

Case Report - Year 2016 - Volume 31 -
Micrographic surgery in the treatment of microcystic adnexal carcinoma
Cirurgia micrográfica no tratamento do carcinoma microcístico anexial
ABSTRACT
Microcystic adnexal carcinoma is an uncommon, locally aggressive eccrine sweat gland tumor that is easily confused with benign adnexal tumors and has a high rate of recurrence after incorrect treatment. The present study was aimed at increasing awareness on the importance of knowing and properly managing this disease among plastic surgeons by using Mohs micrographic surgery. A literature review from 1982 to January 2014 and a prospective case study of a microcystic adnexal carcinoma treated with Mohs micrographic surgery, at the plastic surgery clinic of Felício Rocho Hospital in March 2011, were performed. First described in 1982, microcystic adnexal carcinoma now has at least 700 cases reported in the international literature. The case reported herein was treated with Mohs micrographic surgery, including a 3-year follow-up without recurrence. In conclusion, Mohs micrographic surgery is an excellent therapeutic tool to preserve vital tissues and ensure a reduced recurrence rate.
Keywords: Mohs surgery; Cutaneous tumor; Surgical flaps.
RESUMO
Carcinoma microcístico anexial é uma neoplasia de glândulas sudoríparas écrinas incomum, localmente agressivo. Facilmente confundido com tumores anexiais benignos e com alto índice de recidiva após tratamento incorreto. O objetivo é divulgar aos cirurgiões plásticos a importância do conhecimento e manejo correto desta afecção com utilização da cirurgia micrográfica de Mohs. Foi realizada revisão da literatura de 1982 a janeiro de 2014 e análise prospectiva de um caso de carcinoma microcístico anexial conduzido na Clínica de Cirurgia Plástica do Hospital Felício Rocho em março de 2011, utilizando a cirurgia micrográfica de Mohs. Descrito em 1982, menos de 700 casos de carcinoma microcístico anexial relatados na literatura mundial. O caso apresentado foi tratado com o uso da cirurgia micrográfica de Mohs, com seguimento 3 anos sem recidiva. Podemos concluir que a cirurgia micrográfica de Mohs é um excelente arsenal terapêutico, com menor índice de recidiva e preservação de tecidos vitais.
Palavras-chave: Cirurgia de Mohs; Neoplasias cutâneas; Retalhos cirúrgicos.
Microcystic adnexal carcinoma (MAC) is a rare, low-grade, locally aggressive eccrine sweat gland tumor that rarely metastasizes1,2. MAC most often affects the head and neck3, with differential diagnosis for benign adnexal tumors such as trichoepithelioma, syringoma, and malignant skin tumors, including basal and squamous cell carcinomas, among others4,5. Given its subclinical progression, MAC is diagnosed at later stages, and incorrect treatment causes a high recurrence rate6.
OBJECTIVES
The present study was aimed at reporting a case of MAC treated with Mohs micrographic surgery at the plastic surgery clinic of Felício Rocho Hospital (Belo Horizonte, Minas Gerais [MG], Brazil) and performing a nonsystematic literature review toward increasing awareness on the importance of knowing and properly managing this rare disease among plastic surgeons.
METHODS
The study was conducted prospectively in accordance with the ethical principles of the Declaration of Helsinki revised in 2000. A patient diagnosed as having MAC was examined in March 2011, underwent Mohs micrographic surgery at the plastic surgery clinic of Felício Rocho Hospital (Belo Horizonte, MG, Brazil), and followed up for 3 years.
The Mohs technique used begins with enucleation, which is the excision of the visible tumor, and histopathological analysis. The post-Mohs micrographic surgical margin is then excised. The fragments are then mapped and subsequently frozen in a cryostat, with deep fragment embedding. The location of the residual tumor, when present, is accurately identified, and a new stage of local excision is performed until complete tumor excision7,8.
Defect reconstruction was performed during the same surgery. All surgical specimens were examined at the department of pathological anatomy of the same institution by using the standardized criteria to confirm the diagnosis.
The review included studies from 1982, when the first report of a MAC case was published, to January 2014, when the most recent study was published. The databases searched were Medline and PubMed. The search was limited to manuscripts written in English or Portuguese languages, and "microcystic adnexal carcinoma" was used as the search keyword. Articles published in oncology, dermatology, head and neck surgery, and other journals were selected because of the lack of studies published in plastic surgery journals.
Studies published in the references of the articles initially identified were selected, and articles with repeated data or available in other studies were excluded from the present study. The authors evaluated all the articles. The main inclusion criteria of the study were clinical presentation, histological features, immunohistochemistry, differential diagnosis, and treatment options.
RESULTS
A female patient aged 66 years underwent incisional biopsy of a light-brown plaque-like lesion with imprecise limits in the right inferomedial subpalpebral area (Figure 1) at another hospital department, in February 2011. Histopathological findings were indicative of squamous cell carcinoma. The pathologist suggested that diagnosis could be made with a larger lesion sample.
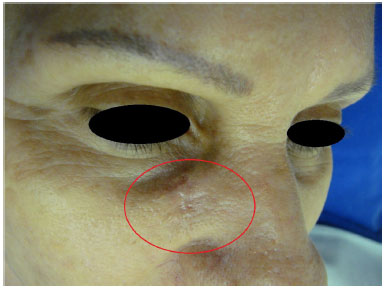
Figure 1. Plaque-like cutaneous lesion with imprecise limits in the right inferomedial subpalpebral area.
Subsequently, the patient was managed by the plastic surgery team of Felício Rocho Hospital, wherein a new incisional biopsy was performed in March 2011, with findings suggestive of infiltrating carcinoma with atypical tumor cell nests infiltrating the dermis and perineural invasion foci, which most likely originated from the eccrine glandular structures.
Immunohistochemical analysis was performed to aid diagnosis. Tumor cells were negative for epithelial antigen (Ber-EP4) and positive for p63, indicating squamous or pillar differentiation. p63 reactivity suggests a primary cutaneous lesion. Tumor cells were also positive for epithelial membrane antigens (EMAs) and cytokeratin (AE1 and AE3 clones). The combined findings suggest the diagnosis of MAC.
In April 2011, micrographic surgery of the facial lesion and immediate reconstruction with a local flap for defect repair were performed at only one stage, under local anesthesia and sedation (Figure 2).
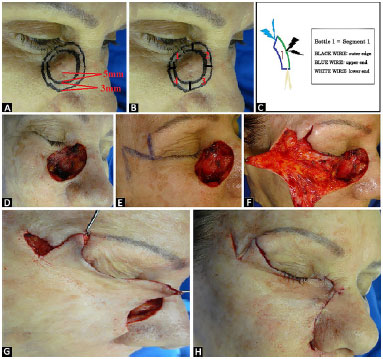
Figure 2. Surgical technique. A and B: Cutaneous lesion, 5-mm lateral excision margin, and staining of four 3-mm-wide fragments. Surgical margins are examined in frozen sections; C: Mapping scheme and identification of each fragment; D: Loss of substance; E, F, G, H: Defect repaired by using the McGregor technique (lateral periorbital zetaplasty).
The patient responded well, without signs of tumor recurrence during a 3-year follow-up (Figure 3).
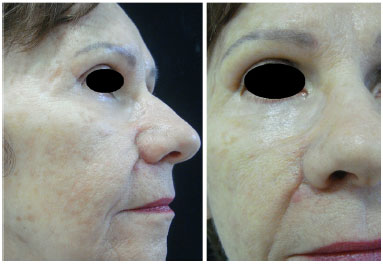
Figure 3. Appearance at 3-year postoperative follow-up.
DISCUSSION
First described in 1982 by Goldstein et al.1, MAC, also known as sclerosing sweat duct carcinoma, malignant syringoma, and syringoid carcinoma, is an uncommon cutaneous tumor, with approximately 700 cases reported in the international literature.
A retrospective case series over a 16-year period was conducted at a Mohs surgical center based in Pennsylvania (USA)3. The number of cases per year was 1.63.
Another large, American retrospective study with a statistical analysis of 223 patients identified the prevalence in females (57%) and Caucasians (90%). Cases have been reported in African Americans, Japanese, Puerto Ricans, Spanish, Koreans, and Jews9.
MAC rarely occurs in children, predominantly affecting the elderly, with a mean age of 68 years and higher incidence in the head and neck, including the lips (74%). Similar data were reported in another large case series conducted at the University of California, San Francisco, which also reported predominance of the left side among patients with MAC10.
MAC consists of low-grade lesions, albeit locally aggressive, which are clinically characterized as a hardened, solitary plaque, nodule, or cyst, with imprecise limits, skin color, or yellowish plaque, most commonly located in the central area of the face. Other uncommon locations include the scalp, armpits, buttocks, and genitals10,11.
MAC is usually asymptomatic. Increased morbidity occurs in cases with perineural invasion. Patients may experience symptoms, including paresthesia, anesthesia, and local pain12.
Ultraviolet (UV) radiation may be involved in the tumor pathogenesis, and cases reported in the literature were associated with immunosuppression5.
Given its subclinical presentation, MAC is commonly diagnosed at late stages and mostly confined to the skin (75%), with lesions averaging 2 cm in size. However, soft tissue and even bone invasion may occur because of local aggressiveness, causing high morbidity. Lymph nodes are affected in 1% of cases, and distant metastasis is extremely rare13.
Clinical differential diagnosis includes benign adnexal tumors such as trichoepitheliomas, trichoadenoma, syringoma, and malignant skin tumors, including squamous cell carcinoma, basal cell carcinoma, and even melanoma14.
Histopathological analysis with appropriate tissue sampling is required to avoid misdiagnosis because the histological features of MAC include few atypical cells, of which few or none are mitotic. In a case series, misdiagnosis occurred in 27% of cases because bland microscopic features associated with inadequate samples complicated the diagnosis10. Therefore, the biopsy tissue must have adequate thickness and size.
The disease is characterized by multiple islands of epithelial basaloid cells, ductal structures, and keratinized cysts, intradermally located, stretching as thin threads of tumor cells interspersed between collagen bundles. It typically features deep components, small tumor nests, and cell strands embedded in a hyalinized stroma. Perineural and intramuscular invasions commonly occur, and such features are not evident in superficial biopsies14.
Perineural invasion has been reported in 17.5% to 59% of cases in a surgical series and is commonly associated with tumor recurrence15.
Immunohistochemistry may be useful in diagnosis and differentiation from other tumors. Eccrine and pillar differentiations usually stand out. The most reliable positive markers are the EMAs, which stain ductal structures; AE1 and AE3, which strongly stain epithelial cells in MAC; and carcinoembryonic antigen (CEA), which is useful to differentiate MAC from desmoplastic trichoepitheliomas3.
Several retrospective studies and a prospective study confirmed the efficacy of Mohs micrographic surgery for the treatment of MAC. Conventional excisions have recurrence rates higher than 47%, usually in the first 3 years after operation. Conversely, Mohs surgery has been recognized as the treatment of choice for cutaneous tumors whose surgical margins are difficult to define, primarily because it increases the chance of cure and spares healthy tissues, showing a 0-22% recurrence rate in a 5-year follow-up period. Nevertheless, approximately 87% and 10% of primary MACs are currently treated with conventional and Mohs surgical techniques, respectively6.
Mohs surgical technique is named after its pioneer, the physician Dr. Frederic Edward Mohs, from the University of Wisconsin, USA. It was developed in the 1930s. The technique involves excising skin and mucosal tumors with maximum efficiency and reduced tissue loss, and is characterized by surgical mapping combined with judicious perioperative microscopic examination of all surgical margins16.
Complementary treatment with radiotherapy and chemotherapy is not yet consensual, given the rarity and few cases reported. Authors believe that adjuvant radiotherapy may contribute to local control in patients for whom additional surgery is contraindicated, although tumor conversion into more aggressive forms has been reported as an adverse event17. Even if not the first-line treatment option, chemotherapy should be useful in patients at advanced disease stages, who lack treatment options18.
A long follow-up is important for patients with MAC considering the fact that reports of recurrence after 30 years of treatment have been published.
CONCLUSION
Knowledge of the lesion and adequate physical examination favor clinical suspicion, which provides accurate diagnosis when aided by histopathological and immunohistochemical analyses. When properly executed, Mohs micrographic surgery is an excellent therapeutic tool, with reduced recurrence rate, and should be considered as a differential diagnostic technique for cutaneous tumors, especially of the face.
COLLABORATIONS
ACMA Data analysis and/or interpretation, statistical analysis, and manuscript writing; and study design.
AFSF Content review analysis, performing the surgeries and/or experiments, and final manuscript approval.
JCRRA Content review analysis, performing the surgeries and/or experiments, and final manuscript approval.
RLFS Literature search.
REFERENCES
1. Goldstein DJ, Barr RJ, Santa Cruz DJ. Microcystic adnexal carcinoma: a distinct clinicopathologic entity. Cancer. 1982;50(3):566-72.
2. Yugueros P, Kane WJ, Goellner JR. Sweat gland carcinoma: a clinicopathologic analysis of an expanded series in a single institution. Plast Reconstr Surg. 1998;102(3):705-10.
3. Thomas CJ, Wood GC, Marks VJ. Mohs micrographic surgery in the treatment of rare aggressive cutaneous tumors: the Geisinger experience. Dermatol Surg. 2007;33(3):333-9.
4. Gabillot-Carré M, Weill F, Mamelle G, Kolb F, Boitier F, Petrow P, et al. Microcystic adnexal carcinoma: report of seven cases including one with lung metastasis. Dermatology. 2006;212(3):221-8.
5. Abbate M, Zeitouni NC, Seyler M, Hicks W, Loree T, Cheney RT. Clinical course, risk factors, and treatment of microcystic adnexal carcinoma: a short series report. Dermatol Surg. 2003;29(10):1035-8.
6. Tse JY, Nguyen AT, Le LP, Hoang MP. Microcystic adnexal carcinoma versus desmoplastic trichoepithelioma: a comparative study. Am J Dermatopathol. 2013;35(1):50-5.
7. Mohs FE. Chemosurgery: a microscopically controlled method of cancer excision. Arch Surg. 1941;42(2):279-95.
8. Leibovitch I, Huilgol SC, Selva D, Lun K, Richards S, Paver R. Microcystic adnexal carcinoma: treatment with Mohs micrographic surgery. J Am Acad Dermatol. 2005;52(2):295-300.
9. Nadiminti H, Nadiminti U, Washington C. Microcystic adnexal carcinoma in African-Americans. Dermatol Surg. 2007;33(11):1384-7.
10. Yu JB, Blitzblau RC, Patel SC, Decker RH, Wilson LD. Surveillance, Epidemiology, and End Results (SEER) database analysis of microcystic adnexal carcinoma (sclerosing sweat duct carcinoma) of the skin. Am J Clin Oncol. 2010;33(2):125-7.
11. Carroll P, Goldstein GD, Brown CW Jr. Metastatic microcystic adnexal carcinoma in an immunocompromised patient. Dermatol Surg. 2000;26(6):531-4.
12. Thosani MK, Marghoob A, Chen CS. Current progress of immunostains in Mohs micrographic surgery: a review. Dermatol Surg. 2008;34(12):1621-36.
13. Todd MM, Lee JW, Marks VJ. Rapid toluidine blue stain for Mohs' micrographic surgery. Dermatol Surg. 2005;31(2):244-5.
14. Wang SQ, Goldberg LH, Nemeth A. The merits of adding toluidine blue-stained slides in Mohs surgery in the treatment of a microcystic adnexal carcinoma. J Am Acad Dermatol. 2007;56(6):1067-9.
15. Diamantis SA, Marks VJ. Mohs micrographic surgery in the treatment of microcystic adnexal carcinoma. Dermatol Clin. 2011;29(2):185-90.
16. Albom MJ, Swanson NA. Mohs Micrographic Surgery for the Treatment of Cutaneous Neoplasms. In: Friedman RJ, Rigel DS, Kopf AW, Harris MN, Baker D, eds. Cancer of the skin. Philadelphia: WB Saunders; 1991. p.484-529.
17. Pugh TJ, Lee NY, Pacheco T, Raben D. Microcystic adnexal carcinoma of the face treated with radiation therapy: a case report and review of the literature. Head Neck. 2012;34(7):1045-50.
18. Tawfik AM, Kreft A, Wagner W, Vogl TJ. MRI of a microcystic adnexal carcinoma of the skin mimicking a fibrous tumour: case report and literature review. Br J Radiol. 2011;84(1002):e114-7.
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
2. Hospital Monte Sinai, Juiz de Fora, MG, Brazil
3. Hospital e Maternidade Therezinha de Jesus, Juiz de Fora, MG, Brazil
4. Hospital Felício Rocho, Belo Horizonte, MG, Brazil
5. Instituto de Cirurgia Plástica Avançada, Belo Horizonte, MG, Brazil
6. Universidade Federal de Minas Gerais, Belo Horizonte, MG, Brazil
7. Faculdade de Medicina da Universidade de Itaúna, Itaúna, MG, Brazil
Institution: Clínica de Cirurgia Plástica do Hospital Felício Rocho, Belo Horizonte, MG, Brazil.
Corresponding author:
Augusto César de Melo Almeida
Rua Ouro Preto, 617/1904 - Barro Preto
Belo Horizonte, MG, Brazil Zip Code 30170-040
E-mail: almeidaugusto@hotmail.com
Article received: April 14, 2015.
Article accepted: July 20, 2015.
Conflicts of interest: none.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter