

Articles - Year 1999 - Volume 14 -
Botulinum Toxin in Plastic Surgery: Experience and Indications in 1200 Treated Areas
Toxina Botulínica na Cirurgia Plástica: Indicações e Experiência em 1200 Áreas Tratadas
ABSTRACT
The author reports his experience with the use of botulinum toxin for the treatment of facial wrinkles, facial asymmetry and other pathologies in 509 patients, with a total of 1200 areas applied for a period of 32 months. Botox® was injected into muscles responsible for expression and facial wrinkles. The main zones treated were periorbital, frontal, glabellar and cervical. The toxin was also used aiming at improving facial asymmetry caused by nervous injury, blepharospasm, cervical dystonia, masseteric hypertrophy and axillary hyperhidrosis. Pharmacological aspects are reviewed, as well as the infection technique, dose, complications and results.
Keywords: Wrinkles; botulinum toxin; facial asymmetry; facial paralysis.
RESUMO
O autor relata sua experiência com a utilização da toxina botulínica no tratamento das rugas faciais, assimetria facial e outras patologias, em 509 pacientes, perfazendo 1200 áreas aplicadas em um período de 32 meses. O Botox® foi injetado nos músculos responsáveis pelas rugas de expressão da face. As principais zonas tratadas foram a periorbital, frontal, glabelar e cervical. A toxina foi também utilizada visando à melhora da assimetria facial provocada por injúria nervosa, blefaroespasmo, distonia cervical, hipertrofia massetérica e hiperidrose axilar. Aspectos farmacológicos são revistos, assim como a técnica de injeção, dose, complicações e resultados.
Palavras-chave: Rugas; toxina botulínica; assimetria facial; paralisia facial.
Botulism is known since the 18th Century. In 1897, Clostridium botulinum bacteria was found to produce eight different types of toxins. Type A, or Botox®, is a potent neurotoxin. It acts in cholinergic termination zones, blocking acetylcholine at neurimotor plate and leading to muscle paralysis.
As a function of the traction that given muscle groups exert on the skin surface, the formation and stressing of expression wrinkles occur with the course of time.
Thus, an important improvement of expression lines is obtained through the muscle paralysis action obtained with the drug application. This blocking action brings benefits to other conditions where muscle hyperactivity exists.
PHARMACOLOGY
From the eight toxins produced by Clostridium botulinum bacteria (A, B, C1 C2, D, E, F and G), seven are associated to muscle paralysis. Type A botulinum toxin is a neuromuscular joint blocking agent. It promotes the neurotransmitter - acetylcholine - blockade at the presynaptic neuron, leading to muscle paralysis. Type B and F toxins are indicated to cases of type A immunity after repeated injections.
The drug inhibition mechanism comprises three distinct processes. First, there occurs the binding of toxin to presynaptic neuron specific receptors at the neuromuscular joint plane. Following, there occurs the internalization or penetration of the specific receptor.
Finally, there is the inhibition of acetylcholine release.
The effects of striated musculature weakening and paralysis appears around the 2nd or 3rd day after injection, remaining for an approximate period of six months, depending on the dose applied and on each case.
In spite of the deinnervation being permanent, muscle function recovery generally occurs. This is due to the neurogenesis phenomena, with the formation of axonal sprouts, new plates and nervous terminations and the absorption of devitalized neurons(4).
Drug dosage is expressed in international units (IU).
Toxicity is expressed in units, 1 toxin unit (0.4 mg) representing the average lethal dose observed in a group of Webster mice (LD50). Toxic amount for on individual weighting approximately 70 kilograms is believed to be from 2,500 to 3,000 units. Botulinum toxin is free from great side effects. It does not surpass the cerebral barrier and symptoms related to central nervous system do not occur.
INDICATIONS
The first clinical use of toxin is dated 1973 through injection into monkey extraocular muscles. In 1980, Scott started its use in patients bearing cross-eye(7, 8).
Then, it was used for the treatment of blepharospasm(9), paralysis of nystagmus eye rectus muscle(5), hemifacial spasm(11) and facial asymmetry(3).
As from there, a number of clinical indications were encountered, as in cervical dystonia (spasmodic wry neck), oromandibular dystonia, spasmodic dystonia, thrills, cerebral paralysis, masseteric hypertrophy(10), anal fissura, achalasia and others. In 1991, Carruthers showed his experience with the use of the drug in patients bearing glabellar wrinkles(2). In 1996, Matsudo reported his preliminary evaluation of frontoglabellar wrinkles(6). In 1996, Bushara suggested the use of the toxin in axillary hyperhidrosis(1).
CONTRAINDICATIONS
Despite no teratogenicity reports exist,, the employment of the toxin in pregnant or lactant women is not indicated. Neuromuscular alterations are relative contraindications. Patients bearing allergy to human albumin, one of the drug components, shall not use it. Care should be taken specially when there is concomitant use of aminoglucosides, once its toxic action is potentialized.
MATERIAL AND METHODS
Type A botulinum toxin - Botox®, stored under refrigeration was reconstituted into 4 ml of 0.9% physiological saline without preservative, in a concentration of 25 U/ml.
During 32 months, 509 patients bearing wrinkles localized at frontal, glabellar, orbital and cervical regions, as well as those bearing facial asymmetry and other pathologies, were selected for treatment, with a total of 1,200 applied areas. The photographic documentation followed a strict distance and luminosity pattern.
Photos were taken before and between three and six month after application.
The regions treated were the frontal, glabellar, periorbital, cervical, axillary and masseteric ones, besides nasogenian furrow. The main muscles treated were the frontal, corrugator, procerus, eyelid orbicular, platysma, masseter, sternocleidomastoid ones and, in case of facial asymmetry, the major and minor zygomaticus and the risorius ones (Table I). The injections were applied with the patient seated, without anesthesia, using insulin-type syringe with fine needle (30 grams). After passing through the skin and reaching the muscle, the needle was pulled approximately 1 millimeter and the solution injected. Local manual compression followed. Recommendations post-treatment were reduced to exercising facial mirnics within the first hours, remaining in orthostatic or seated position and not handling the region.
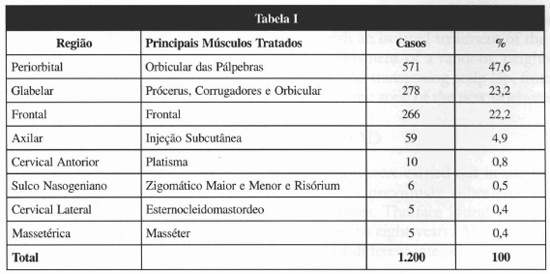
RESULTS
During 32 months 509 patients were treated with a total of 1200 applied areas. A high muscle paralysis degree was reached with the conseguent improvement of symptoms in practical1y al1 cases (Figs. 1 to 4). Only 17 patients needed reapplication before a four month period (3.3%). The effects were noticed within 24 - 48 hours after injection, being enhanced in up to seven or ten days and remaining stable for approximately four to six months. Patients bearing facial asymmetry had a more transient result (three months), probably due to the fact that the dose used had been lower, as well as in the case of blepharospasm (Figs. 5 and 6).
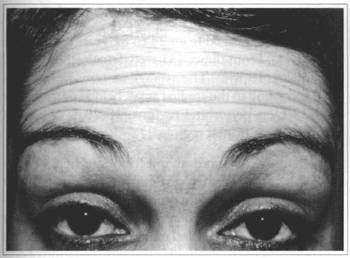
Fig. la - 43 y. o. parient with wrinkles at frontal region.
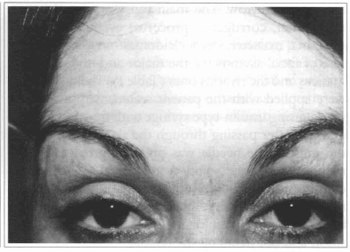
Fig. 1b - Five months after treatment.
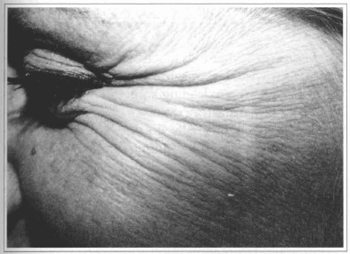
Fig. 2a - 37 year-old patient with wrinkles at periorbital region ("crowfoot").
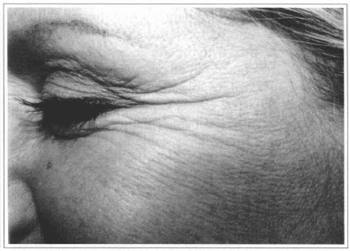
Fig. 2b - Five months after treatment.
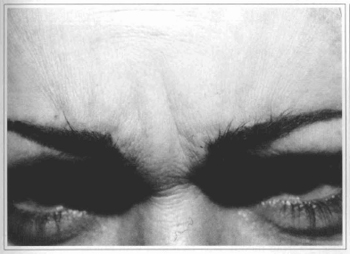
Fig. 3a - 36 year-old patient with wrinkles at glabellar region.
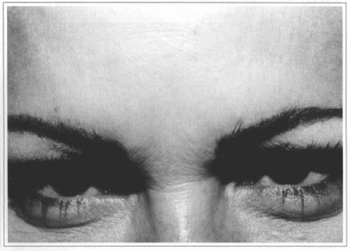
Fig. 3b - Four months after treatment.
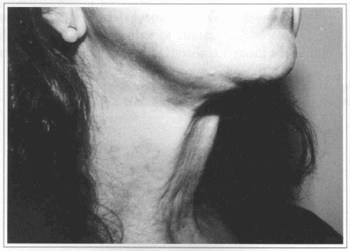
Fig. 4a - 52 year-old patient with platysma bands at cervical region.
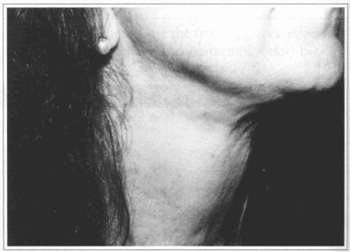
Fig. 4b - Four months after treatment with Botox.

Fig. 5a - Patient with facial paralysis of traumatic origin, mouth deviation and asymmetry of nasogenian furrow.
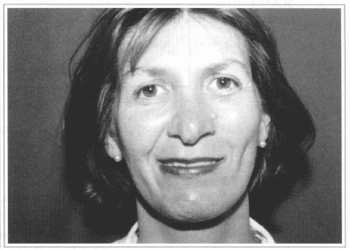
Fig. 5b - Three months after application into zygomaticus and risorius muscles.
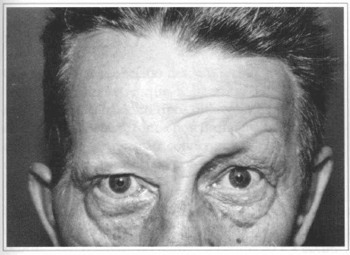
Fig. 6a - 50 year-old patient with congenital facial paralysis.
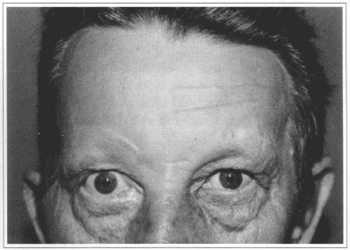
Fig. 6b - Five months after treatment with Botox.
There was, however, an important improvement in the harmony and balance between the two sides of the face. At periorbital region - the most applied area - there was a significant increase of side portion of eyebrow, providing a more jovial aspect to the patient.
Patient's age ranged from 24 to 81 years old (average of 49.44). From these, 431 were females(84.7%) and 78 males (15.3%). The average amount injected varied according to the applied areas, being approximately 20-25 U at periorbital regions, 15-20 U at glabella and 15 U at frontal region.
Ecchymosis occurred at injection site in four cases (0.8%). Short-term cephalea was reported by four patients (0.8%). Major complications related to weakening of muscle next to applied areas by migration or accidental injection. Unilateral eyelid ptosis occurred in one patient (0.2%) with a discrete deviation of ocular globe that persisted for approximately ten days, resolving spontaneously.
DISCUSSION
Several are the methods described aiming at attenuating facial wrinkles. Surgical intervention is required for many of them, leading some times to delayed recovery, non-satisfactory results or scars. Injectable synthetic products general1y do not have a definite result, are expensive, may migrate and some of them require previous testing before use. Fatty graft does not give good results concerning periorbital wrinkles, and its results are transient, besides requiring a donor site.
Botulinum toxin application showed to be a simple, non-surgical, safe and efficient method for the treatment of facial wrinkles, mainly in its lower third. It presents few complications, is reversible, may be reapplied several times and is easy to be obtained.
Some classical surgeries, such as the corrogator muscle excision, may be satisfactorily substituted by the repeated application of this substance. Selected cases of facial asymmetry caused by iatrogenic lesion or other etiologies also get good results with this drug. Transient paralysis of frontal region post ritidoplasty may be properly controlled with counterlateral small injections of the toxin till recovery of injured side, exceedingly decreasing patient and surgeon's anxiety. This non-surgical option shows important advantages for bearers of masseteric hypertrophy and axillary hyperhidrosis in relation to traditional treatments.
CONCLUSION
The use of botulinum toxin is adjoining traditional methods and represents a safe and effective non-surgical option for the treatment of facial expression wrinkles and in selected cases of facial asymmetry and finds more and more important indications in medical practice.
REFERENCES
1. BUSHARA KO et al. Botulinum toxin - a possible new treatment for axillary hyperhidrosis.
Clinical Ex. Dermatol. 1996;21:276-278.
2. CARRUTERS J, CARRUTERS JA. Botulinum toxin use for glabellar wrinkles. IN: Annual Meeting of the American Society for Dermatologic Surgery, 1991, Orlando. Annals... p. 13-17.
3. CLARK RP, BERRIS CE. Botulinum toxin: A treatment for facial asymmetry caused by facial nerve paralysis. Plast. Reconstr Surg. 1989; 84:353.
4. HASSAN SM et alo Elimination of superfluous neuromuscular junctions in rat calf muscles recovering from botulinum toxin-induced paralysis. Muscle Nerve. 1994; 17:623-631.
5. HELVESTON EM. Treatment of acquired nystagmus with botulinum toxin. Am. J. Ophtalmol. 1988;106:584-586.
6. MATSUDO PKR. Botulinum toxin for correction of fronto-glabella wrinkles: preliminary evaluation. Aesth. Plast. Surg. 1996; 20:439-441.
7. SCOIT AB et aloPharmacologic weakening of extraocular muscles. Invest. Oftalmol. Soc. 1973; 12:924-927.
8. SCOTT AB. Botulinum toxin injection into extraocular muscles as an alternative to strabismus surgery. Ophtalmology. 1980;87: 1044-1049.
9. SHORR N et al. The use of botulinum toxin in blepharospasm. Am. J. Ophtalmol. 1985; 99:542-546.
10. SMYTH AG. Botulinum toxin treatment of bilateral masseteric hypertrophy. Br. J. Oral Maxillofac. Surg. 1994; 32:29-33.
11. SAVINO PJ et al. Hemifacial spasm treated with botulinum injection. Arch. Ophtalmology. 1985; 103:1305-1306.
12. TSUI JK et al. A pilot study on the use of botulinum toxin in spasmodic torcicollis. Can. J. Neuro. Sci. 1985; 12:314-316.
I - Senior Member of the Brazilian Society of Plastic Surgery.
Address for correspondence:
Alberto Goldman, MD
R. Quintino Bocaiúva, 683 conj. 203
Porto Alegre - RS
90440-051
Phone: (55-51) 331-7377
e-mail: goldman@pro.via-rs.com.br


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter