

Original Article - Year 2017 - Volume 32 -
Procedures in the care of hand burns in the acute phase
Procedimentos no atendimento para queimaduras nas mãos na fase aguda
ABSTRACT
INTRODUCTION: The hand represents 3% of total body surface, but its involvement in severe trauma, such as burns, can lead to serious functional sequelae. The present study describes procedures in the care of patients in the acute phase with burns involving the hands.
METHODS: 122 patients with thermal burns involving one or both hands were included in the study between August 2011 and July 2014. On initial physical examination, the extent and depth of the lesions were determined; deep and circular burns, with signs and symptoms of inadequate perfusion, had escharotomies performed to avoid the loss of extremity. Burns of partial deep thickness or full thickness on the back of the hands received tangential excision within 5 days after the trauma; partial skin grafting was performed on all patients before the 15th day of the trauma.
RESULTS: Men represented the majority of cases (58%). In relation to the burn agent, flammable liquids caused 46.7% of admissions. In 50.8% of the cases, the burns were of partial deep thickness or of total thickness and required an operative procedure. No graft loss was identified in the group. Escharotomies were performed in 12.3% of the patients.
CONCLUSION: The burned hand needs proper local attention and care, along with early excision and skin grafting.
Keywords: Hands; Burns; Injuries; Skin transplantation; Reconstructive surgical procedures.
RESUMO
INTRODUÇÃO: A mão representa 3% da superfície corporal total, porém seu envolvimento em trauma grave, como uma queimadura, pode levar a sequelas funcionais graves. O presente estudo descreve os procedimentos no atendimento de pacientes na fase aguda com queimaduras envolvendo as mãos.
MÉTODOS: 122 pacientes com queimaduras térmicas envolvendo uma ou ambas as mãos foram incluídos no estudo, entre agosto de 2011 a julho de 2014. No exame físico inicial, determinou-se a extensão e a profundidade da lesão; as queimaduras profundas e circulares, com sinais e sintomas de perfusão inadequada, tiveram escarotomias realizadas para evitar perda de extremidade. As queimaduras de espessura parcial profunda ou de espessura total no dorso das mãos foram submetidas à excisão tangencial até 5 dias após o trauma; e o enxerto de pele parcial foi realizado em todos os pacientes antes do 15º dia do trauma.
RESULTADOS: Os homens representaram a maioria (58% da casuística). Em relação ao agente das queimaduras, os líquidos inflamáveis corresponderam a 46,7% das internações. Em 50,8% dos casos, as queimaduras eram de espessura parcial profunda ou de espessura total e necessitaram de procedimento operatório. Não foi identificada perda de enxerto no grupo. As escarotomias foram realizadas em 12,3% dos pacientes.
CONCLUSÃO: A mão queimada necessita de atenção e cuidados locais adequados, juntamente com excisão precoce e enxertia de pele.
Palavras-chave: Mãos; Queimaduras; Lesões; Transplante de pele; Procedimentos cirúrgicos reconstrutivos.
Burns are a severe trauma with social and economic repercussions that affect all age groups, but their incidence is higher in children and adults during their active years1-3. Thus, the home environment is the most common place where accidents occur, followed by work accidents in adults. In addition, their treatment is basically funded by the government; therefore, education and prevention measures must be pursued.
In this context, the hand is more susceptible to burn trauma because it is usually closer to the causative agent or because it is used by the victims in an attempt to protect themselves at the time of the accident1. Epidemiological studies have shown that the majority of large burns (over 25 % of burned body surface) have one or both of the hands affected, reaching 90% of the cases1-3. Children are the most affected group in the domestic environment and men in the workplace2-4.
The treatment of hand burns aims to restore local coverage and maintain functional movements in the first 15 days after thermal injury2. Decisions on the proper treatment of burns vary mainly depending on the depth and location of the burn and the age of the patient4. The depth or "degree" of burns is one of the most influential factors in initial treatment measures4-6. The hand comprises a small portion of body surface, but its functional loss prevents a normal adult from performing more than 50% of daily functions7.
The American Burn Association considers hand burns a serious injury with indications for specialized treatment in a dedicated unit1. The Ministry of Health Guidelines for Burns in Brazil also consider cases of patients with one or both burned hands to be of high severity; they must be quickly transferred to specialized units8.
Given all these factors involved in hand burns and the importance of minimizing the sequelae that can affect patients, this study presents a standardized clinical and surgical treatment of burns involving the hands.
OBJECTIVE
To describe the routine treatment of patients with burns involving one or both hands.
METHODS
Data were collected from the patients treated by the author from August 2011 to July 2014, at the Burns Treatment Unit of the Department of Plastic Surgery of the Paulista School of Medicine, Federal University of São Paulo (EPM/UNIFESP). The study followed the principles of the Declaration of Helsinki, 1964. Patients were included after reading and signing the Informed Consent Form. One hundred and twenty-two patients with thermal burns involving one or both hands were included in the protocol for the early treatment of burns involving the upper and lower extremities. The study population included only adults older than 18 years of age.
Procedures used in patient care with hand burns
Early Care
In the initial phase of care of patients with burns, the guidelines of the Advanced Trauma Life Support - ATLS® and the Ministry of Health Care Guidelines were followed.
The treatment of severe trauma was a priority in relation to the treatment of hand burns. However, early attention to lesions in special areas (face, ears, hands, feet, and genitalia) was considered fundamental to a successful outcome.
The history of the patient, as well as the mechanism of the injury and the conditions at the trauma site, were collected. The patient history also included occupation, dominant hand, and comorbidities.
The extent and depth of the burn was determined during the initial physical examination of the burned hand. In addition, warning signs of vascular insufficiency (decreased pulses and slow capillary refill) were assessed.
Calculation of burn injury extension (Lund and Browder Chart) and replacement fluid (Parkland Formula) was performed simultaneously. To assist in this calculation, complete removal of clothes and cleaning of burned areas with removal of phlyctena were necessary. Care to avoid hypothermia was utilized in all patients (infusion of heated crystalloids, ambient temperature above 25ºC).
Assessment of the depth of burns, especially if there are deep partial thickness and total thickness lesions, should be performed as early as possible, in addition to special attention for deep circumferential lesions in the limbs.
Escharotomy
Escharotomies were performed on clearly deep and circular burns, with signs and symptoms of inadequate perfusion, such as decreased capillary refill of the fingers (less than 3 seconds), claw hand deformity, pain on passive extension of the fingers, and paresthesia (Figure 1).
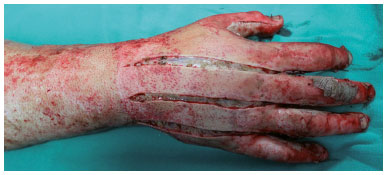
Figure 1. Escharotomy performed on the back of the hand with total thickness burn. The incision reaches the full thickness of the eschar and exposes the subcutaneous tissue. The incisions between the interdigital spaces allow access and release the space between the interosseous muscles, aiding in the return of tissue perfusion to the fingers.
The procedure was performed according to the following criteria:
- Escharotomy was indicated within the first 24 hours after trauma;
- Escharactomy was completed in one's own bed;
- Analgesia with intravenous opioids, preferably morphine was provided;
- In previously sedated patients, due to the need for mechanical ventilation, fentanyl or propofol were used to enhance analgesia;
- Asepsis and antisepsis care were provided;
- Sterile surgical fields were used;
- Attention was paid to avoid lesions of underlying noble structures;
- Electrocautery was used to prevent major bleeding;
- Operative technique: Axial incisions were performed using a cold scalpel on the radial side of the upper limb, from the distal to the proximal end, reassessing the perfusion return so as not to invade areas of superficial burn or whole skin. If this was not sufficient, the author repeated the same type of incision on the ulnar side of the limb;
- The incision reached only the thickness of the eschar until the subcutaneous tissue was visualized;
- Immediate return of limb perfusion was the expected result. In the absence of improvement, fasciotomy was indicated.
Wound Dressings
The wide range of dressing material available for burn care today is not addressed in this study.
At the time of admission, blisters or phlyctena were ruptured and removed, and any foreign material from the wounds was removed prior to the application of the dressings.
In deep partial thickness and full thickness burns, silver sulfadiazine was applied on non-adherent gauze, followed by a dry gauze, cotton, and bandage (dressing in four layers). This procedure was performed, at most, daily for the first five days after the trauma.
For partial-thickness superficial burns and for epithelial burns, non-adherent gauze with vegetable oil (essential fatty acid) was used. The other layers of the dressing were maintained.
Each layer of dressing used has its function: the first layer with non-adherent gauze acts to keep the more suitable topical agent in contact with the wound bed. It is important to use non-adherent material to avoid traumatizing the lesion bed, major pain during the dressing exchange, and removing cells that have migrated to the surface to form new epithelium. The second layer of dry gauze absorbs exudate from the wound. The third layer with hydrophilic cotton also absorbs secretions and assists in maintaining body temperature. The fourth layer keeps the other layers in place; we usually use bandages.
The fundamental principle of the hand dressing was respected; that is, the hands were held in an anatomical functional position to facilitate movement.
Surgical treatment
Burns of partial deep thickness or full thickness on the dorsum of the hands received tangential excision within five days after the trauma. However, patients with burn extensions above 25%, with associated trauma, or with comorbidities had the surgical treatment delayed. The author avoided this period for more than ten days and pursued the goal of grafting within the first 15 days after the trauma. All patients were treated in a similar manner.
Excision of non-viable tissue was performed using a Blair knife. Tangential excision was chosen because it allows for the controlled debridement of non-viable tissues without exposing noble surface structures, such as the extensor tendons.
Following the desired excision of the necrosis, an epinephrine-soaked swab (concentration 1: 200,000) was applied to the wound bed for 10 minutes to aid in hemostasis. In punctual bleeds, electrocautery was used. After this step, a four-layer compressive dressing was applied to maintain hemostasis, without impairing the perfusion of the extremities.
Sheet grafting was chosen as the standard by the author to cover the dorsum of the hands and the dorsal surface of the fingers. The patients' vastus lateralis was the most used donor site. The skin was removed using an electric dermatome with a thickness of 0.5 mm (0.02 inches). The slides were fixed with suture (Nylon # 5). A four layer dressing with non-adherent gauze was used. Opening the dressing occurred from the first to the third post-operative day, according to the need to evaluate hematoma formation between the recipient bed and the sheet graft.
Rehabilitation in a hospital environment
After stabilizing the patient, the hands were elevated for 24 h to 48 h after the burn, to assist in the control of edema in this area. Perfusion of the extremities was re-evaluated every hour for the first 24 hours after the trauma.
During the period of expectant management, the thickness of the lesions was superficial and non-surgical treatment was preferable. Thus, we released patients for physiotherapy at least three times a day, starting soon after the injury to minimize edema and maximize the amplitude of movement. If the patient was sedated and intubated, i.e. incapable of actively moving the hand and digits, passive physiotherapy was performed, also early and daily.
Rehabilitation after Hospital Discharge
All patients received instructions about the importance of post-discharge rehabilitation. The following topics were presented and explained to the patients and family members.
- Need for physiotherapy;
- Use of compressive mesh;
- Use of silicone patches on scars with undesirable evolution;
Hydration of healed, donor, and grafted areas;
- Solar protection;
- Psychological follow-up;
- Occupational therapy and/or participation in support groups;
- Follow-up of the evolution of scars over time by specialized medical staff capable of correcting the onset of sequelae, and who could discontinue the above mentioned adjuvant treatments.
RESULTS
Men represented the majority of patients, 71 patients (58%), and women represented 51 cases (42%). The mean age of the men was 37 years and of the women 43.7 years. Regarding the burn agent, an alarming finding was discovered; flammable liquids caused 46.7% of hospitalizations, and in this group, liquid alcohol accounted for 87% of burns. Heated liquids occupied the second place (19.6% of the accidents), followed by flame (17.2%), and hot gas (9.8%). Finally, heated solids accounted for 6.5% of the cases.
Half of the patients enrolled in the study needed operative treatment for the hands (50.8%), and in this group, the lesions were partial deep thickness or total thickness. The superficial partial thickness burns corresponded to 49% of the affected hands and a conservative treatment with dressings was the adopted procedure. No graft loss was identified in the operated group. Escharotomies were performed in 12.3% of the patients, and in this group, all patients presented at least 30% of burned body surface. Nine patients died (7.3% of the series).
DISCUSSION
Burns cover three zones. The first, "hyperemia", is the outermost zone. Usually, the cells are not killed in this area and can heal quickly. The skin displays erythema, similar to a sunburn without blistering. The second zone, called "stasis," consists of an area with vascular damage that is potentially reversible if appropriate treatment is initiated early. Restoration of adequate blood flow to the stasis zone, with the correction of hypovolemia, allows recovery of the injured tissue.
The central area of the burn is the coagulation zone (third zone), where irreversible damage with cell death has already occurred at the time of trauma. The immediate and adequate treatment of the damage in the first two zones, those of hyperemia and stasis, prevents the progression of the severity of the burn. Without adequate treatment, the burn damage advances to the next zone, resulting in a greater area of deep and irreversible injury9.
Patients with large extensive burns may have initially neglected lesions on the hands and upper limbs, while more critical lesions are treated. Thus, rapid determination of lesion depth is important during initial care. Burns are classified according to the degree of damage to the epithelium, dermis, subcutaneous tissue, and other underlying structures.
First-degree burns involve damage only to the epidermis and do not show any open wounds or blisters. These heal without scarring and do not require surgical treatment. Second-degree burns are defined as partial thickness burns, which means that the lesion covers the epidermis and different degrees of dermis. These burns are classified into superficial and deep. This concept is important because second-degree superficial burns usually recover with local care within 10 to 14 days. These lesions present blistering and are painful due to exposure of the nerve endings in the dermis (Figures 2 and 3). Generally, scars and loss of function are not associated with these burns because the inflammatory phase is interrupted with the formation of the epidermis9.
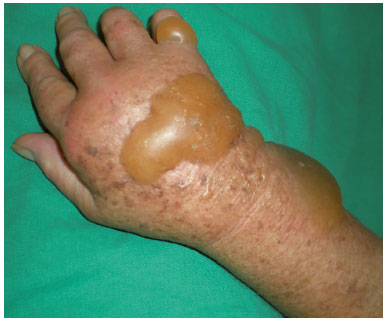
Figure 2. Second-degree burn with undamaged phlyctena.
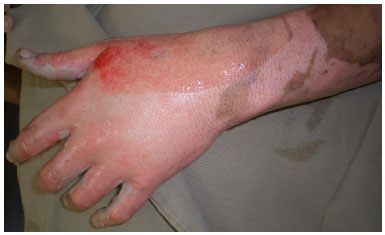
Figure 3. Burn of partial superficial thickness (second-degree) on the back of the hand, after removal of blisters and local cleaning. Presence of intact hair follicles, showing that the deepest part of the dermis is preserved and high probability of restoration without surgical intervention.
Deep second-degree burns affect the reticular dermis and destroy more epidermal attachments and nerve endings, causing less painful lesions characterized by an "eschar" rather than a blister. These lesions present an inflammatory phase of prolonged scarring caused by delayed restoration of the epidermis and extensive deposition of collagen, resulting in pathological scarring and functional impairment. These burns should be treated similarly to full-thickness burns, with excision of the devitalized tissue and skin grafts (Figures 4A, 4B, 4C, and 4D).
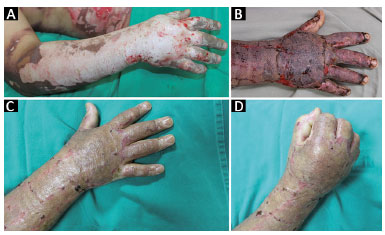
Figure 4. A: Male patient, deep second-degree burn (or partial deep thickness) caused by liquid alcohol and reaching the dominant upper limb; B: Sheet grafting performed on the dominant hand. Procedure was performed on the 10th day after the burn and on the third postoperative day; C: Thirty-days after skin grafting with good integration; D: Hand with normal functionality on the thirtieth day after skin grafting.
Third-degree burns involve the full thickness of the epidermis, dermis, and subcutaneous tissue. These wounds cannot be restored because of the total loss of epidermal attachments and dermis. They are routinely treated in the same way, with excision and grafting (Figure 5). Fourth-degree burns involve underlying structures, such as muscles, tendons, and bones. They are serious injuries requiring elaborate reconstructions and, occasionally, amputations9.
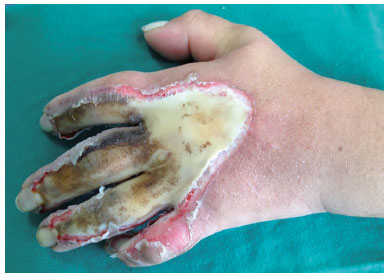
Figure 5. Full thickness (third-degree) burn in the dominant upper limb of a female patient caused by heated solid.
The diagnosis of inadequate vascular perfusion of the burned hand cannot be underestimated, and can be extremely difficult to achieve due to the change in skin color and damage to the normal anatomy. Usually, in superficial and partial thickness burns, perfusion of the hand is not compromised. However, full-thickness burns deserve attention. Secondary edema in large burns and subsequent volume replacement may also compromise hand perfusion. High-risk injuries such as circumferential burns, crush injuries, and lacerations can easily lead to vascular impairment. Thus, escharotomies should be performed within the first 24 hours after the burn10,11.
The dorsum of the hand has a relatively thin soft tissue cover. In the treatment of burns in general, the surgical option chosen depends on the location of the burn and the depth and experience of the surgeon. To address hand burns, tangential excision allows controlled debridement of non-viable tissues. There are a number of coverage options available; however, the partial thickness skin graft is the most common and allows a recovery of the amplitude of hand movements, in addition to a pleasant esthetic appearance (Figures 6A, 6B, 6C, 6D and 6E)12-14.
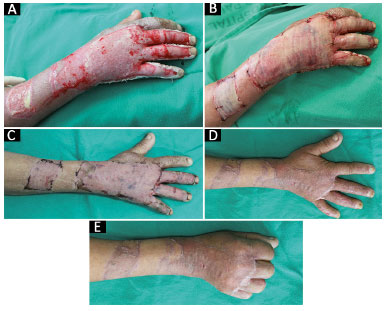
Figure 6. A: Male patient with deep second-degree burn caused by hot liquid (pre-operative); B: Intraoperative sheet skin graft, the most common procedure used to cover this area; C: Post-operative (one week) aspect; D: Postoperative (one month) aspect with agreeable esthetic appearance; E: Post-operative (one month) aspect with return of the amplitude of the movements of the hand.
Hypertrophic scarring, digital contractures, and losses (functional, physical, and psychological) are some of the complications that can occur with inadequate initial treatment. The characteristic appearance of the dorsum of the inadequately treated burned hand with the extension of the wrist and metacarpophalangeal joints is a deformity that can be avoided with early treatment.
Fixation of metacarpophalangeal joints flexed between 60 and 70 degrees with stainless steel wire, thumb fully adhered, and fully extended interphalangeal joints avoids deformity in patients who are unable to participate in therapy. Various splint modalities can be used to maintain the functional position of the hands before and after the skin graft. The splint should be removed daily for physical therapy (passive or active) and occupational therapy, according to the condition of the patient15.
The goals of dressings are to prevent heat and water loss, prevent infection, and promote skin regeneration. They should also be easy to apply, relatively painless, and inexpensive. Thus, any dressing should allow the patient to exercise and use the functions of the hand15.
CONCLUSION
Local care to prevent infection and promote good evolution of burns, along with early excision and grafting, are key elements for the treatment of burns in the hands.
COLLABORATIONS
AFO Analysis and/or interpretation of data; statistical analyses; conception and design of the study; completion of surgeries and/or experiments; writing the manuscript.
LMF Final approval of the manuscript; critical review of its contents.
REFERENCES
1. Gragnani A, Ferreira LM. Pesquisa em queimaduras. Rev Bras Queimaduras. 2009;8(3):91-6.
2. Dias LDF, Oliveira AF, Juliano Y, Ferreira LM. Unidade de Tratamento de Queimaduras da Universidade Federal de São Paulo: estudo epidemiológico. Rev Bras Cir Plást. 2015;30(1):86-92.
3. Narikawa R, Michelski DA, Hiraki PY, Ueda T, Nakamoto HA, Tuma Jr. P, et al. Análise epidemiológica da mão queimada no Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo. Rev Bras Queimaduras. 2011;10(3):89-92.
4. Kreymerman PA, Andres LA, Lucas HD, Silverman AL, Smith AA. Reconstruction of the burned hand. Plast Reconstr Surg. 2011;127(2):752-9. PMID: 21285778 DOI: http://dx.doi.org/10.1097/PRS.0b013e3181fed7c1
5. McKeeDM. Acute management of burn injuries to the hand and upper extremity. J Hand Surg Am. 2010;35(9):1542-4. DOI: http://dx.doi.org/10.1016/j.jhsa.2010.03.019
6. Greco Júnior JB, Moscozo MVA, Lopes Filho AL, Mendes CMGG, Tavares FMO, Oliveira GM, et al. Tratamento de pacientes queimados internados em hospital geral. Rev Bras Cir Plást. 2007;22(4):228-32.
7. Sterling J, Gibran NS, Klein MB. Acute management of hand burns. Hand Clin. 2009;25(4):453-9. DOI: http://dx.doi.org/10.1016/j.hcl.2009.06.010
8. Pan BS, Vu AT, Yakuboff KP. Management of the Acutely Burned Hand. J Hand Surg Am. 2015;40(7):1477-84. DOI: http://dx.doi.org/10.1016/j.jhsa.2015.02.033
9. Hettiaratchy S, Dziewulski P. ABC of burns: pathophysiology and types of burns. BMJ. 2004;328(7453):1427-9. PMID: 15191982 DOI: http://dx.doi.org/10.1136/bmj.328.7453.1427
10. Piccolo NS, Piccolo MS, Piccolo PD, Piccolo-Daher R, Piccolo ND, Piccolo MT. Escharotomies, fasciotomies and carpal tunnel release in burn patients--review of the literature and presentation of an algorithm for surgical decision making. Handchir Mikrochir Plast Chir. 2007;39(3):161-7. DOI: http://dx.doi.org/10.1055/s-2007-965322
11. Wong L, Spencer RJ. Escarothomy and fasciotomy of the burned upper extremity. Hand Clin. 2000;16(2):165-74.
12. Kamolz LP, Kitzinger HB, Karle B, Frey M. The treatment of hand burns. Burns. 2009;35(3):327-37. DOI: http://dx.doi.org/10.1016/j.burns.2008.08.004
13. Richards WT, Vergara E, Dalaly DG, Coady-Fariborzian L, Mozingo DW. Acute surgical management of hand burns. J Hand Surg Am. 2014;39(10):2075-85. DOI: http://dx.doi.org/10.1016/j.jhsa.2014.07.032
14. Mohammadi AA, Bakhshaeekia AR, Marzban S, Abbasi S, Ashraf AR, Mohammadi MK, et al. Early excision and skin grafting versus delayed skin grafting in deep hand burns (a randomised clinical controlled trial). Burns. 2011;37(1):36-41. PMID: 20537468 DOI: http://dx.doi.org/10.1016/j.burns.2010.02.005
15. Kowalske KJ. Hand burns. Phys Med Rehabil Clin N Am. 2011;22(2):249-59. DOI: http://dx.doi.org/10.1016/j.pmr.2011.03.003
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
2. Universidade Federal de São Paulo, Escola Paulista de Medicina, São Paulo, SP, Brazil
Institution: Universidade Federal de São Paulo, Escola Paulista de Medicina, São Paulo, SP, Brazil.
Corresponding author:
Andrea Fernandes de Oliveira
Rua Napoleão de Barros, 715, 4º andar - Vila Clementino
São Paulo, SP, Brazil Zip Code 04024-002
E-mail: sandra.dcir@gmail.com / dra.afo@gmail.com
Article received: August 11, 2015.
Article accepted: September 29, 2015.
Conflicts of interest: none.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter