
Rua Monsenhor Bruno, 1101, Apto. 403 - Aldeota
Fortaleza, CE, Brazil Zip Code 60115-190
E-mail: vitordvm@yahoo.com.br
Article received: August 19, 2012.
Article accepted: October 15, 2012.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter