

Review Article - Year 2021 - Volume 36 -
What is the role of rapid diagnostic tests for COVID-19 IgM/IgG in the pre-operative period of cosmetic plastic surgery?
Qual o papel dos testes rápidos de diagnóstico de COVID-19 IgM/IgG no pré-operatório de cirurgia plástica estética?
ABSTRACT
Introduction: The disease by the new coronavirus 2019 (COVID-19) emerged in China and spread globally with sustained worldwide transmission from human to human. The COVID-19 IgM/IgG rapid diagnostic tests performed at the time of hospital admission, before elective surgery, are among the most widely used pre-operative screening methods.
Objectives: This study aims to evaluate the role of the rapid test of COVID-19 antibodies as screening in outpatients in aesthetic plastic surgery. Methods: A systematic review was carried out for studies published since December 2019, with several search terms related to the rapid antibody test for COVID-19 and SARS-CoV-2. The relevant articles were selected through the evaluation of titles and abstracts. Relevant articles have been revised. Data on the level of evidence, sensitivity, and specificity were collected.
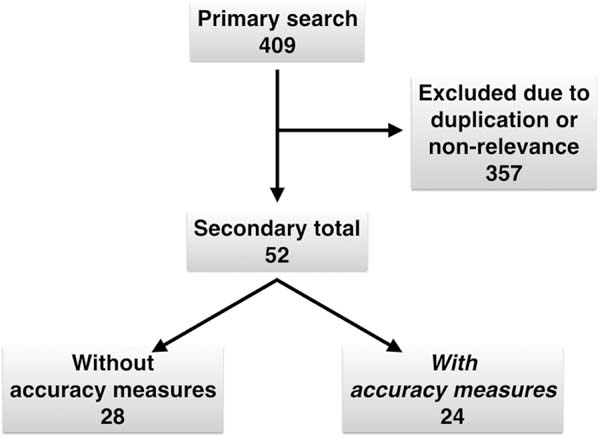
Results: The review strategy produced 409 manuscripts. A total of 357 studies were duplicated or proved to be irrelevant to the research question. Among the remaining articles, 28 were studies without precision information, and 24 were manuscripts describing precision measures. The sensitivity varied from 18.4 to 100%; the positive predictive value between 19.7 and 100%; specificity between 94 and 100%; and the negative predictive value between 20 and 100%.
Conclusion: COVID-19 IgM / IgG rapid diagnostic tests may be inaccurate. We found no evidence to support the rapid antibody test COVID-19 or SARS-CoV-2 for outpatients in cosmetic plastic surgery.
Keywords: Coronavirus infections; Pre-operative care; Plastic surgery; Epidemiology; Severe acute respiratory syndrome
RESUMO
Introdução: A doença pelo novo coronavírus 2019 (COVID-19) surgiu na China e se espalhou globalmente com transmissão mundial sustentada de humano para humano. Os testes de diagnóstico rápido COVID-19 IgM/IgG realizados no momento da admissão hospitalar, antes de cirurgia eletiva, estão entre os métodos de rastreamento pré-operatórios mais amplamente utilizados.
Objetivos: O objetivo deste estudo é avaliar o papel do teste rápido de anticorpos COVID-19 como triagem em pacientes ambulatoriais em cirurgia plástica estética.
Métodos: Uma revisão sistemática foi realizada para estudos publicados desde dezembro de 2019, com vários termos de pesquisa relacionados ao teste rápido de anticorpos para COVID-19 e SARS-CoV-2. Os artigos relevantes foram selecionados por meio da avaliação de títulos e resumos. Artigos pertinentes foram revisados. Dados sobre o nível de evidência, sensibilidade e especificidade foram coletados.
Resultados: A estratégia de revisão produziu 409 manuscritos. Um total de 357 estudos foram duplicados ou mostraram-se não relevantes para a questão de pesquisa. Entre os artigos restantes, 28 eram estudos sem informações sobre precisão e 24 eram manuscritos descrevendo medidas de precisão. A sensibilidade variou de 18,4 a 100%, o valor preditivo positivo variou entre 19,7 e 100%, a especificidade variou entre 94 e 100%, e o valor preditivo negativo ficou entre 20 e 100%.
Conclusão: Os testes de diagnóstico rápido COVID-19 IgM/IgG podem ser imprecisos. Não encontramos nenhuma evidência para apoiar o teste rápido de anticorpos COVID-19 ou SARS-CoV-2 para pacientes ambulatoriais em cirurgia plástica estética.
Palavras-chave: Infecções por coronavírus; Cuidados pré-operatórios; Cirurgia plástica; Epidemiologia; Síndrome respiratória aguda grave.
INTRODUCTION
Coronavirus disease 2019 (COVID-19) emerged in China and spread globally with sustained human-to-human transmission1. Due to its highly contagious nature, unprecedented global spread, aggressive clinical presentation, and the lack of effective treatment, the acute coronavirus 2 (SARS-CoV-2) respiratory infection syndrome is causing the loss of thousands of lives and repercussions unmatched in health systems worldwide2.
The infection caused by COVID-19 is a highly transmissible disease that presents a significant risk for both patients and health professionals3. Recently, it was demonstrated that high levels of the virus are present in respiratory secretions during the pre-symptomatic period, which can last from days to weeks before the characteristic symptoms of COVID-194. The virus’s ability to be transmitted by people without symptoms is one of the main reasons for the pandemic5.
The diagnosis of COVID-19 is made using clinical, laboratory, and radiological characteristics6. As the signs and radiological findings of COVID-19 are nonspecific, infection with SARS-CoV-2 must be confirmed by laboratory tests. Polymerase chain reaction tests with reverse transcriptase (RT-PCR) are the gold standard for the diagnosis of COVID-19. However, it is challenging to collect tests, and its results are not immediately available7. The rapid diagnostic tests for COVID-19 IgM / IgG were developed using lateral flow technology to find antigens from the SARS-CoV-2 virus and detect antibodies produced by patients infected with COVID-198.
Screening tests are widely used to assess the probability of members of a defined population having a specific disease; with few exceptions, screening tests do not diagnose the disease9. The rapid serological diagnostic test performed at the time of admission, before elective surgery, is among the most widely used pre-operative screening methods for COVID-1910-18.
OBJECTIVES
The aim of this study is to evaluate the role of rapid tests for COVID-19 antibodies in outpatients being admitted for aesthetic plastic surgery.
METHODS
A systematic review of the literature was performed using the search engines in PubMed, Web of Science, and SciELO journals, for studies with animals and humans published from December 2019 to July 30, 2020. We consider specific terms about COVID-19 or SARS- CoV-2 and plastic surgery. The following descriptors were used: “plastic surgery”, “elective surgery”, “COVID-19”, “COVID-19 diagnostic test”, “COVID-19 blood antibody test”, “SARS-CoV-2 test”. Many terms and words were displayed similarly when searching for articles. Words like “pre-operative,” “surgical,” and “surgery” showed similar results. The results of the words and phrases investigated were analyzed by quantity and quality. Documents written in English, Spanish, French, Italian, and Portuguese were included. Videos, posters, and letters to the editor were disregarded. Two researchers independently selected the relevant articles through the evaluation of titles and abstracts. The third researcher reviewed relevant articles. Data on the level of evidence, sensitivity, specificity, and predictive values of rapid diagnostic tests were collected.
This study follows Helsinki’s declaration and does not need to be evaluated by an ethics committee since it does not directly involve collecting data or tissues from human beings, only research conducted exclusively with scientific texts.
RESULTS
Using our active search strategy, the database review found 409 articles (Figure 1). A total of 357 studies were duplicated or considered not relevant to our research question. Among the remaining articles, 28 were studies without information on the accuracy of rapid diagnostic tests, and 24 were studies describing measures of accuracy7,8,19-40.
The level of evidence varied from V to III. The sensitivity varied from 18.4 to 100%, the specificity varied from 94 to 100%, the positive predictive value varied between 19.7 and 100%, and the negative predictive value was between 20 and 100%.
DISCUSSION
The limited experience accumulated during the COVID-19 pandemic has shown that the management of all medical conditions, including elective surgeries, has undergone some degree of change2. We all want to go back to work without the COVID-19 spectrum. During the extraordinary conditions of the COVID-19 pandemic, the ideal strategies for treating aesthetic patients individually are unknown. There is no consensus in the literature regarding pre-operative care, except that all patients should be screened for symptoms before being presented to the operating room, and those who report symptoms of COVID-19 should be referred for further evaluation.
The rapid diagnostic test can be produced quickly and cheaply. This qualitative test is small and portable, usually similar to a pregnancy test, showing to the user colored lines to indicate positive or negative results8. Rapid diagnostic tests do not measure the number of antibodies in the patient’s serum or whether these antibodies can protect against future infections. However, they do have the ability to detect exposure and can identify asymptomatic people and people who have cleared the virus. Many of the rapid diagnostic tests available so far lack analytical performance concerning sensitivity and specificity and need to be better validated before being used preoperatively21.
For a medical diagnosis, the test’s sensitivity is its ability to correctly identify those with the disease (true positive rate), while the test’s specificity is its ability to accurately identify those without the disease (true negative rate). In this research, sensitivity ranged from 18.4 to 100%, reflecting a potential inability to identify people who have antibodies to COVID-19 correctly. Specificity varied between 94 and 100%, demonstrating a high ability to identify all patients who do not have COVID-19 antibodies.
The negative predictive value is the probability that patients with a negative result in a rapid diagnostic test do not have COVID-19 antibodies; in our research, their values were between 20 and 100%, we can say that, in some circumstances, 80% of individuals with a negative test may have COVID-19 antibodies. Positive predictive value is the likelihood that individuals with a rapid positive diagnostic screening test will have the disease; in our research, its variation was between 60 and 100%. Consequently, we can affirm that, in some circumstances, 40% of the individuals with positive rapid tests may not have antibodies to COVID- 19. Therefore, the rapid test results seem to be scientifically unreliable, and the recommendation to perform this testing in a generalized way by patients or hospital institutions seems inadequate.
It is estimated that SARS-CoV-2 IgM antibodies can be detected in a blood sample after three days and IgG antibodies eight days after the onset of symptoms7. The seroconversion rate for IgM and IgG was described as 82.7% and 64.7%, respectively5. To date, we do not know whether everyone who has recovered from COVID-19 has developed antibodies, and we do not know to what extent these antibodies protect patients from reinfection. The antibody tests do not detect an active infection but look for signs that a person has been previously infected, as shown by the antibodies his immune system has produced to fight the coronavirus. With other diseases, the presence of antibodies usually means acquired immunity for at least some period, but this is not yet known in the case of COVID-194.
Patients should be screened only if a positive test results in mandatory action. This is not the case for rapid diagnostic tests for COVID-19 before cosmetic surgery because the procedure will be performed regardless of antibody detection status. In the case of the new COVID-19 virus and the SARS-CoV-2 disease it causes in humans, the objective of pre-operative testing would be straightforward: to identify infected patients and isolate them by postponing their surgeries, trying to reduce the morbidities of the procedure and thus reducing the risk of infection for health professionals8. Nevertheless, without a perfect test, false positives and false negatives can lead to significantly worse outcomes.
Both false positives and false negatives pose their own unique dangers wherever testing occurs, but false negatives are particularly dangerous for COVID-19. Two weeks after surgery, some patients may be positive for COVID-19; despite the negative pre-operative result, important medico-legal implications may arise. Was the infection contracted during hospitalization? Did the surgeon or his team contaminate the patient during outpatient postoperative follow-up? Did seroconversion occur because of the surgery-induced immunosuppression? A negative result in a rapid diagnostic test for COVID-19 performed preoperatively can be dangerous medico-legal evidence for surgeons and hospital entities.
Antibody tests are versatile: these serological tests are of critical importance to determine seroprevalence, prior exposure and to identify highly reactive human donors for the generation of therapeutic convalescent serum4. They will also support contact screening and screening for healthcare professionals to identify those who are already immune.
It is plausible that several limitations may have influenced the results obtained in this research. The exclusion of articles in Asian languages is one of them since much of the knowledge about COVID-19 comes from this geographic area. However, there was none among the researchers with knowledge of these languages , and we consider that electronic translators are not reliable. However, many of these studies would provide information with limited external validity for patients in the Americas since COVID-19 mutations are frequent, and most of the rapid diagnostic tests used there are not available on our continent. A well-designed systematic review benefits the evolution of knowledge, identifying a lack of scientific information and providing a synopsis of the available evidence. The credibility of systematic reviews can be compromised by reporting bias, which arises when the results’ nature influences the dissemination of published articles. Our findings are based on a limited number of articles; therefore, the results of such analysis should be treated with utmost caution.
Controlled clinical trials are lacking, and future studies should examine the safety and efficacy of rapid diagnostic tests for COVID-19 to obtain more consistent results and establish recommendations for their appropriate use.
CONCLUSION
The COVID-19 IgM / IgG rapid diagnostic tests appear to be inaccurate. We found no evidence to support COVID-19 or SARS-CoV-2 antibodies’ rapid testing to screen outpatients for cosmetic plastic surgery. Future studies on the subject are needed to validate different laboratory diagnostic tests.
REFERENCES
1. Contini C, Di Nuzzo M, Barp N, Bonazza A, De Giorgio R, Tognon M, et al. The novel zoonotic COVID-19 pandemic: an expected global health concern. J Infect Dev Ctries. 2020 Mar;14(3):254-64. DOI: https://doi.org/10.3855/jidc.12671
2. Patel V, Jimenez E, Cornwell L, Tran T, Paniagua D, Denktas AE, et al. Cardiac surgery during the COVID-19 pandemic: perioperative considerations and triage recommendations. J Am Heart Assoc. 2020 Jul;9(13):e017042. DOI: https://doi.org/10.1161/jaha.120.017042
3. He W, Yi GY, Zhu Y. Estimation of the basic reproduction number, average incubation time, asymptomatic infection rate, and case fatality rate for COVID-19: meta-analysis and sensitivity analysis. J Med Virol. 2020 Mai;92(1):2543-50. DOI: https://doi.org/10.1002/jmv.26041
4. Kim SE, Jeong HS, Yu Y, Shin SU, Kim S, Oh TH, et al. Viral kinetics of SARS-CoV-2 in asymptomatic carriers and presymptomatic patients. Int J Infect Dis. 2020 Jun;95:441-3.
5. Wong MC, Teoh JY, Huang J, Wong SH. Strengthening early testing and surveillance of COVID-19 to enhance identification of asymptomatic patients. J Infect. 2020 Ago;81(2):E112-3. DOI: https://doi.org/10.1016/j.jinf.2020.05.048
6. Luo N, Zhang H, Zhou Y, Kong ZX, Sun WH, Huang N, et al. Utility of chest CT in diagnosis of COVID-19 pneumonia. Diagn Interv Radiol. 2020 Mai;26:437-42. DOI: https://doi.org/10.5152/dir.2020.20144
7. Li Z, Yi Y, Luo X, Xiong N, Liu Y, Li S, et al. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J Med Virol. 2020 Fev;92(9):1518-24. DOI: https://doi.org/10.1002/jmv.25727
8. Van Elslande J, Houben E, Depypere M, Bracknier A, Desmet S, André E, et al. Diagnostic performance of 7 rapid IgG/IgM antibody tests and the Euroimmun IgA/IgG ELISA in COVID-19 patients. Clin Microbiol Infect. 2020 Ago;26(8):1082-7. DOI: https://doi.org/10.1016/j.cmi.2020.05.023
9. Maxim LD, Daniel Maxim L, Niebo R, Utell MJ. Screening tests: a review with examples. Inhal Toxicol. 2014 Set;26(13):811-28. DOI: https://doi.org/10.3109/08958378.2014.955932
10. The Aesthetic Society (AS). COVID-19 updates [Internet]. Garden Grove: AS; 2020; [acesso em 2020 Ago 01]. Disponível em: https://www.surgery.org/professionals/covid-19/covid-19-updates
11. Kaye K, Paprottka F, Escudero R, Casabona G, Montes J, Fakin R, et al. Elective, non-urgent procedures and aesthetic surgery in the wake of SARS-COVID-19: considerations regarding safety, feasibility and impact on clinical management. Aesthetic Plast Surg. 2020 Mai;44:1014-42. DOI: https://doi.org/10.1007/s00266-020-01752-9
12. Flexman AM, Abcejo AS, Avitsian R, Sloovere V, Highton D, Juul N, et al. Neuroanesthesia practice during the COVID-19 pandemic: recommendations from Society for Neuroscience in Anesthesiology and Critical Care (SNACC). J Neurosurg Anesthesiol. 2020 Jul;32(3):202-9. DOI: https://doi.org/10.1097/ANA.0000000000000691
13. Zeegen EN, Yates AJ, Jevsevar DS. After the COVID-19 pandemic: returning to normalcy or returning to a new normal?. J Arthroplasty. 2020 Jul;35(7 Supl 1):S37-S41. DOI: https://doi.org/10.1016/j.arth.2020.04.040
14. Liu Z, Zhang Y, Wang X, Zhang D, Diao D, Chandramohan K, et al. Recommendations for surgery during the novel coronavirus (COVID-19) epidemic. Indian J Surg. 2020 Abr;1-5.
15. Paraiso MFR, Brown J, Abrão MS, Dionisi H, Rosenfield RB, Lee TM, et al. Surgical and clinical reactivation for elective procedures during the COVID era: a global perspective. J Minim Invasive Gynecol. 2020 Jul;27(5):1188-95. DOI: https://doi.org/10.1016/j.jmig.2020.05.012
16. Carugno J, Di Spiezio Sardo A, Alonso L, Haimovich S, Campo R, De Angelis C, et al. COVID-19 Pandemic. Impact on hysteroscopic procedures: a consensus statement from the global congress of hysteroscopy scientific committee. J Minim Invasive Gynecol. 2020 Jul;27(5):988-92. DOI: https://doi.org/10.1016/j.jmig.2020.04.023
17. Meng Y, Leng K, Shan L, Guo M, Zhou J, Tian Q, et al. A clinical pathway for pre-operative screening of COVID-19 and its influence on clinical outcome in patients with traumatic fractures. Int Orthop. 2020 Ago;44(8):1549-55.
18. Ren X, Chen B, Hong Y, Liu W, Jiang Q, Yang J, et al. The challenges in colorectal cancer management during COVID- 19 epidemic. Ann Transl Med. 2020 Abr;8(7):498. DOI: https://doi.org/10.21037/atm.2020.03.158
19. Jacofsky D, Jacofsky EM, Jacofsky M. Understanding antibody testing for COVID-19. J Arthroplasty. 2020 Jul;35(7 Supl 1):S74-S81. DOI: https://doi.org/10.1016/j.arth.2020.04.055
20. Ragó Z, Szijjártó L, Duda E, Bella Z. Opportunity of periodic monitoring of COVID-19 patients, asymptomatic virus carriers, and postinfectious individuals with IgM/IgG rapid antibody tests among healthcare workers during SARS-CoV-2 pandemic. Orv Hetil. 2020 Mai;161(21):854-60.
21. Shen B, Zheng Y, Zhang X, Zhang W, Wang D, Jin J, et al. Clinical evaluation of a rapid colloidal gold immunochromatography assay for SARS-Cov-2 IgM/IgG. Am J Transl Res. 2020;12(4):1348-54.
22. Spicuzza L, Montineri A, Manuele R, Crimi C, Pistorio MP, Campisi R, et al. Reliability and usefulness of a rapid IgM-IgG anti- body test for the diagnosis of SARS-CoV-2 infection: a preliminary report. J Infect. 2020 Ago;81(2):53-4. DOI: https://doi.org/10.1016/j.jinf.2020.04.022
23. Cassaniti I, Novazzi F, Giardina F, et al. Performance of VivaDiag COVID-19 IgM/IgG Rapid Test is inadequate for diagnosis of COVID-19 in acute patients referring to emergency room department. J Med Virol. 2020 Mar;92(10):1724-7. DOI: https://doi.org/10.1002/jmv.25800
24. Padoan A, Cosma C, Sciacovelli L, Faggian D, Plebani M. Analytical performances of a chemiluminescence immunoassay for SARS-CoV-2 IgM/IgG and antibody kinetics. Clin Chem Lab Med. 2020;58(7):1081-8. DOI: https://doi.org/10.1515/cclm-2020-0443
25. Rosa MI, Prestes GS, Macedo ACL, Colonetti T, Uggioni MLR, Grande AJ. Accuracy of rapid IgM and IgG antibody test for SARS-CoV-2 infection diagnosis: a systematic review and meta analysis. Braz J Infect Dis. 2020. DOI: https://doi.org/10.37766/inplasy2020.4.0099
26. Pulia MS, O'Brien TP, Hou PC, Schuman A, Sambursky R. Multi-tiered screening and diagnosis strategy for COVID-19: a model for sustainable testing capacity in response to pandemic. Ann Med. 2020 Ago;52(5):207-14.
27. Castro R, Luz PM, Wakimoto MD, Veloso VG, Grinsztejn B, Perazzo H. COVID-19: a meta- analysis of diagnostic test accuracy of commercial assays registered in Brazil. Braz J Infect Dis. 2020 Mar/Abr;24(2):180-7.
28. Saenz-Flor KV, Santafe LM. Concordance of "rapid" serological tests and IgG and IgM chemiluminescence for SARS-COV-2. medRxiv [Internet]. 2020 Jun 03; [Epub preprint]. DOI: https://doi.org/10.1101/2020.06.01.20114884
29. Prazuck T, Colin M, Giachè S, Gubavu C, Seve A, Rzpecki V, et al. Evaluation of performance of two SARS-CoV-2 rapid whole-blood finger-stick IgM-IgG combined antibody tests. medRxiv [Internet]. 2020 May 27; [Epub preprint]. DOI: https://doi.org/10.1101/2020.05.27.20112888
30. Di Lorenzo G, Toniolo P, Lurani C, Foresti L, Carrisi C. Evaluating the adequacy of Prima Covid-19 IgG/IgM Rapid Test for the assessment of exposure to SARS-CoV-2 virus. medRxiv [Internet]. 2020 Jun 03; [Epub preprint]. DOI: https://doi.org/10.1101/2020.05.30.20117424
31. Ying, Yue-Ping L, Bo D, Fei-Fei R, Yue W, Jinya D, et al. Diagnostic indexes of a rapid IgG/IgM combined antibody test for SARS-CoV-2. medRxiv [Internet]. 2020 Mar 30; [Epub preprint]. DOI: https://doi.org/10.1101/2020.03.26.20044883
32. Hoffman T, Nissen K, Krambrich J, Rönnberg B, Akaberi D, Esmaeilzadeh M, et al. Evaluation of a COVID-19 IgM and IgG rapid test; an efficient tool for assessment of past exposure to SARS-CoV-2. Infect Ecol Epidemiol. 2020 Abr;10(1):1754538. DOI: https://doi.org/10.1080/20008686.2020.1754538
33. Pérez-García F, Pérez-Tanoira R, Romanyk J, Arroyo T, Gómez-Hurruz P, Cuadros-González J. Rapid diagnosis of SARS-CoV-2 infection by detecting IgG and IgM antibodies with an immunochromatographic device: a prospective single-center study. medRxiv [Internet]. 2020 Abr 24; [Epub preprint]. DOI: https://doi.org/10.1101/2020.04.11.20062158
34. Minteer C, Casanovas-Massana A, Li T, McDonald D, Wang L, Pan SH, et al. Multi-site validation of a SARS-CoV-2 IgG/IgM rapid antibody detection kit. medRxiv [Internet]. 2020 Mai 26; [Epub preprint]. DOI: https://doi.org/10.1101/2020.05.25.20112227
35. Yangchun F. Optimize clinical laboratory diagnosis of COVID-19 from suspect cases by likelihood ratio of SARS-CoV-2 IgM and IgG antibody. medRxiv [Internet]. 2020 Abr 08; [Epub preprint]. DOI: https://doi.org/10.1101/2020.04.07.20053660
36. Kohmer N, Westhaus S, Rühl C, Ciesek S, Rabenau HF. Clinical performance of SARS-CoV-2 IgG antibody tests and potential protective immunity. medRxiv [Internet]. 2020 Mai 10; [Epub preprint]. DOI: https://doi.org/10.1101/2020.05.08.085506
37. Qu J, Wu C, Li X, Zhang G, Jiang Z, Li X, et al. Profile of immunoglobulin G and IgM antibodies against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020 Out;71(16):2255-8. DOI: https://doi.org/10.1093/cid/ciaa489
38. Capello F, Cipolla M, Cosco L, Gnasso A, Mancini R, Nichelatti M, et al. The VivaDiag COVID-19 lgM/IgG rapid test for the screening and early diagnosis of COVID-19 in patients with no clinical signs of the disease. Int J Endocr Metab Dis. 2020;6(1):1-4. DOI: https://doi.org/10.16966/2380-548x.167
39. Xie J, Ding C, Li J, Wang Y, Guo H, Lu Z, et al. Characteristics of patients with coronavirus disease (COVID-19) confirmed using an IgM-IgG antibody test. J Med Virol. 2020 Abr;92(10:2004-10. DOI: https://doi.org/10.1002/jmv.25930
40. Ma H, Zeng W, He H, Zhao D, Jiang D, Zhou P, et al. Serum IgA, IgM, and IgG responses in COVID-19. Cell Molec Immunol. 2020 Mai;17:773-5. DOI: https://doi.org/10.1038/s41423-020-0474-z
1 . Pontifical Catholic University of the Rio
Grande do Sul, Postgraduate Program in Medicine and Health Sciences, Porto
Alegre, RS, Brazil.
2 . Mãe de Deus Health System, Hospital Mãe de Deus
Carlos Gomes, Porto Alegre, RS, Brazil.
RKZ Análise e/ou interpretação dos dados, Análise estatística, Aprovação final do manuscrito, Aquisição de financiamento, Coleta de Dados, Conceitualização, Concepção e desenho do estudo, Gerenciamento do Projeto, Investigação, Metodologia, Realização das operações e/ ou experimentos, Redação - Preparação do original
SSV Análise e/ou interpretação dos dados, Análise estatística, Aprovação final do manuscrito, Coleta de Dados, Conceitualização, Concepção e desenho do estudo, Gerenciamento do Projeto, Investigação, Metodologia, Realização das operações e/ou experimentos, Redação - Preparação do original, Visualização
DSV Análise e/ou interpretação dos dados, Análise estatística, Aprovação final do manuscrito, Coleta de Dados, Conceitualização, Concepção e desenho do estudo, Investigação, Metodologia, Realização das operações e/ou experimentos, Redação - Preparação do original, Redação - Revisão e Edição, Supervisão, Visualização
Corresponding author: Denis Souto Valente Rua Antônio Carlos Berta, 475/702, Jardim Europa, Porto Alegre, RS, Brazil. Zip Code: 91340-020 E-mail: denisvalentedr@gmail.com
Article received: July 05, 2020.
Article accepted: January 10, 2021.
Conflicts of interest: none



 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter