

Original Article - Year 2016 - Volume 31 -
Effects of radiofrequency on the healing of skin wounds in rats: analysis using digital planigraphy and histological evaluation
Efeitos da radiofrequência na cicatrização de feridas cutâneas em ratos: análise por planigrafia digital e avaliação histológica
ABSTRACT
INTRODUCTION: Given the extensive use of plastic surgery and the search for better aesthetic and functional results, it is necessary to research ways to improve healing and scarring. The objective is to evaluate the effects of three radiofrequency (RF) sessions in healing the skin of mice.
METHODS: Forty-eight rats were divided into four groups by day of sacrifice and treatment (RF group: RG; control group: CG). Dissection of the excisional wound of 2 cm x 2 cm (4 cm²) was performed and a 6-mm punch was used to hold two excisional wounds 0.6 cm in diameter. After 24 h, radiofrequency was performed using Spectra® directly on the wound in the dorsal region for 7 minutes at 38ºC. This was repeated three times on alternate days. For the control group, the radiofrequency protocol was performed with the device switched off.
RESULTS: A larger area of the square wound was measured on postoperative day three in RG (RG7: 3.3 cm² ± 0.7 cm² vs. CG7: 2.4 cm² ± 0.4 cm²; p = 0.009). On day 14, the square wound in RG was greater than in CG (RG14: 1.9 cm² ± 0.5 cm² vs. CG14: 1.0 cm² ± 0.3 cm²; p = 0.001). There was a 90% closure of wounds in CG14. In RG14, 60% of the wounds were re-epithelized while 40% remained ulcerated. In CG7, 70% of the remaining wounds were ulcerated and 30% were re-epithelized. In RG7, 8% were re-epithelized and 92% remained ulcerated.
CONCLUSION: Radiofrequency has a negative influence on the healing process, as indicated by mice that received radiofrequency having a persistent ulcerated wound.
Keywords: Healing; Scar; Fibroblasts; Re-epithelialization.
RESUMO
INTRODUÇÃO: Tendo em vista o número de cirurgias plásticas e a busca por melhores resultados estético-funcionais fazem-se necessárias pesquisas para encontrar meios para melhorar a cicatrização e as cicatrizes. O objetivo é avaliar os efeitos de três sessões de radiofrequência na cicatrização da pele de ratos.
MÉTODOS: Quarenta e oito ratos machos foram divididos em quatro grupos conforme ao grupo que pertenciam e o dia do sacrifício (grupo radiofrequência - GR - e grupo controle - GC). Realizada a dissecação da ferida excisional de 2 cm x 2 cm (4 cm²) e utilizou-se um punch de 6 mm para a realização de duas feridas excisionais de 0,6 cm de diâmetro. Após 24 h, foi realizada a radiofrequência com o equipamento Spectra® na região dorsal, diretamente sobre as feridas por 7 minutos, com temperatura de 38ºC. Repetida por três vezes, em dias alternados. No grupo controle foi realizada com o aparelho desligado.
RESULTADOS: Foi encontrada área maior na ferida quadrada, no 3º dia pós-operatório do GR (GR7 3,3 cm² ± 0,7 cm² vs. GC7 2,4 cm² ± 0,4 cm², p = 0,009). No 14º dia a ferida quadrada do GR foi maior do que no GC (GR14 1,9 cm² ± 0,5 cm² vs. GC14 1,0 cm² ± 0,3 cm², p = 0,001). Houve fechamento de 90% das feridas no GC14. No GR14, 60% das feridas foram reepitelizadas enquanto 40% permaneceram ulceradas. No GC7, 70% das feridas de permaneceram ulceradas e 30% foram reepitelizadas. Já no GR7, 8%, foram reepitelizadas e 92% permaneceram ulceradas.
CONCLUSÃO: A radiofrequência tem influência negativa no processo cicatricial, mostrando que, nos ratos que receberam a radiofrequência, o quadrado permaneceu ulcerado.
Palavras-chave: Cicatrização; Cicatriz; Fibroblastos; Reepitelização.
Healing after cosmetic surgery is of great interest given the number of plastic surgeries in Brazil and the search for better aesthetic and functional results. It is thus necessary to research and improve techniques and identify solutions to perfect skin repair.
Collagen fibers provide tensile strength in the healing stage of tissue repair. Their metabolism is a balance between biosynthesis and degradation; they are resorbed during growth, remodeling, involution, inflammation and tissue repair. Resorption is initiated by specific collagenases that digest fiber and tropocollagen molecules1.
Collagen, as its primary function, supports the extracellular matrix by providing strength and structural integrity of various tissues and organs, including the dermis. In addition, when immature, collagen is characterized by large number of young fibroblasts and less dense collagen fibers due to the large amount of water within its structure. Over time, the collagen matures and thus becomes less cellular and with less water content. The connective tissue will gradually mature with a higher deposition of more mature collagen1.
The ability to contract collagen with thermal energy is not new in medicine. Thermal-induced collagen contraction has been previously documented in sports medicine, where it facilitates the stretching of the ligaments responsible for shoulder instability. This concept of contraction, with minimal or no epidermal damage, is extremely interesting2.
Physical therapy has therapeutic resources that can influence this process, among which radiofrequency (RF) is a primary tool given its ability to heat deep tissue while protecting the skin3.
However, the effect of RF healing remains unknown. Thus, the aim of the study in experimental animals was to evaluate its use in the healing of skin wounds in rats by computerized digital planigraphy with macroscopic and histological analysis.
OBJECTIVE
The objective of this study was to evaluate the RF effects on skin healing in rats.
METHODS
This controlled experimental and quantitative research was approved by the Research Ethics Committee of the Pontifical Catholic University of Paraná (PUCPR) under the registration number 684 on February 28, 2013.
The experiments were performed in the laboratory of PUC-PR, Curitiba-PR. The histopathologic study was developed at St. Luke's Hospital laboratory in the same city.
This study included 48 adult male rats of the species Rattus norvegicus (i.e., albino Wistar rats), weighing between 300 g and 350 g, aged ± 3 months, and kept in individual cages with temperature controlled by stable air conditioning at 21 ± 2ºC and an artificial light/dark cycle of 12 h.
The animals were randomly divided into four groups of 12 animals, identified by a number and the group. The groups included control group (CG) with radiofrequency device switched off, and radiofrequency group (RG), with the radiofrequency device switched on. These groups were further, classified according to the day euthanasia was performed (i..e, 7th or 14th postoperative day (PO):
• CG7: Control Group 7.First, rats were anesthetized with ketamine 80 mg/kg and xylazine 8 mg/kg administered intramuscularly in a thigh.
• RG7: Radiofrequency Group 7.
• CG14: Control Group 14.
• RG14: Radiofrequency Group 14.
After anesthesia, trichotomy and antisepsis with povidone-iodine (PVP-I) was performed. Next, the animals were placed in the prone position with the four limbs extended and the head aligned with the trunk. Marking was made using black ink, using a plastic mold 2 cm x 2 cm (4 cm²) that was made by the author.
Incisions were made using a 20-blade scalpel along the edges of the square used to demarcate the dissection of the skin segment. In addition, there were two circular excisional wounds, which were made with a 6 mm metallic punch using a cutting blade at its lower edge. This was followed by the removal of two skin fragments of 0.6 cm² in diameter to expose the dorsal fascia.
During the next five days, rats were administered 5 mg/kg of the analgesic dipyrone intramuscularly twice daily to minimize pain or discomfort the animal might be feeling.
At 24h after the injury, anesthesia was repeated. The 10 x 5 cm² dispersion electrode was placed in the abdominal region for radiofrequency application in the dorsal region. The active electrode was applied for 7 minutes in total: 5 minutes on the dorsal region and 2 minutes for the electrode to reach the ideal temperature of 38ºC.
This procedure was repeated three times on alternate days with the rat under anesthesia. A radiofrequency modulator with an amplitude of 100% and a neutral gel was used. The handle was positioned on the rat dorsum skin at 90º, and rectilinear and circular movements were performed. For this, the temperature was checked with an infrared thermometer, which allows for accurate, real-time measurement. The thermometer was maintained at a distance of 10 cm and placed at 90º to the skin. In all groups, the same procedure was performed; however, in the control group, the equipment was turned off.
Photographic documentation was performed on the day of surgery as well as at postoperative days 1, 7, and 14.
The scar contraction of the back of the rats wound was compared using computer analysis of digital photos using standardized ImageJ® software (National Institutes of Health, USA).
The samples were taken on the 7th and 14th day and stored in 10% formaldehyde solution and were sent to the PUC laboratory for slide preparation and histological analysis.
After this procedure, the animals were euthanized in a chamber with CO2 saturation at 70%.
The fixed samples were cleaved, place in histological cassettes and subjected to technical processing for paraffin embedding. The samples were embedded, cut in a microtome to 3 µm, stained with hematoxylin-eosin method (HE), and mounted.
Microscopic analysis under an optical microscope Olympus CX31 model® was performed by a pathologist who completed the report without knowing to which group the mouse belonged. This allowed us to semi-quantitatively evaluate cellular inflammatory components, the cellular elements of chronic and acute inflammation, repair and fibroblast proliferation.
Data are expressed as the mean and standard deviation. Student's t-test and a one-way ANOVA were used for group comparisons p values < 0.05 were considered statistically significant.
RESULTS
The development of the reduction of the lesion area in each experiment, as analyzed by digital planigraphy, is shown in the figures below (Figures 1 and 2).
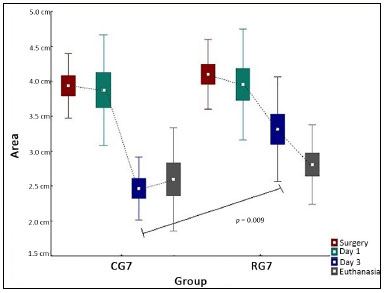
CG7: Control Group 7; RG7: Radiofrequency Group 7.
Figure 1. Comparative assessment of RG7 and CG7 at surgery, POD1, POD3, and euthanasia. POD: post-operative day.
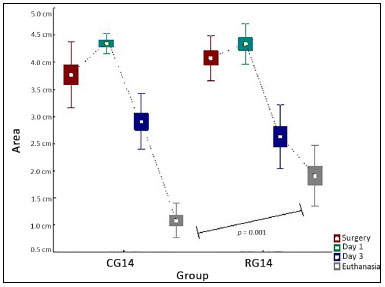
CG14: Control Group 14; RG14: Radiofrequency Group 14.
Figure 2. Comparative assessment of RG14 and CG14 at surgery, POD1, POD3, and euthanasia. POD: post-operative day.
The average wound area on day 3 in RG7 was larger than the area in CG7 (RG7: 3.3 ± 0.7 cm² vs. CG7: 2.4 ± 0.4 cm²; p = 0.009).
There was a significant difference in wound size between RG14 and CG14 groups on the day of euthanasia (RG14: 1.9 ± 0.5 cm² vs. CG14: 1.0 ± 0.3 cm²; p = 0.001). We observed that both groups experienced a decline in the area on day three; however, in RG14, there was a smaller reduction in the wound area on the day of euthanasia than there was in CG14.
On postoperative days seven and 14, the wounds of the control and experimental groups were different (Table 1).
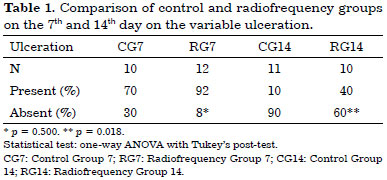
There was 90% closure of wounds in CG14, while 10% remained ulcerated. In RG14, 60% of mice with punch wounds were re-epithelized, while 40% remained ulcerated, thereby showing a significant difference between RG14 and CG14 with p = 0.018. In CG7, 70% of the punch wounds remained ulcerated and 30% were epithelized. In RG7, 8% of mice with punch wounds were re-epithelized and 92% of the rats were ulcerated.
Figures 3 and 4 include an example of the details of the punch and square wounds.
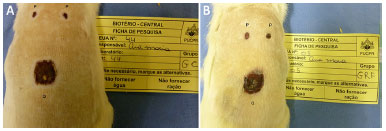
Figure 3. A and B: Example of punch and square wounds represented by P (punch) and Q (square). CG7 (A) and RG7 (B) do not present macroscopic differences.
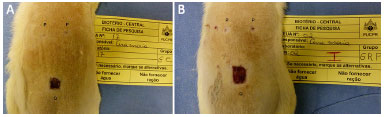
Figure 4. A and B: Example of punch and square wounds represented by P (punch) and Q (square). In A, CG14 punches are shown closed, while in B, punches are more open in RG14. It is also apparent that, in RG14, the square is larger compared to the square of CG14.
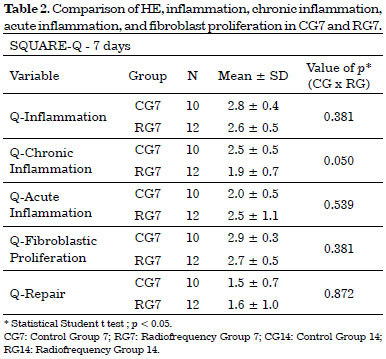
Only in RG7 did the evaluation of chronic inflammation cells indicated a difference between groups (GR7: 1.9 ± 0.7 vs. CG7: 2.5 ± 0.5; p = 0.05) (Table 2).
In RG14, an increase in acute inflammation cells and inflammatory markers was observed (Table 3).
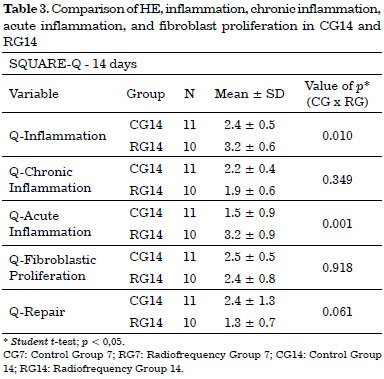
The average of inflammation was similar in both RG7 and CG7 (p = 0.3), while there was a greater average in RG14 relative to CG14 (p = 0.01).
In the analysis of acute inflammation, RG7 had a similar number of cells relative to CG7 (p = 0.53) and an increased number of neutrophils in RG14 vs. CG14 (p = 0.001).
Chronic inflammation has persisted in RG7 and RG14 (p = 0.82), while in CG7 and CG14 these cellular elements of chronic inflammation exhibited a moderate number of lymphocytes and/or plasma cells (p = 0.22).
Regarding fibroblast proliferation, both RG7 and CG7 groups (p = 0.38) as well as RG14 and CG14 (p = 0.91) were similar.
During repair, macrophages were present in small numbers in both RG7 and CG7 (p = 0.87); however, RG14 exhibited less macrophages when compared to CG14 (p = 0.06) (Figure 5).
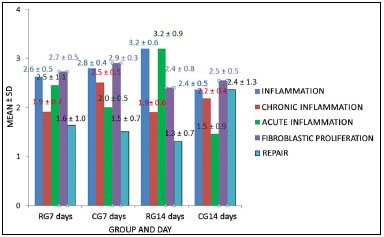
Figure 5. Comparison of the analysis made by HE in relation to inflammation, chronic inflammation, acute inflammation, fibroblast proliferation and repair in the control group (CG) and radiofrequency group (RG) following euthanasia on day 7 or 14.
DISCUSSION
Although many mammalian species simulate human healing, there are no sufficiently similar models. In mouse skin, there is no definition between the papillary dermis and reticular dermis. This animal does not form hypertrophic scars or keloids, has a thicker dermis, and has no subcutaneous fat tissue or subcutaneous muscle. The subdermal blood vessels are present and they present the same infusion changes of macro- and microvascularization.
In this study, monopolar radiofrequency was chosen because of its excellent results in the treatment of skin laxity as well as increased fibroblast and collagen induction, which greatly influence healing4.
However, the radiofrequency device was developed for use in humans. The size of its application handle and especially the size of the conductive current board, are relevant factors that may have influenced the results4,5.
Comparing CG7 and RG7 regarding wound contraction assessments in the area at times surgery, 1 day, 3 days and day of euthanasia, it was found that on day 3 a smaller area of CG7 when compared against RG7, thus demonstrating a significant difference in p = 0.009.
When compared to the area on the day of euthanasia of CG14 and RG14, a significantly smaller area was found in CG14 relative to RG14 (p = 0.001). This shows that, macroscopically, the healing of skin wounds undergoes prolonged inflammatory process following exposure to radiofrequency, which prevents the wound from contracting and closing in the groups in which the radiofrequency was applied.
In the macroscopic analysis of the punch, it was observed whether or not re-epithelialization of the wound occurred. In CG7, 30% were re-epithelialized while only 8.34% closed at RG7; 90.91% of the wounds in CG14 closed, while 60% closed in RG14. These results demonstrate the possible influence of radiofrequency on the re-epithelialization of wounds.
Histological evaluation of the healing process was performed with hematoxylin-eosin staining to quantify and identify the cellular elements in the inflammatory, proliferative and remodeling phases of the healing process. The inflammatory response is important for healing, but it is can be harmful since it can impair local microcirculation and the proliferation of fibroblasts6.
Correlating the contraction of the wound with HE, it was observed that the wound did not heal completely and remained ulcerated in GR14. On day 14, a higher number of inflammatory cells and cellular elements of acute inflammation were observed (i.e., neutrophils that were not present in the control group).
In RG7, chronic inflammatory cells were reduced, presumably due to the offending agent (radiofrequency in this case), which demonstrates the influence of heat on the healing of damaged tissue through connective tissue growth7.
Inflammation was similar in CG7 and RG7, which is within the expected repair time. In CG14, attenuation of inflammation was observed on day 14, while, in RG14, it was persistent. These findings suggest that radiofrequency increases vasodilation, heat, and edema, thereby lengthening the inflammatory process and impairing repair. Borges et al.8 and Carvalho et al.3 reported that the action of chemotactic agents during the inflammatory phase ends after seven days, even though the number of blood vessels is increased in the region.
Chronic inflammation was present in CG14 and RG14, which is similar to previous reports given that chronic inflammation has a prolonged duration and can last for weeks or months7.
In the HE analysis of RG14, we found that there is an increase in acute inflammation cells. According to Abbas et al.7, this should not occur at this stage since acute inflammation is a response that serves to bring leukocytes and plasma proteins to the injured site.
Since inflammation only subsides when the offending agent is eliminated, these findings suggest that the influence of radiofrequency in this group was to prolong the inflammatory process. The inflammatory response is linked to the repair process, while it destroys, dilutes and removes injurious agents, it tries to heal damaged tissue. The repair begins during inflammation, but usually ends after the injurious influence (radiofrequency in this case) has been terminated.
In the study by Choi et al.9, in which radiofrequency sessions were conducted in rabbits, histological analysis demonstrated the most prominent inflammatory reactions with internal inflammatory cell growth were associated with acute inflammation compared to the control group. This effect became more prominent with evolution due to nanostructural changes in dermal collagen fibrils.
In a study by Carvalho et al.3, which assessed the effects of applying radiofrequency on the skin of 20 mice in three sessions on alternate days, collagen became more dense after seven days after applications and included neocollagenesis. After 15 days, there was no more important neocollagenesis; however, neoelastogenesis was present on days 15 and 21.
An intense inflammatory process was observed in the presence of epithelial tear and edematous areas after 24 hours of RF application. This is similar to the results of the study by Brown & Almeida10, which suggests that RF leads to inflammation in the tissue and induces the formation of edema and increased vascularization, which may give rise to the onset of hemorrhages and the stimulation of fibroblasts3,10. Similar findings were also found in this work.
In the studies by Carvalho et al.3, three RF applications using a temperature of 37ºC for two minutes in the Wistar rat dorsum were shown to result in neocollagenesis in analyses at sacrifice as well as at 24 hours and seven days after the last application. In groups with sacrifice at 15 and 21 days after the last application could not find changes in tissue collagen nor could they detect the presence of neocollagenesis. However, after investigating possible changes in elastic fibers, neoelastogenesis was observed.
Similar studies exist, but with the use of other treatment modalities, such as laser-induced wound healing in rats. Heat can affect the scar healing process, which is often a complex and unpredictable process, and it is not uncommon to see the development of the healing process present with hypertrophic scars or keloids.
In the study of Mordon et al.11, a laser was applied immediately after the sutured incision (T) in the breast with a fluence of 110 J/cm². This study showed a marked reduction of type I collagen and fibronectin in lasertreated scars, while there was a decrease in type III collagen. This indicates that the pathological healing process is associated with increased rates of collagen type I and III formation, while a low amount supports regeneration.
According to Brown & Almeida10, radiofrequency causes a series of inflammatory events in the tissue, especially increased vascularization, blood flow, edema, which may cause the appearance of bleeding and the stimulation of fibroblasts. This corroborates the work of Del Pino et al.12, which demonstrated that radiofrequency increases the temperature in the deeper layers of the dermis, thereby causing morphological changes, such as increased vascularization and fibroblast induction.
Abercrombie et al. apud Yaguishita13 showed that contraction and physiological collagenization of rats in the quadrangular lumbar back injury mechanisms are the most important mechanisms for tissue repair in these animals. Contraction began on POD5 and ended 10 days later, while fibrosis began on POD5 and continued after the contraction has ended and the collagen fibers appeared 25 days after the experiment.
Therefore, the effects of RF in contraction and wound healing appear to be damaging during the initial postoperative period; however, the data of this study suggest an effect of increased fibroblast induction in fibroblastic proliferation. Nevertheless, these observations were not significant, which encourages further research to assess the level of collagen induction.
Prospects point to a late application of radiofrequency in skin healing after surgery in order to observe the results in another phase of healing, corroborating Agne14 and Moreira & Giusti15, who demonstrated that the effects of RF persist 1 to 4 months after application. Another possible treatment improvement is to reduce the RF dose.
Further studies with other types of animals that have greater similarity to human tissue should be performed while considering different findings concerning the dosimetry of radio frequency, temperature, the number of sessions and collagen cell count.
CONCLUSION
In relation to the effects of radiofrequency, we conclude that, after three RF applications on alternate days at 38ºC on the back of Wistar rats, radiofrequency-treated wounds showed delayed contraction of exceptional wounds and extension of the acute inflammatory process time, which was maintained for a longer period than the physiological time. It is speculated that radiofrequency adversely affects the healing process by excessive thermal injury.
COLLABORATIONS
AMCC Analysis and/or interpretation of data; statistical analysis; conception and design of the study; surgeries and/or experiments; writing of the manuscript or critical review of its content.
ACC Drafting of the manuscript or critical review of its content.
GFDE Surgeries and/or experiments.
PB Surgeries and/or experiments.
REFERENCES
1. Guirro E, Guirro R. Fisioterapia Dermato-Funcional: fundamentos recursos e patologias. 3a ed. São Paulo: Manole; 2007.
2. Atiyeh BS, Dibo SA. Nonsurgical nonablative treatment of aging skin: radiofrequency technologies between aggressive marketing and evidence-based efficacy. Aesthetic Plast Surg. 2009;33(3):283-94.
3. Carvalho GF, Silva RMV, Mesquita Filho JJT, Meyer PF, Ronzio AO, Medeiros JO, et al. Avaliação dos efeitos da radiofrequência no tecido conjuntivo. Arq Med. 2011;11(68)10-25.
4. Santos MFS, Czeczko NG, Nassif PAN, Ribas-Filho JM, Alencar BLF, Malafaia O, et al. Avaliação do uso do extrato bruto de Jatropha gossypiifolia L. na cicatrização de feridas cutâneas em ratos. Acta Cir Bras. 2006;21(suppl.3):2-7.
5. Bettes PSL. Análise comparativa histológica e tensiométrica entre a cicatrização deferidas cutâneas tratadas com o adesivooctil-2-cianoacrilato e com sutura intradérmica em ratos [Tese doutorado]. Curitiba: Universidade Federal do Paraná; 2003.
6. Gomes CS, Campos ACL, Torres OJM, Vasconcelos PRL, Moreira ATR, Tenório SB, et al. Efeito do extrato de Passiflora edulis na cicatrização da parede abdominal de ratos: estudo morfológico e tensiométrico. Acta Cir Bras. 2006;21(suppl.2):9-16.
7. Abbas K, Kumar V, Fausto N. Robbins l & Cotran R - Patologia - Bases Patológicas das Doenças. Rio de Janeiro: Elsevier; 2010.
8. Borges FS. Modalidades terapêuticas nas disfunções estéticas. São Paulo: Phorte; 2010.
9. Choi S, Cheong Y, Shin JH, Lee HJ, Lee GJ, Choi SK, et al. Short-term nanostructural effects of high radiofrequency treatment on the skin tissues of rabbits. Lasers Med Sci. 2012;27(5):923-33.
10. Brown A, Almeida G. Novel radiofrequecy device for cellulite & body reshaping therapy. Alma Laser. 2005 [citado 2014 Ago 30]. Disponível em: http://www.almalasers.com/sites/default/files/downloads/Novel_RF_Device_WP.pdf
11. Mordon S, Capon A, Fournier N, Iarmarcovai G. Lasers thermiques et cicatrisation cutanée. Med Sci (Paris). 2010;26(1):89-94.
12. Emilia del Pino M, Rosado RH, Azuela A, Graciela Guzmán M, Argüelles D, Rodríguez C, et al. Effect of controlled volumetric tissue heating with radiofrequency on cellulite and the subcutaneous tissue of the buttocks and thighs. J Drugs Dermatol. 2006;5(8):714-22.
13. Yaguishita N. Cicatrização induzida pela membrana de celulose porosa (Membracel®) em dorso de ratos [Dissertação de mestrado]. Curitiba: Universidade Federal do Paraná; 2006.
14. Agne JE. Eu sei eletroterapia. 2a ed. Santa Maria: Pallotti; 2009.
15. Moreira JAR, Giusti HHKD. A fisioterapia dermato-funcional no tratamento de estrias: revisão de literatura. Rev Cient Uniararas. 2013;1(2):22-32.
1. Universidade Federal do Paraná, Curitiba, PR, Brazil
2. Curso de Fisioterapia na Faculdade Paranaense, Curitiba, PR, Brazil
3. Pontifícia Universidade Católica do Paraná, Curitiba, PR, Brazil
Institution: Universidade Federal do Paraná, Curitiba, PR, Brazil.
Corresponding author:
Ana Maria Cardoso Cepeda
Rua Marechal José Bernardino Bormann, 1508, apt 103 - Bigorrilho
Curitiba, PR, Brazil Zip Code 80730-350
E-mail: anamariaceped@hotmail.com
Article received: August 13, 2015.
Article accepted: June 5, 2016.
Conflicts of interest: none.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter