

Original Article - Year 2017 - Volume 32 -
Symmetrization using submuscular implants to achieve lasting results in breast reconstruction
Simetrização com implantes submusculares na busca de resultados duradouros em reconstruções mamárias
ABSTRACT
INTRODUCTION: In breast reconstruction with implants, ptosis is uncommon over time, in contrast to symmetrization mammoplasty without implants, which causes premature recurrence of breast asymmetry. The objective of this study was to describe the procedure and results of contralateral symmetrization mammoplasty with the use of submuscular implants.
METHODS: A total of 18 cases of symmetrization mammoplasty with submuscular implants performed over a 2-year period were retrospectively studied.
RESULTS: The most common comorbidity was obesity (p = 0.007). Obese patients had a higher volume of resected breast tissue (p = 0.0013). The mean follow-up was 13 months. The reoperation rate was 5.5% (1 case) for pseudoptosis. There were no implant infections. Complications included partial necrosis of the nipple-areola complex in 1 case, superficial wound dehiscence in 1 case, seroma in 3 cases, and moderate pain in 3 cases.
CONCLUSION: Contralateral symmetrization mammoplasty with submuscular implants, associated with adequate emptying of breast tissue, provided long-lasting results with a low rate of complications, minimizing the sequelae of mastectomy.
Keywords: Mammoplasty; Breast implant; Mastectomy; Breast cancer.
RESUMO
INTRODUÇÃO: Nas reconstruções de mama com implante é incomum a ptose no decorrer do tempo, fato não observado nas mamoplastias de simetrização, principalmente sem implantes, ocasionando precocemente a recidiva da assimetria mamária. O objetivo desse trabalho foi demonstrar a mamoplastia de simetrização contralateral com uso de implantes submusculares e os resultados alcançados.
MÉTODOS: No período de 2 anos foram estudados, retrospectivamente, 18 casos de mamoplastia de simetrização com implante submuscular.
RESULTADOS: A comorbidade mais encontrada foi a obesidade, com p = 0,007. As pacientes obesas tiveram maior volume de tecido mamário ressecado, com p = 0,0013. O acompanhamento médio foi de 13 meses. A taxa de reoperação foi de 5,5% (1 caso) devido à pseudoptose. Não houve infecção dos implantes. As complicações encontradas foram: necrose parcial do complexo areolopapilar em 1 caso, deiscência superficial de ferida em 1 caso, seroma em 3 casos e dor moderada em 3 casos.
CONCLUSÃO: A mamoplastia de simetrização contralateral com uso de implantes submusculares, associada ao adequado esvaziamento do tecido mamário, proporcionou resultados duradouros com baixo índice de complicações, minimizando as sequelas decorrentes da mastectomia.
Palavras-chave: Mamoplastia; Implante mamário; Mastectomia; Neoplasias da mama.
According to estimates by the National Cancer Institute in Brazil, approximately 57,120 women were diagnosed with breast cancer in 20141. The standard treatment for non-metastatic breast cancer is surgery, with or without radiotherapy and/or chemotherapy in accordance with the cancer stage. Breast reconstruction preserves self-image while minimizing the psychosocial sequelae resulting from mastectomy. This results in a positive impact on the quality of life2 and makes the procedure more accepted and requested by younger patients3, in whom breast expanders and implants have been increasingly used4.
Regardless of the technique, there will almost always be asymmetry between the breasts after unilateral breast reconstruction, especially when a silicone prosthesis is used in only one breast. Therefore, breast symmetrization surgery is, almost always, an inevitable indication.
In a breast reconstructed with an implant, ptosis is uncommon over time due to the lack of tissue, inelasticity of the skin due to radiotherapy, and the presence of the implant in the submuscular position. However, in symmetrized breasts, early progression is common, with development of pseudoptosis and even grade I5 ptosis in less than 12 months, resulting in recurrence of mammary asymmetry (Figure 1).
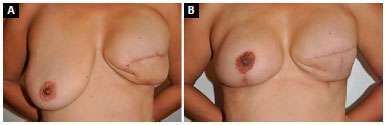
Figure 1. A: Reconstruction of left breast with expander and asymmetry arising from contralateral breast ptosis; B: Recurrence of breast asymmetry at 9 months after mastopexy with subglandular implant.
OBJECTIVE
The objective of this study was to describe the procedure and results of contralateral symmetrization mammoplasty with the use of submuscular implants.
METHODS
This was a retrospective study based on the analysis of medical records of patients seen in a private practice who underwent surgery in three private hospitals in the Federal District (Hospital Daher Lago Sul, Brasilia Hospital, and HOME Hospital), between March 2013 and March 2015. All cases underwent surgery by the same surgeon, the author of the study. Patients who underwent breast symmetrization mammoplasty with submuscular implants were included in the study. There were no exclusion criteria.
All patients signed an Informed Consent Form and the study followed the guidelines of the Declaration of Helsinki, pending approval by an ethics committee.
Statistical analysis
The data were collected and stored in a database created with Microsoft Excel, which was also used for statistical analysis and construction of graphs and tables. The distribution frequencies of the categorical and ordinal variables collected from medical records and clinical-surgical observations were analyzed. All data are presented in the tables as measures ± standard deviation or absolute values (percentage). The Kolmogorov-Smirnov test was used to evaluate the normality of the quantitative variables. Continuous variables were analyzed with Student's t-test. Categorical variables were analyzed with the chi-square or Fisher's exact test when appropriate. A p value was considered significant when less than 0.05. The analysis was performed with SPSS software for Macintosh version 20.0 (SPSS, Chicago, IL, USA).
Description of the operative technique
During a mastectomy, the excess skin and degree of ptosis in the cancerous breast were evaluated and, when necessary, Pitanguy-type or similar marking, was performed to remove excess skin. In the second symmetrization phase in the contralateral breast, we followed the same skin marking as for correction of ptosis and removed adequate mammary tissue to mimic the mastectomy, but preserved the vascularization of the nipple-areola complex (NAC) and the contour of the breast for adequate coverage of the implant to be placed in the submuscular plane. In the mastectomy area, we performed fat grafting following the same concept (Figure 2).
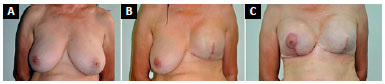
Figure 2. A: Preoperative aspect of patient with left breast cancer associated with grade III ptosis; B: At 6 months after breast reconstruction with a
submuscular expander and Pitanguy-type marking for correction of excess skin; C: At three months after mammoplasty with submuscular implant.
The placement of the submuscular implant follows a previously published approach6: with access between the fibers of the pectoralis major muscle and preparation of a partial submuscular pocket, through the inferior disinsertion of the pectoral muscle by incision and release of the connection between the muscle and the fascia of the rectus abdominis, but with preservation of the adherence of the muscle to the fat pad for coverage and non-exposure of the implant at the lower pole, enabling lower anatomical positioning in relation to the mammary groove. This same concept was used in the placement of the expander in immediate reconstruction (Figure 3).
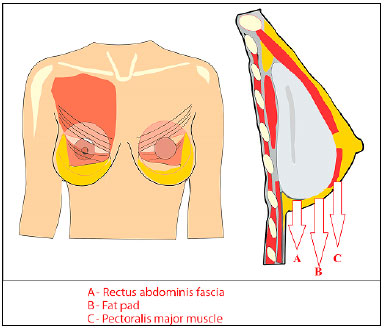
Figure 3. Schematic drawing of the partial submuscular implant with coverage in the lower pole by preservation of the fat pad (B) and section of the connection
of the pectoral muscle (C) to the rectus abdominis fascia (A), thus achieving a better implant position in relation to the mammary groove.
Another important point is the disinsertion of muscle in the mid-sternal region up to the level of the 4th intercostal space, minimizing the inconvenience resulting from mobilization of the implant with muscle contraction7. The total closure of the fibers and global coverage of the implant in the central portion may not be possible, depending on the size of the implant, and one should only perform approximation of the fibers without compromising the stability of the implant (Figure 4).
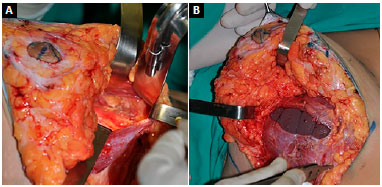
Figure 4. A: Preparation of the submuscular pocket via mammoplasty with disinsertion of muscle fibers in the inferomedial portion; B: Implant positioned with partial closing of muscle fibers and preservation of the fat pad in the lower pole for coverage of the implant.
Elevation of the NAC is performed with a superior pedicle or medial pedicle technique, depending on the case. Tall, textured, round implants are used; the implant pocket is irrigated with 500 ml of saline solution plus 1 g of cefazolin. Tubular drains are placed routinely, and remain until drainage is less than 50 ml/24 h.
RESULTS
Symmetrization with submuscular implantation was performed in 18 patients (Figures 5, 6, 7, and 8). Seventeen cases underwent reconstructed breast symmetrization with expanders and 1 case was reconstructed with a latissimus dorsi muscle flap and prosthesis. Ten patients, about 56%, underwent adjuvant radiotherapy. The age ranged from 32 to 77 years, with a mean of 52 years. The most frequent comorbidity was obesity in 8 cases (p = 0.007). Hypercholesterolemia was present in 7 cases, hypertension in 4, diabetes in 3, and arrhythmia, depression, and hypothyroidism in 1 case each. Some patients had combined comorbidities, e.g., 1 patient with NAC necrosis had diabetes, hypertension, hypercholesterolemia, and obesity. No patient reported smoking (Figure 9).
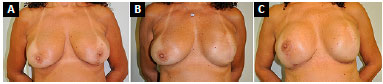
Figure 5. A: Preoperative aspect of left breast cancer; B: Seven months after left breast reconstruction with submuscular expander; C: Three months after postoperative exchange of expander with a prosthesis and symmetrization with submuscular implant.

Figure 6. A: Preoperative right breast cancer; B: 5 months after right breast reconstruction with submuscular expander after mastectomy, with preservation of the nipple areola complex (NAC); C: 3 months after expander replacement with a prosthesis, in addition to mastopexy and contralateral breast symmetrization with submuscular implant.
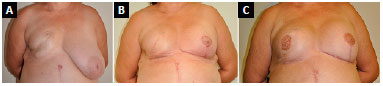
Figure 7. A: Postoperative reconstruction of right breast with submuscular expander, followed by adjuvant radiotherapy; B: 5 months after expander replacement with a prosthesis associated with fat grafting and contralateral breast symmetrization with submuscular implant; C: 11 months after new fat grafting in the right breast and reconstruction of the nipple-areola complex without evidence of ptosis in the symmetrized breast.
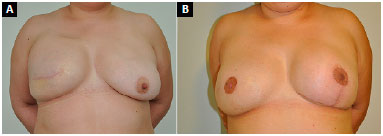
Figure 8. A: Postoperative appearance of right breast reconstruction with submuscular expander; B: 10 months after expander exchange with a prosthesis, combined with fat grafting and contralateral breast symmetrization with a submuscular implant, and a third operation 3 months later for right nipple-areola complex reconstruction.
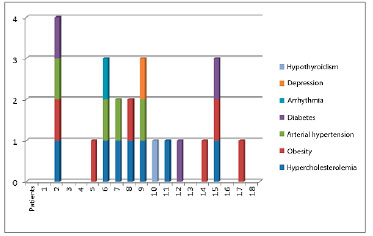
Figure 9. Distribution of patients and comorbidities, with 13 patients having one or more comorbidities and only 5 patients without other diseases.
Three patients had breasts with grade I ptosis, 2 had grade II ptosis, and 13 had grade III ptosis (Figure 10). The resection of breast tissue ranged from 177 to 1,152 g, with an average of 500 g. Obese women had a greater volume of breast tissue removed (p = 0.013). The 18 symmetrization procedures were performed with round, high-profile textured implants, with volumes ranging from 175 to 275 cm3. The time from breast reconstruction until symmetrization surgery ranged from 3 to 50 months, with an average of 10 months.
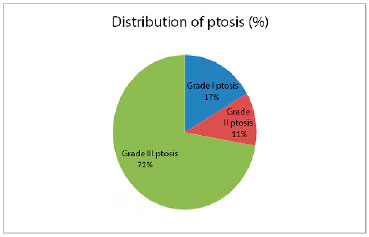
Figure 10. This graph demonstrates that most patients had severe ptosis of the breast, which was not a contraindication for the technique.
Follow-up ranged from 4 to 25 months, with an average of 13 months. A new mastopexy was required in a 77-year-old patient for pseudoptosis after 12 months of follow-up (Figure 11). There were no implant infections. One patient had vascular distress and partial necrosis (20%) of the NAC after elevation with a medial pedicle, without need for surgical reintervention, and with resolution by second intention healing (Figure 12). One patient had superficial dehiscence of the vertical mammoplasty scar, without exposure of the implant, and with resolution by secondary intention healing. Three patients developed seromas, with spontaneous resolution within 45 days. Three patients reported moderate pain in the symmetrized breast (Table 1).
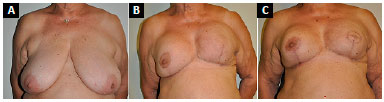
Figure 11. A: Preoperative appearance of patient with left breast cancer associated with grade III ptosis; B: 12 months after surgery with pseudoptosis of symmetrized right breast with submuscular prosthesis and expander replacement with a prosthesis in left breast reconstruction; C: Three months after new right mastopexy with maintenance of implant position and reconstruction of the left nipple-areola complex.
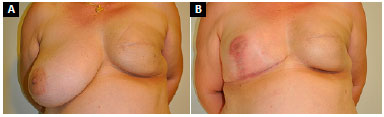
Figure 12. A: Postoperative appearance of left breast reconstruction with submuscular expander followed by adjuvant radiotherapy; B: 3 months after expander exchange with a prosthesis combined with fat grafting and right breast symmetrization with submuscular prosthesis, complicated by partial necrosis of right nipple-areola complex, and resolution by second intention healing.
DISCUSSION
After mastectomy, reconstruction with expanders or permanent submuscular prostheses tends to result in a breast cone without ptosis and with a shape similar to the implant, because of resection of breast tissue and sagging of subcutaneous tissue. Despite the availability of different techniques, we considered three major points as a basis for the choice of symmetrization method.
The first is the use of a contralateral breast prosthesis; the second is the placement of a prosthesis in the submuscular plane, similar to that in the reconstructed breast; and the third is the proper emptying of tissue in the remaining breast to minimize future ptosis and the development of a double-bubble deformity8.
In this sense, mastopexy or reductive mammoplasty alone, in which a good part of the breast tissue is preserved, would not be adequate, due to the high rate of recurrence of ptosis and asymmetry. In addition, the surgical tactic of breast emptying may have the benefit of exploring the contralateral breast and diagnosing a hidden cancer in up to 10 to 15% of cases9. Moreover, according to Spear et al.10, prophylactic subcutaneous adenomastectomy can be indicated in patients over 65 years old and in those with mutation in BRCA genes 1 and 2.
The results of this study suggest that the described technique can also be used in patients with obesity, which is the most frequent comorbidity (p = 0.007); these patients also had a larger resected breast volume (p = 0.0013). Thus, gigantomastia and grade III ptosis would not be contraindications for the technique, which in this study showed low reoperation rates (5.5%), varying only in the resulting scar and the NAC elevation technique. We believe it is important to continue the study to strengthen the results.
In this case, fat grafting was an important step in symmetrization. It was used to model the new breast, improve the thickness of subcutaneous tissue, and to minimize the complications of radiodermatitis and inelasticity of the skin resulting from radiotherapy. Ten patients who underwent radiotherapy benefited from this procedure without increasing locoregional recurrence11.
We irrigate the surgical area with saline solution mixed with antibiotic, although studies have demonstrated no statistical difference when compared to irrigation with saline solution alone, with regard to the rate of infection and capsular contracture12. These complications were more evident in patients with comorbidities13, and may be subject of a future study.
CONCLUSION
Contralateral symmetrization mammoplasty with the use of submuscular implants, after adequate emptying of breast tissue, provided long-lasting results, with a low rate of complications, and enhanced the quality of breast reconstruction results.
COLLABORATIONS
DBP Analysis and/or interpretation of data; final approval of the manuscript; conception and design of the study; completion of surgeries and/or experiments; writing the manuscript or critical review of its contents.
FTM Statistical analyses.
REFERENCES
1. Brasil. Ministério da Saúde. Instituto Nacional do Câncer José de Alencar Gomes da Silva [acesso 2017 Out 20]. Disponível em: http://www.inca.gov.br/rbc/n_60/v01/pdf/11-resenha-estimativa-2014-incidencia-de-cancer-no-brasil.pdf
2. Rondelo JC, Martino MD, Mermerian T, Veiga DF, Abla LEF, Gebrin LH, et al. Qualidade de vida em pacientes submetidas à reconstrução de mama com retalho miocutâneo transverso do reto abdominal. Rev Bras Cir Plást. 2014;29(1):79-83.
3. Keith DJ, Walker MB, Walker LG, Heys SD, Sarkar TK, Hutcheon AW, et al. Women who wish breast reconstruction: characteristics, fears, and hopes. Plast Reconstr Surg. 2003;111(3):1051-6. DOI: http://dx.doi.org/10.1097/01.PRS.0000046247.56810.40
4. Esteves BP, Cammarota MC, Daher JC, Camara Filho JPP, Borgatto MS, Curado DMC. Reconstrução mamária em mulheres jovens: análise de 5 anos de experiência. Rev Bras Cir Plást. 2013;28(3):67.
5. Regnault P. The hypoplastic and ptotic breast: a combined operation with prosthetic augmentation. Plast Reconstr Surg. 1966;37(1):31-7. PMID: 5903218 DOI: http://dx.doi.org/10.1097/00006534-196637010-00004
6. Daher JC, Amaral JDLG, Pedroso DB, Cintra Júnior R, Borgatto MS. Mastopexia associada a implante de silicone submuscular ou subglandular: sistematização das escolhas e dificuldades. Rev Bras Cir Plást. 2012;27(2):294-300. DOI: http://dx.doi.org/10.1590/S1983-51752012000200021
7. Spear SL, Schwartz J, Dayan JH, Clemens MW. Outcome assessment of breast distortion following submuscular breast augmentation. Aesthetic Plast Surg. 2009;33(1):44-8. PMID: 19052809 DOI: http://dx.doi.org/10.1007/s00266-008-9275-y
8. Tebbetts JB. Dual plane breast augmentation: optimizing implant-soft-tissue relationships in a wide range of breast types. Plast Reconstr Surg. 2006;118(7 Suppl):81S-98S.
9. Petit JY, Rietjens M, Contesso G, Bertin F, Gilles R. Contralateral mastoplasty for breast reconstruction: a good opportunity for glandular exploration and occult carcinomas diagnosis. Ann Surg Oncol. 1997;4(6):511-5. DOI: http://dx.doi.org/10.1007/bF02303678
10. Spear SL, Carter ME, Schwarz K. Prophylactic mastectomy: indications, options, and reconstructive alternatives. Plast Reconstr Surg. 2005;115(3):891-909. PMID: 15731693 DOI: http://dx.doi.org/10.1097/01.PRS.0000153220.66955.39
11. Coelho GVBF, Nogueira FVM, Silveira Junior VF, Andrade CZN, Carrara HHA, Oliveira HF, et al. Avaliação oncológica após reconstrução mamária com lipoenxertia. Rev Bras Cir Plást. 2014;29(2):243-7.
12. Drinane JJ, Kortes MJ, Bergman RS, Folkers BL. Evaluation of Antibiotic Irrigation Versus Saline Irrigation in Reducing the Long-Term Incidence and Severity of Capsular Contraction After Primary Augmentation Mammoplasty. Ann Plast Surg. 2016;77(1):32-6. DOI: http://dx.doi.org/10.1097/SAP.0000000000000302
13. Cosac OM, Camara Filho JPP, Barros APGSH, Borgatto MS, Esteves BP, Curado DMDC, et al. Reconstruções mamárias: estudo retrospectivo de 10 anos. Rev Bras Cir Plást. 2013;28(1):59-64. DOI: http://dx.doi.org/10.1590/S1983-51752013000100011 NAC: Nipple-Areola Complex.
1. Sociedade Brasileira de Cirurgia Plástica, São Paulo, SP, Brazil
2. Hospital Daher Lago Sul, Brasília, DF, Brazil
3. Centro de Câncer de Brasília, Brasília, DF, Brazil
4. Sociedade Brasileira de Anestesiologia, Rio de Janeiro, RJ, Brazil
Institution: Hospital Daher Lago Sul, Brasília, DF, Brazil.
Corresponding author:
Diogo Borges Pedroso
SEPS 709/909, Centro Médico Julio Adnet, Clínica S-20
Asa Sul, Brasília, DF, Brazil - Zip Code 70390-095
E-mail: dpedroso@gmail.com
Article received: April 10, 2017.
Article accepted: September 23, 2017.
Conflicts of interest: none.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter