

Original Article - Year 2018 - Volume 33 -
Breast reconstruction with a latissimus dorsi myocutaneous flap and implants: evaluation of strategies adopted to improve the results and evaluation of satisfaction by Breast-Q
Reconstrução mamária com o retalho miocutâneo do grande dorsal e implantes: avaliação de estratégias adotadas para melhoria dos resultados e avaliação de satisfação pelo Breast-Q
ABSTRACT
Introduction: The paper describes a 14-year experience with
breast reconstruction using a latissimus dorsi myocutaneous
flap (LDMF) and breast implants. The objective was to
delineate the experience with LDMF and breast implants, skin
island schedules and dissection details, flap transposition and
placement of the implant under a double layer, strategies to
minimize damage in the donor area, and strategies of breast
symmetry and reconstruction of the nipple-areola complex, in
association with evaluation using the Breast-Q questionnaire.
Methods: A review of medical records was performed between
April 2003 and June 2017.
Results: In the period, 76 patients
with a mean age of 50.09 years underwent reconstruction with
a LDMF, which was bilateral in 11, for the right breast in 34, for
the left breast in 31, immediate in 41, late in 22, and for rescue
in 13.
Conclusion: We conclude and verified with the Breast-Q
questionnaire that with a precise indication, the proposed
reconstruction technique with a double subpectoral plane and
coverage with a LDMF is safer with a lower complication rate.
Keywords: Breast neoplasms; Surveys and questionnaires; Myocutaneous flap; Treatment outcome; Breast implants
RESUMO
Introdução: O trabalho descreve 14 anos em reconstrução mamária com o retalho miocutâneo
do grande dorsal (RGD) e implantes mamários. O objetivo é delinear
experiência com o RGD e implantes mamários, programações das ilhas de pele e
detalhes da dissecção, transposição de retalho e colocação do implante sob
dupla camada muscular, estratégias para minimizar os danos na área doadora,
estratégias de simetrização mamária e reconstrução do complexo
areolopapilar, associado a avaliação do questionário
Breast-Q.
Métodos: Foi realizada revisão de prontuários entre abril de 2003 a junho de 2017.
Resultados: No período 76 pacientes foram reconstruídas com o RGD, com idade média de
50,09 anos, sendo 11 bilaterais, 34 mama direita e 31 mama esquerda. 41
imediatas, 22 tardias e 13 de resgate. A média de satisfação foi 72,36% por
meio do Breast-Q.
Conclusão: Concluímos e comprovamos com o questionário do Breast-Q que
a indicação precisa aliada a técnica de reconstrução proposta com plano
duplo subpeitoral associada a cobertura do RDG, propicia uma loja mais
segura, diminuído o índice de rippling.
Palavras-chave: Neoplasias da mama; Inquéritos e questionários; Retalho miocutâneo; Resultado do tratamento; Implantes de mamas
INTRODUCTION
Breast cancer is the most common type of cancer among women worldwide, including Brazil, after non-melanoma skin cancer, accounting for approximately 28% of new cases each year. Breast cancer also affects men, though rarely, representing only 1% of the total number of cases. It is relatively rare before the age of 35 years, but at older ages, its incidence increases progressively, especially after 50 years. Data indicate an increase in incidence in both developed and developing countries.
Breast cancer is of several types. Some types progress quickly and even devastatingly, while others do not. Most cases have a good prognosis. The estimated number of new cases is 57,960 (2016 INCA), while the number of deaths is 14,388, of which 181 occur in men and 14,206 occur in women (2013 SIM)1. Mastectomy is often used as treatment and prophylaxis for malignant breast cancer and can save the lives of women with breast cancer. However, the loss of the breast can cause psychological and psychosocial trauma. Breast reconstruction is an important step in post-mastectomy recovery, which may help patients recover their sense of femininity2. To do so, the degree of satisfaction of patients who have undergone breast reconstruction must be evaluated.
Within the context of private medicine (in which this study was developed), the early detection rate of breast cancer has increased, which has led to a higher number of professionals and teams requiring to offer a greater number of breast reconstructions. The multidisciplinary interaction and cooperation between the medical specialties (mastology, plastic surgery, oncology, radiotherapy (RT), physiotherapy, etc.) can alleviate the suffering of patients by using more-efficient treatments and techniques.
Of the several existing techniques for breast reconstruction, which range from techniques using local flaps such as the plug flap3 and mammoplasty techniques using neighborhood flaps such as the thoracodorsal flap4 to the use of alloplastic materials (expanders and prostheses) and numerous autologous flaps5, combined techniques have also been used. We consider that no procedure is superior to another in all issues; however, patients benefit from a judicious choice5 based on the knowledge of the surgeon, the patient’s wishes, and, especially, the indications/contraindications of each method.
Although it does not provide a large volume to the new breast when used in isolation, the latissimus dorsi muscle flap (LDMF), which was described by Tansini6 and modified by Bostwick et al.7 in 1978, ensures good results when associated with a breast implant. Owing to its versatility and safety, coupled with a short learning curve, the LDMF is often a salvage alternative if the initial reconstruction fails5.
However, in some cases, initial reconstruction using a LDMF is appropriate for the following indications: absence of an abdominal donor area for possible reconstruction with a transverse rectus abdominis muscle (TRAM) flap, risk factors that contraindicate the use of other flaps, impossibility of reconstructions with local flaps, lack of tissue for reconstructions with alloplastic materials (prostheses and expanders), and acceptance by the patients8.
With the popularization of breast reconstructions and with a better knowledge on the part of patients, expectations from breast reconstructions have changed, not only as reconstructive procedures but also as aesthetic procedures, thereby requiring more harmonious results9. Thus, in cases of unilateral or even bilateral reconstruction with the most varied techniques, symmetrization becomes an essential step. Although several studies have compared the different breast reconstruction techniques, few studies have evaluated the factors that influence breast symmetry after reconstruction10.
OBJECTIVE
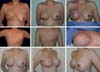
The objective was to outline our 14-year experience with breast reconstruction with an LDMF and implants, programming of skin islands, dissection details, transposition of the flap, and implant placement under a double muscle layer, in addition to strategies to minimize the damage in the donor area. The study also details the strategies of breast symmetrization and reconstruction of the nipple-areola complex (NAC) together with an assessment of patient satisfaction through the Breast-Q questionnaire.
METHODS
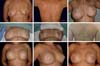
All 76 patients who underwent breast reconstruction with a LDMF signed an informed consent form before surgery and were operated by the same senior surgeon with the same routine. Medical charts and preoperative and postoperative photographs were reviewed, and the Breast-Q questionnaire was administered from April 2003 to June 2017 at the author’s (J.D.L.G.A.) private clinic in Brasília, DF.
The Breast-Q questionnaire was administered in all the patients who underwent breast reconstruction with a LDMF after mastectomy for breast cancer. The Breast-Q was applied from the SurveyMonkey system, which is the most popular online survey software in the world. This platform makes it easy to create polls and research surveys that would help to better manage the critical aspects of a business, such as customer satisfaction and employee involvement.
The exclusion criteria were patients who knowingly had an indication for post-reconstruction RT and therefore were not treated with the LDMF technique, and patients who refused to receive myocutaneous flaps.
RESULTS
From April 2003 to June 2017, 76 patients were evaluated in accordance with the following data:
Epidemiological data: Age, type of cancer, and laterality. Bilateral, right, and left reconstructions were performed in 11, 34, and 31 patients, respectively. The mean age at the moment of reconstruction was 50.09 years. Of the patients, 23 were aged <45 years. Biopsy revealed invasive ductal carcinoma in 29 patients, invasive lobular carcinoma in 5 patients, and invasive papillary carcinoma in 1 patient. Forty-one patients did not have pathology records in their medical charts.
Forty-one reconstructions were immediate, 22 were late, and 13 were salvage reconstructions.
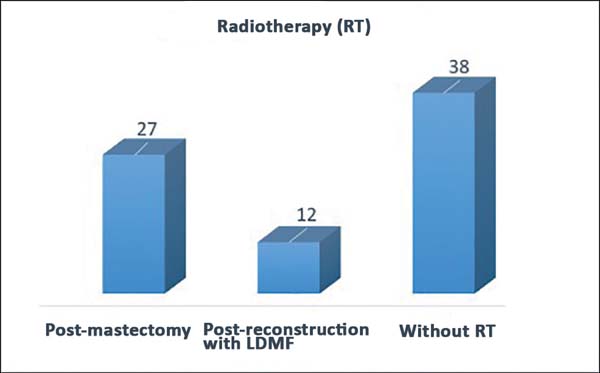
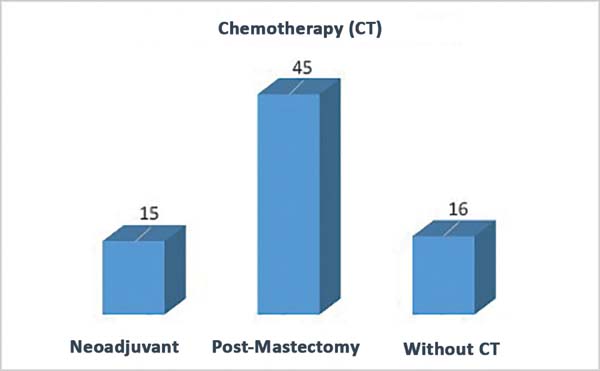
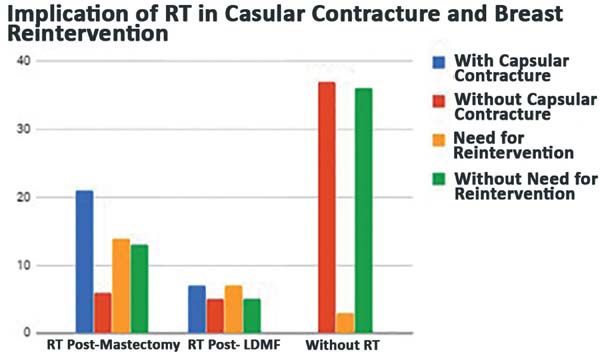
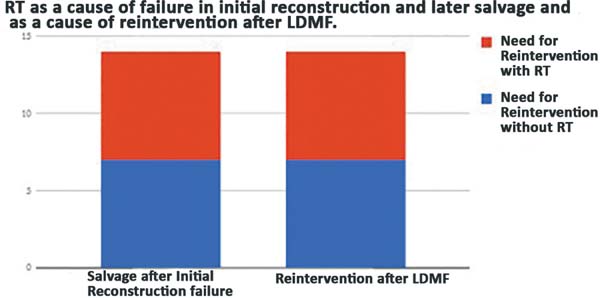
Completion of chemotherapy and/or RT: Twenty-seven patients received RT after mastectomy (late and salvage reconstructions); 12 had a reconstruction with a LDMF, even if they had no indication in the preoperative period; and 38 did not receive RT (Figure 1). Fifteen patients underwent neoadjuvant chemotherapy after mastectomy, 45 had a mastectomy, and 16 did not receive chemotherapy (Figure 2). A direct relationship was found between the patients who had RT and those who had to undergo salvage reconstruction with a LDMF or reintervention for the breasts (Figure 3).
- Intervals between the first, second, and third reconstructions: The
mean time was 8.6 months between the first and second surgeries and
11 months between the second and third surgeries. Thirty-five
patients completed the reconstruction in 2 stages; 12, 3 stages; and
28, only 1 stage and refused to have the second/third stage.
- Types of implants: Textured implants were used in 61 patients;
polyurethane implants, 11 patients; permanent textured two-chambered
expanders, 2; and no implants, 2.
- Plan for the inclusion of implants: Seventy patients received an
implant with a LDMF in the double plane (under the greater pectoral
and latissimus dorsi muscles). In the symmetrization of the opposite
breast, 49 patients had an implant in the retropectoral plane, 15
did not undergo symmetrization, 2 underwent mastopexy without
prosthesis, and 3 had an implant in the subglandular plane.
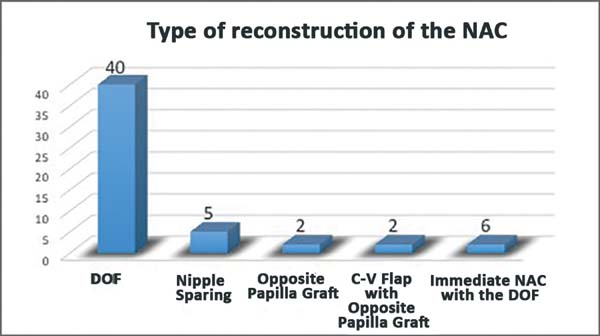
- Type of reconstruction of the NAC: 51 NAC reconstructions were
performed in the 76 patients, 40 double opposing flap (DOF), 5
nipple sparing, 2 opposite papilla graft, 2 C-V flap with graft of
the opposite papilla, 6 immediate NAC with the DOF (Figure 4).
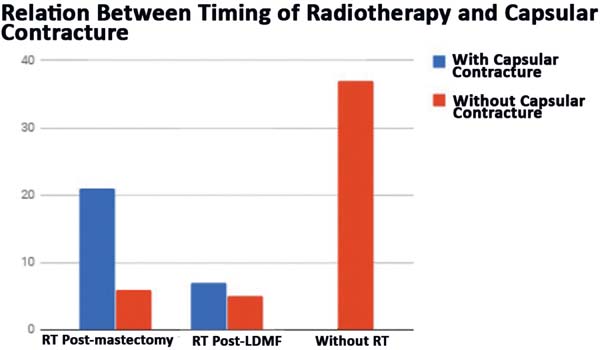
- Intercurrent and complication rates: Of the 12 patients who
underwent post-reconstruction RT, even if not foreseen/programmed in
the preoperative period, 7 developed a capsular contracture
requiring reintervention (Figure 5).
Loss of implant by infection and extrusion occurred in 2 patients and minor complications in 14 patients (seroma, small dehiscence, partial necrosis of the mastectomy flap, early ptosis of the implant, and other less important complications). In 24 patients, reintervention for loss of implant, or capsular contracture or ptosis of the implant was not necessary (Figure 6).
- With regard to the assessment of the degree of satisfaction of
patients after reconstruction, 23 patients answered the Portuguese
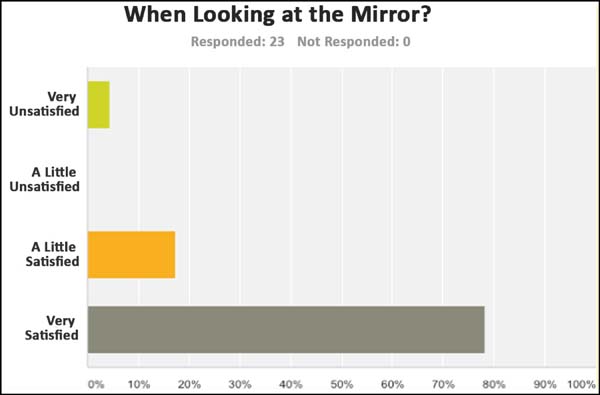
version of the Breast-Q questionnaire. Of these
patients, 78.26% reported being highly satisfied when looking in the
mirror (Figure 7). With regard
to the shape of the reconstructed breast, 69.57% of the patients
reported being highly satisfied, and in relation to the size of the
breast, this rate was 81.82%. Regarding symmetrization, 72.73% of
patients were satisfied. In terms of the appearance of the
reconstructed breast in comparison with the appearance before having
any breast surgery, 63.64% of patients reported being highly
satisfied and 95.65% of the patients thought that having a
reconstructed breast was much better than not having a breast, and
the same percentage (95.65%) of patients would like to encourage
other women to have breast reconstruction surgery. None of the
patients regretted having undergone breast reconstruction. With
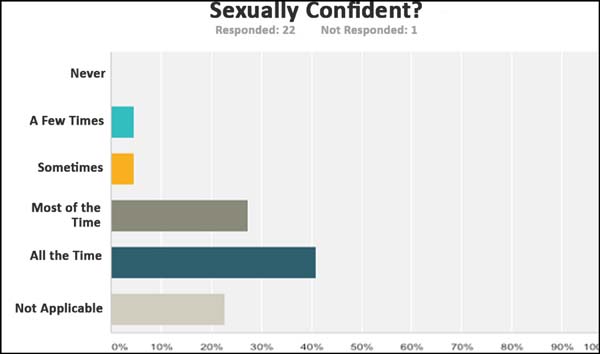
respect to self-confidence, 60.87% reported having self-confidence
in the social environment at all times and 40.91% reported being
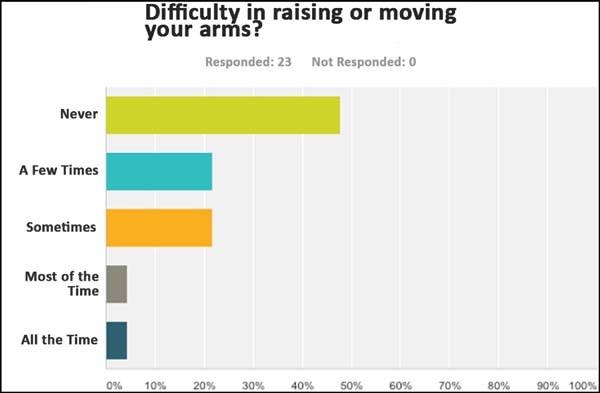
sexually confident at all times (Figure 8). Of the patients, 47.83% responded not having
any difficulty in raising or moving the arms (Figure 9), 50% stated not having any
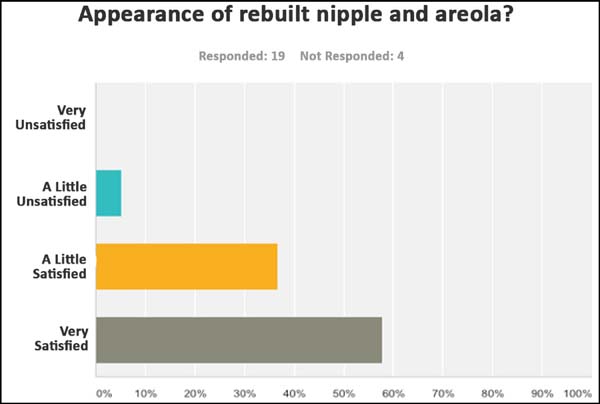
sensitivity, 65% were highly satisfied with the appearance of the
nipple; and 57.89% claimed to be highly satisfied with the
appearance of the reconstructed nipple and areola (Figure 10). Overall, 72.36% of
patients assessed with the Breast Q questionnaire
were highly satisfied.
DISCUSSION
Immediate breast reconstruction with the latissimus dorsi muscle was performed in most cases (53.94%), while the demand for late reconstruction by patients was 28.94%. What guided the indication for the LDMF technique was foremost the impossibility of using an expander, which is the first choice of treatment, because it is less invasive; the disagreement of patients to undergo reconstruction with tissue expanders; and the fact that they were stage 1 or 2 tumors with low probability of post-reconstruction RT with a LDMF (Figure 11).
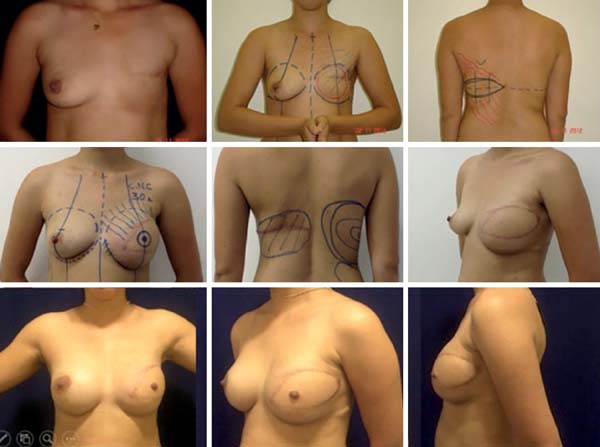
The idea of these concepts goes back >10 years according to the original publication of Di Lamartine et al.11 and the observance of other concepts equally favorable to a good breast reconstruction, such as the use of lipofilling, the tactics of NAC reconstruction, and the strategies for breast symmetrization (Figure 12).
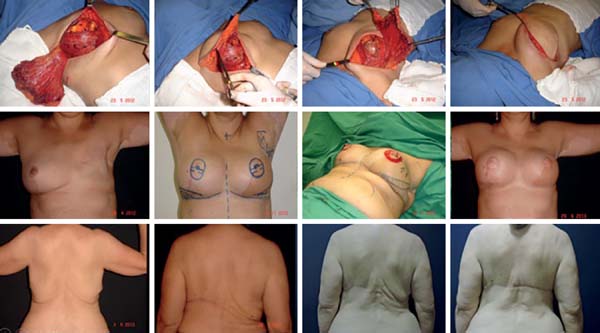
With the progression in early diagnosis and the use of irradiation with less tissue damage, the possibilities of smaller reconstructions with the tissues of the breast itself has also increased, with less indications for surgeries with distal flaps. Without doubt, this is the best prospect in treating patients with early diagnosis or in cases in which the patient has no indications of risk-reducing adenomastectomy
The island plug flap3, with its outstanding versatility, provides good filling of defects generated by the oncological treatment in the upper internal, upper external, and central portions. The ideal donor for island flaps is defined for two reasons. The first is the presence of several perforators that are found from the fourth to the sixth intercostal spaces. The second is that closure of the donor area in this area allows modelling of the breast as if it were a conventional mastoplasty3.
Classically, the relevant distal flaps are the TRAM, which was described by Drever12 in 1977 and modified by Hartrampf et al.13 and Gandolfo14 in 1982, and LDMF, which was described by Tansini15 and modified by Bostwick et al.16 in 1978. The LDMF is often associated with silicone prostheses or expanders to increase the volume and shape the mammary cone. The use of permanent bicompartmental expanders is more recent and has been systematized and diffused by Becker17.
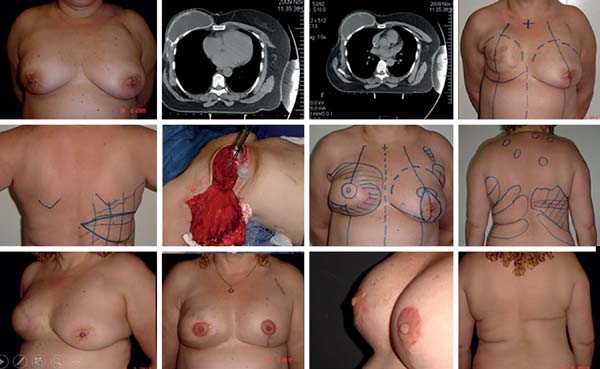
Following the criteria adopted worldwide, contraindicating immediate reconstruction is now scientifically intolerable and contradicts the already preconceived concept of patients that immediate reconstruction is always possible. Thus, the surgeon assumes high rates of capsular contracture when indicating immediate reconstruction, which can reach 68% according to McCarthy et al.17. One encouragement to the surgeon is that this same study shows that the rate of capsular contracture in breast reconstructions with alloplastic material, even without postoperative RT, can reach 40%. The final success rates of the reconstructions were 90% with RT and 99% without RT.The use of radiation on the myocutaneous flap is also associated with capsular contracture, burns, and radiodermatitis, being the most complex surgical treatment with a high possibility of unsatisfactory results. In addition to these complications, hypoesthesia, pain, and lymphedema are described in the literature18. This thought has always guided our indications in the choice of techniques in breast reconstruction so that at the slightest hint of RT, the reconstruction was attempted only with expanders when possible, leaving the LDMF intact and available for a possible failure of the immediate reconstruction because of the potential damage caused by RT on the reconstructions (Figure 13).
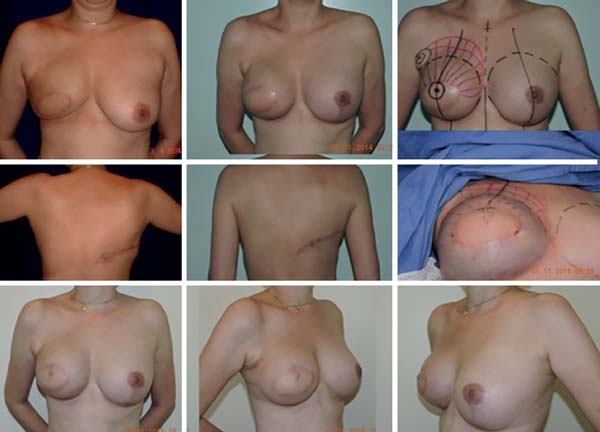
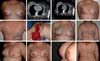
The screening of sentinel lymph nodes has a key role when we maintain conservative surgical treatment in breast cancer. Approximately 25% of patients submitted to lymphatic mapping will present some lymph node involvement, which may imply complementation of treatment with RT. According to the American Society of Clinical Oncologists Guidelines, all cases with ≥4 positive lymph nodes, tumors >5 cm in size with any positive lymph nodes (T3), and all operable tumors in stage 3 have an indication for RT after mastectomy. This will definitely influence the reconstruction strategy and may even postpone reconstruction in specific cases because of tumor or patient factors, together with the undoubtedly deleterious action of RT on implants/reconstructions19.
Skin- or nipple-sparing mastectomy has been more frequently used as the disease is diagnosed in earlier stages. It is known that however effective mastectomy with skin preservation is, approximately 9.5%20 of breast tissue will remain; therefore, it is important to have an appropriate preoperative planning with mastologists and plastic surgeons whenever possible to keep the minimum tissue for maintaining the viability of flaps for the reconstruction and incisions inside an imaginary “T” as described by Pitanguy21 (Figure 14).
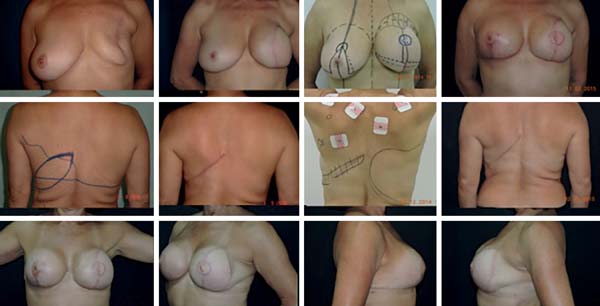
In a prospective study, Sbitany et al.22 compared immediate reconstruction with expanders/implants in patients who underwent or did not undergo RT (pre- or post-mastectomy). A higher rate of complications was observed in the irradiated patients, such as infections, wound dehiscence, skin necrosis, exposure, and loss of implant, with an incidence rate of ≥19.2% as compared with the other group. Other studies have shown that complications after RT can reach 40%. This is due to the lack of complacency of the irradiated tissue, besides damage in vascularization; the higher tendency to have capsular contracture (present in up to 60% of cases); and the higher incidence rates of liponecrosis, lipoatrophies, and radiodermatitis.
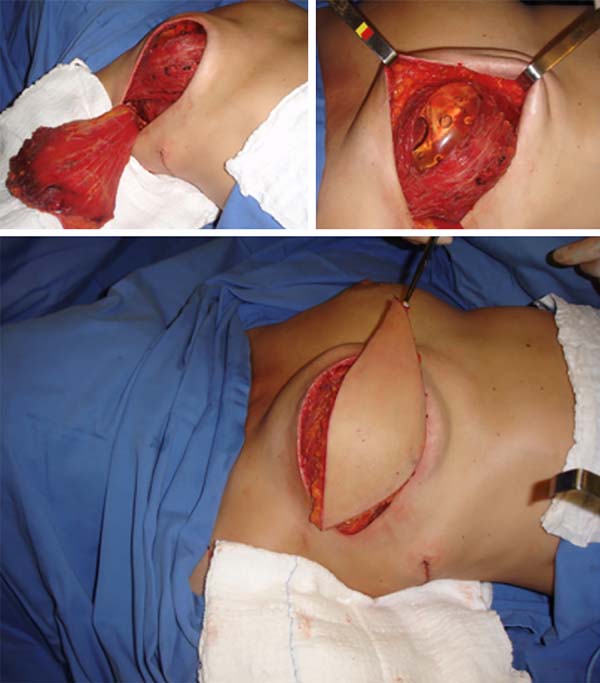
In cases in which the use of a TRAM flap is contraindicated or refused by the patient, the LDMF can be a solution. Although reliable and versatile, its projection is generally inadequate, requiring an implant or expander (in most cases). It is a safe flap with a robust vascular pedicle. For this reason, it may be indicated in patients with major risk factors, which contraindicate other types of autologous flaps, and is often used in salvage reconstructions.
In patients with a history of RT or axillary emptying, assessment should be performed with imaging tests (Doppler) to verify the patency of the thoracodorsal pedicle. Ideally, a horizontal skin spindle should be planned where the resulting scar will be in the bra line. However, in obese patients, the oblique design is preferable in older patients and those requiring large skin islands.
Symmetrization, reconstruction of the NAC, refinements, and complications
In our view, breast symmetrization is an essential step for an adequate breast reconstruction.
Given the most diverse breast shapes and volumes to be treated, the reasoning of the reconstruction surgeon should be sharpened to the point that in the initial assessment, the size, shape, and volume of the breasts that can be achieved are already envisioned. In the same way, we think that the reconstructed breast will have a skin envelope and texture determined by the type of reconstruction adopted.
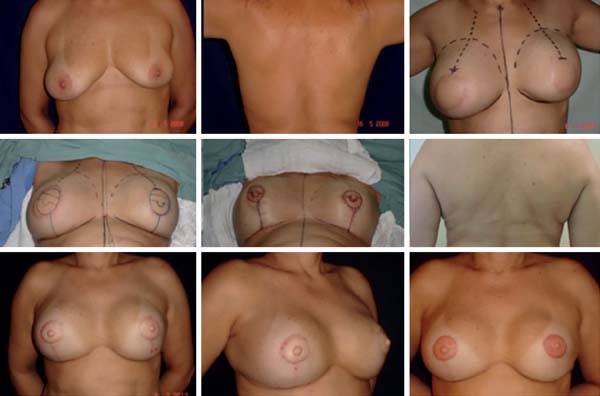
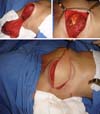
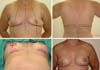
Most of the times, with the exception of reconstructions with the TRAM flap, the coverage that is attained with the implant in cases of reconstruction with implants, even when using the LDMF, is thin, with the possibility of rippling in the late postoperative period. In the strategy that we adopted by placing the implant under a double muscle layer (Figure 15), besides defining a tighter pocket to place the implant (textured in most times) with lesser possibilities of implant dystopia, we have the advantage of preventing rippling. In the second and third moments, fat grafting is still used with the same purpose if necessary.
Symmetrization entails tissue reduction aimed, foremost, toward a similarity of implant coverage between the breasts and to reduce the gland volume in the opposite breast with benefits in the prevention against cancer in the opposite breast.
This strategy to remove 60–70% of the volume of the opposite breast, associated with the inclusion of the implant in retropectoral position, has been quite effective in our previous study (Figure 16).
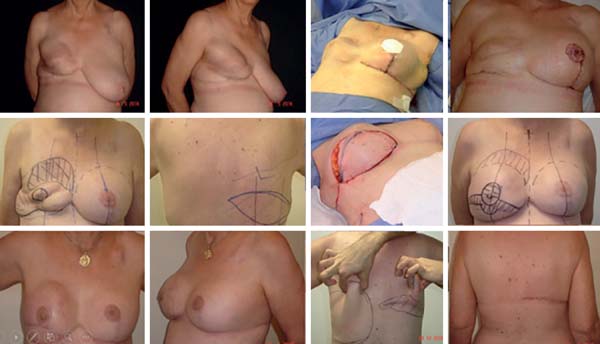
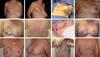
After reconstruction and symmetrization, the new NAC becomes the final touch of the overall reconstruction. With several available NAC reconstruction techniques/strategies to choose, the choice of a few that can be mastered and used with all knowledge is highly important. In our sample, in most cases, we used the DOF, as we found this technique to best mimic the opposite NAC because in addition to providing a shaped and projected papilla that is long-lasting, it creates a raised areola in relation to the neighboring tissues, similar to the anatomy of a natural NAC. One strategy we adopted was to design the flap in an oval/elliptical shape, as in the end, we achieved a rounded NAC, which was not the case with the original design, as the NAC was not actually circular and the nipple was not exactly in the middle of the areola23 (Figure 17).
There were cases in which we performed unilateral or bilateral reconstruction, and already reconstructed the NAC in the first surgical moment because the situation was favorable (Figure 18). The presence of scars and RT do not impede the use of the DOF, as this is a safe and predictable flap given its sessile base23.
During symmetrization and then at the time of reconstruction of the NAC, it is also appropriate for the necessary refinements in cases with asymmetry of the positioning and volume of the implants, capsular contractures, inadequate skin excesses; inadequate scars, thin flaps, and coverage in which lipofilling provides an appropriate solution (Figure 18).
The complications observed that required the removal of the implant or reintervention were related to the infection and exposure of the implant, residual skin necrosis of the mastectomy, and implant dystopia. In cases of loss of implants, their replacement was scheduled after an adequate period of not less than 6 months. In cases of implant dystopia or need of skin adjustments, repair was performed after the reconstruction (Figure 19).
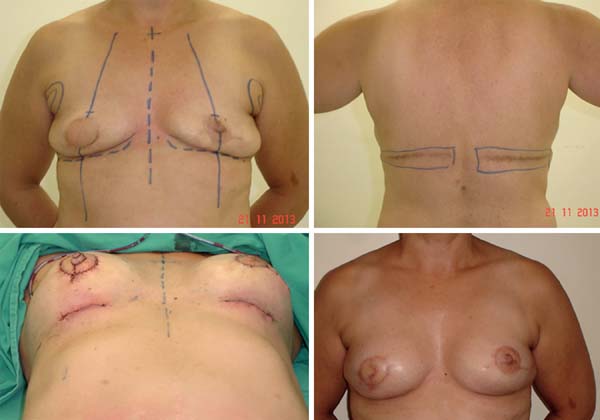
CONCLUSION
We conclude and proved with the Breast-Q questionnaire that a precise indication, together with the reconstruction technique with implants placed under a double muscle layer (pectoralis major and LDMF), provides a safer and stable pocket and decreases the rippling index. Symmetrization with subtotal adenomastectomy in patients with breast cancer presents highly satisfactory results, as it involves a cytoreduction of the opposite breast and provides breasts with better symmetry in terms of volume, texture, projection, and long-term outcome. DOF is an excellent technique that reconstructs NACs with favorable results that are maintained in the long term.
COLLABORATIONS
|
JDLGA |
Analysis and/or interpretation of data; statistical analysis; final approval of the manuscript; funding; data collection; conceptualization; conception and design of the study; project management; research; methodology; writing, including review and editing; and research supervision. |
|
JCD |
Resource management; project management; methodology; writing, including revision and editing; and research supervision. |
|
LMCD |
Data collection, conceptualization, and research; writing, including preparation, revision, and editing of the original manuscript. |
|
JGOJ |
Analysis and/or interpretation of data; statistical analysis; data collection; research; methodology; completion of operations and/or experiments; writing, including preparation, revision, and editing of the original manuscript. |
|
GCS |
Analysis and/or interpretation of data; statistical analysis; final approval of the manuscript; data collection; research; completion of operations and/or experiments; writing, including preparation, revision, and editing of the original manuscript. |
|
ACC |
Analysis and/or interpretation of data; data collection; completion of operations and/or experiments; writing, including preparation, revision, and editing of the original manuscript. |
|
RCSD |
Analysis and/or interpretation of data; statistical analysis; data collection; writing, including preparation, revision, and editing of the original manuscript. |
REFERENCES
1. Brasil. Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Estimativa 2016: Incidência de Câncer no Brasil. Rio de Janeiro: INCA; 2015.
2. Colakoglu S, Khansa I, Curtis MS, Yueh JH, Ogunleye A, Haewyon C, et al. Impact of complications on patient satisfaction in breast reconstruction. Plast Reconstr Surg. 2011 Apr;127(4):1428-36.
3. Daher JC. Breast island flaps. Ann Plast Surg. 1993;30(3):217-23.
4. Holmström H, Lossing C. The lateral thoracodorsal flap in breast reconstruction. Plast Reconstr Surg. 1986;77(6):933-43.
5. Serletti JM, Fosnot J, Nelson JA, Disa JJ, Bucky LP. Breast reconstruction after breast cancer. Plast Reconstr Surg. 2011;127(6):124e-35e.
6. Tansini I. Sopra il mio nuovo processor di amputazione della mammella. (coverage of the anterior chest wall following mastectomy). Guz Mal Ital. 1906;57:141.
7. Bostwick J 3rd, Vasconez LO, Jurkiewicz MJ. Breast reconstruction after a radical mastectomy. Plast Reconstr Surg. 1978;61(5):682-93.
8. Di Lamartine J, Galdino Júnior J, Daher JC, Guimarães GS, Camara Filho JPP, Borgatto MS, et al. Reconstrução mamária com retalho do músculo grande dorsal e materiais aloplásticos: análise de resultados e proposta de nova tática para cobertura do implante. Rev Bras Cir Plást. 2012;27(1):58-66.
9. Hamdi M, Casaer B, Andrades P, Thiessen F, Dancey A, D'Arpa S, et al. Salvage (tertiary) breast reconstruction after implant failure. J Plast Reconstr Aesthet Surg. 2011;64(3):353-9.
10. Leone MS, Priano V, Franchelli S, Puggioni V, Merlo DF, Mannucci M, et al. Factors affecting symmetrization of the contralateral breast: a 7-year unilateral postmastectomy breast reconstruction experience. Aesthetic Plast Surg. 2011;35(4):446-51.
11. Di Lamartine J, Galdino Júnior J, Daher JC, Guimarães GS, Camara Filho JPP, Borgatto MS, et al. Reconstrução mamária com retalho do músculo grande dorsal e materiais aloplásticos: análise de resultados e proposta de nova tática para cobertura do implante. Rev Bras Cir Plást. 2012;27(1):58-66.
12. Drever JM. Total breast reconstruction with either of two abdominal flaps. Plast Reconstr Surg. 1977;59(2):185-90.
13. Hartrampf CR, Scheflan M, Black PW. Breast reconstruction with a transverse abdominal island flap. Plast Reconstr Surg. 1982;69(2):216-25.
14. Gandolfo EA. Breast reconstruction with a lower abdominal myocutaneous flap. Br J Plast Surg. 1982;35(4):452-7.
15. Tansini I. Sopra il mio nuovo processor di amputazione della mammella. (coverage of the anterior chest wall following mastectomy). Guz Mal Ital. 1906;57:141.
16. Bostwick J 3rd, Vasconez LO, Jurkiewicz MJ. Breast reconstruction after a radical mastectomy. Plast Reconstr Surg. 1978;61(5):682-93.
17. Becker H. One stage immediate breast reconstruction with adjustable implants. In: Spear SL, ed. Surgery of the breast: principles and art. 2nd ed. Philadelphia: Lippincott Williams & Wilkins; 2006. p.438-50.
18. Cordeiro PG, Pusic AL, Disa JJ, McCormick B, VanZee K. Irradiation after immediate tissue expander/implant breast reconstruction: outcomes, complications, aesthetic results, and satisfaction among 156 patients. Plast Reconstr Surg. 2004;113(3):877-81.
19. Pedron M, Alves MR, Menk C. Sistematização em mamoplastia oncológica. Rev Soc Bras Cir Plást. 2001;16(3):54-60. Bostwick J 3rd, Paletta C, Hartrampf CR. Conservative treatment for breast cancer. Complications requiring reconstructive surgery. Ann Surg. 1986;203(5):481-90.
20. Torresan RZ, Cabello dos Santos C, Brenelli H, Okamura H, Alvarenga M. Residual glandular tissue after skin-sparing mastectomies. Breast J. 2005;11(5):374-5.
21. Pitanguy I. Surgical treatment of breast hypertrophy. Br J Plast Surg. 1967;20(1):78-85.
22. Sbitany H, Mirzabeigi MN, Kovach SJ, Wu LC, Serletti JM. Strategies for recognizing and managing intraoperative venous congestion in abdominally based autologous breast reconstruction. Plast Reconstr Surg. 2012;129(4):809-15.
23. Di Lamartine J, Cintra Junior R, Daher JC, Cammarota MC, Galdino J, Pedroso DB. Reconstrução do complexo areolopapilar com double opposing flap. Rev Bras Cir Plást. 2013;28(2):233-40.
1. Hospital Daher Lago Sul, Serviço de Cirurgia
Plástica, Brasília, DF, Brazil.
2. Clínica Di Lamartine, Cirurgia Plástica,
Brasília, DF, Brazil.
Corresponding author: Jefferson Di Lamartine Galdino Amaral, SCN QD.02, Torre A, Salas 1121 e 1123 - 11º Andar - “Shopping Liberty Mall” - Brasília, DF, Brazil, Zip Code 70712-903. E-mail: jefferson@dilamartine.com.br
Article received: April 12, 2018.
Article accepted: October 1, 2018.
Conflicts of interest: none.













 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter