

Original Article - Year 2021 - Volume 36 -
Synthetic mesh as a potential low-cost alternative to dermal matrix in mammary reconstructions
Tela sintética como alternativa potencial de baixo custo à matriz dérmica nas reconstruções mamárias
ABSTRACT
Introduction: The use of acellular dermal matrices (ADM) in immediate breast reconstruction with silicone prosthesis (IBRP) led to a decrease in the number of surgical procedures (concerning the silicone expander) secondary to mastectomy, allowing the use of silicone implants >300cc. Its high cost made large-scale employment impossible (mainly in countries outside the US). Synthetic meshes proved to be a viable and widely used alternative (mainly in Europe). This study aimed to identify any difference in the surgical efficiency of IBRP after mastectomy using biological (ADM) or synthetic materials.
Methods: A search was performed in the PubMed and VHL last five years databases (preferably), using the keywords: "mammoplasty" and "mastectomy" and "surgical screens" and "acellular dermal matrix" and "breast implants" and "postoperative complications".
Results: The incidence of infection, hematoma, skin necrosis, seroma, extrusion or loss of implant and capsular contracture found in the research revealed to be complications inherent to the preoperative characteristics of patients: age >65 years, body mass index (BMI) >30kg/m2, large breasts (more than 600g), extensive skin resections and skin incisions in inverted T, smoking, diabetes, hypertension, time of long removal of the drain in the postoperative period, radiotherapy in the pre/postoperative period and that reported postoperative complications were not related to the use of ADM or synthetic mesh.
Conclusion: The consensus of the data presented shows that the IBRP with synthetic meshes produced aesthetic results comparable to the ADMs, with lower costs and complication rates.
Keywords: Surgical screens; Breast implants; Postoperative complications; Mastectomy; Mammoplasty; Breast neoplasms.
RESUMO
Introdução: O emprego das matrizes dérmicas acelulares (ADM) na reconstrução mamária imediata com prótese de silicone (RMIP) levou à diminuição do número de procedimentos cirúrgicos (em relação ao expansor de silicone) secundários à mastectomia, permitindo a utilização de implantes de silicone >300cc. O seu alto custo inviabilizou o emprego em larga escala (principalmente em países fora dos EUA) e a utilização de malhas sintéticas mostrou-se uma alternativa viável e bastante utilizada (principalmente na Europa). Este estudo objetivou identificar se há alguma diferença na eficiência cirúrgica da RMIP após a mastectomia utilizando materiais biológicos (ADM) ou sintéticos.
Métodos: Foi realizada uma pesquisa nos bancos de dados PubMed e BVS dos cinco últimos anos (preferencialmente), usando as palavras-chave: "mamoplastia" e "mastectomia" e "telas cirúrgicas" e "matriz dérmica acelular" e "implantes de mama" e "complicações pós-operatórias".
Resultados: A incidência de infecção, hematoma, necrose cutânea, seroma, extrusão ou perda de implante e contratura capsular encontrados na pesquisa revelou serem complicações inerentes às características pré-operatória dos pacientes: idade >65 anos, índice de massa corpórea (IMC) >30kg/m2, mamas grandes (mais de 600g), ressecções extensas de pele e incisões cutâneas em T invertido, tabagismo, diabetes, hipertensão, tempo de remoção longo do dreno no pós-operatório, radioterapia no pré/pós-operatório e que referidas complicações no pós-operatório não tinham relação com a utilização de ADM ou malha sintética.
Conclusão: O consenso dos dados apresentados mostra que a RMIP com malhas sintéticas produziu resultados estéticos comparáveis às ADMs, com menores custos e taxas de complicações.
Palavras-chave: Telas cirúrgicas; Implantes de mama; Complicações pós-operatórias; Mastectomia; Mamoplastia; Neoplasias da mama
INTRODUCTION
The popularity of breast reconstruction with silicone prosthesis is explained by its technical simplicity and easy reproducibility, lower local morbidity and shorter surgical time when compared to breast reconstruction with autologous tissues.
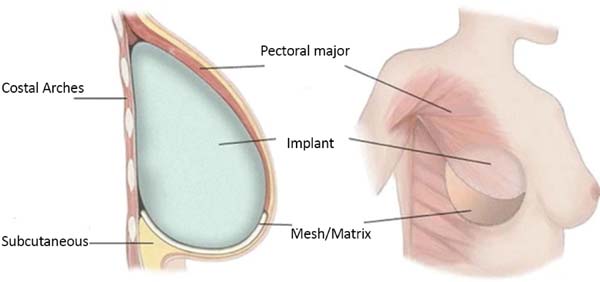
The complete prosthesis coverage by a submuscular pocket to prevent implant exposure during possible dehiscence of the skin/wound has been, for decades, a safer choice for immediate breast reconstruction with a prosthesis (IBRP).
With the introduction of soft tissue replacement materials, biological or synthetic, combined with the submuscular pocket created, an alternative option emerged to perform an IBRP in a single time and with silicone prosthesis of larger volumes (>300cc).
This paper presents a bibliographic review of the use of synthetic screens and biological matrices in the IBRP, comparing differences in aesthetic results, costs and the most reported complications, in addition to the description of the surgical technique employed by the authors and the respective results.
OBJECTIVE
This study aimed to identify, through a literature review, if there is any difference in surgical efficiency in IBRP after mastectomy using a synthetic mesh in place of the biological matrix.
METHODS
Literature research was conducted (considering the period of the last five years, preferably), in the PubMed and VHL databases totaling 20 articles using the keywords “mammoplasty” and “mastectomy” and “surgical screens” and “acellular dermal matrix” and “breast implants” and “postoperative complications”. The number of the opinion in the medical ethics committee was: 4,193,275.
Surgical technique employed by the authors
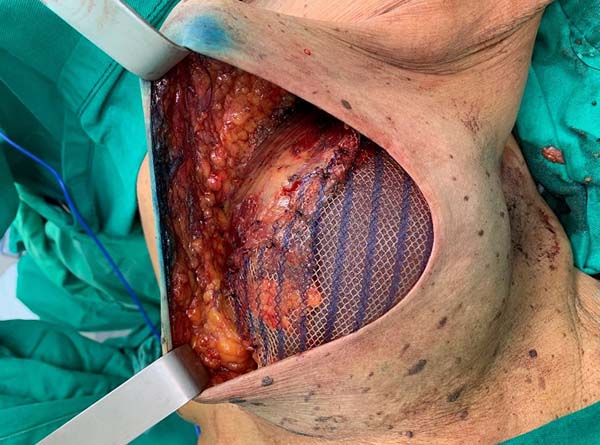
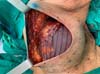
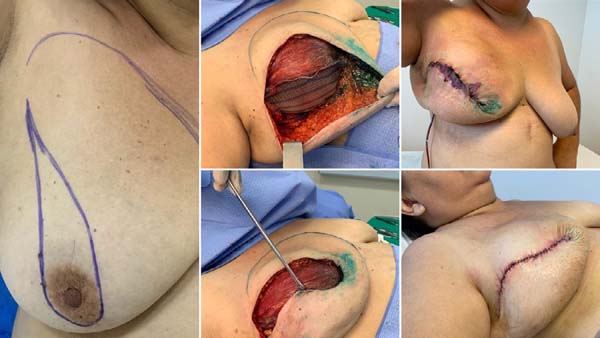
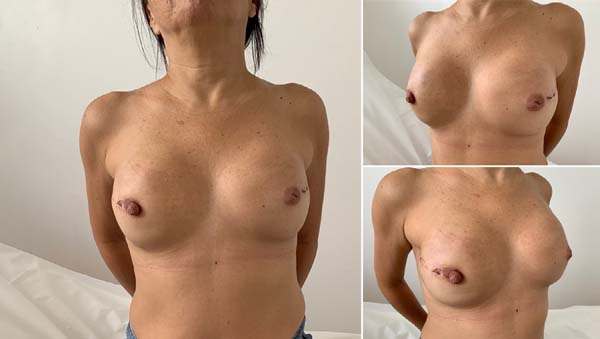
This surgical technique has been used since February 2019, in a private clinic and public health institution, with the use of Ultrapro mesh®. A major pectoral submuscular pocket was dissected in patients undergoing mastectomy and presenting an adequate skin flap, with a minimum of 1 cm thickness and good quality. The inferomedial edge of the pectoral muscle (up to 2nd intercostal space) was elevated for placement of a silicone mold. The synthetic mesh was sutured to this edge of the pectoral muscle, along its entire lower course to the inframammary groove. After choosing the implant, it was introduced under the synthetic mesh. This material was fixed (with Vicryl® 2-0 suture) laterally at the level of the axillary line anterior to the dissected fascia of the anterior serrátil muscle and even the inframammary groove in the desired position. Two suction drains were placed, one in the submuscular spoof and the other in the subcutaneous, through separate cutaneous accesses, followed by surgical wound closure, by planes (Figures 1 and 2).
Materials used universally in the composition of the submuscular pocket:
Acellular dermal matrix (ADM)
A biological mesh, also referred to as acellular dermal matrix (ADM), is a dermis scaffold (skeleton), produced from human cadaver(Alloderm ®, Allomax®, FlexHD® and DermaCell®, pig (Strattice® and Permacol™), bovine (SurgiMend® or bovine pericardium (Veritas® consisting of a flexible tissue blade that is stripped of its antigenic cells through a specific chemical processing. This biological skeleton allows rapid revascularization of the patient (host) and its cell repopulation, optimizing the surgical result.
Synthetic mesh (mesh)
Non-biological materials have been introduced as low-cost alternatives to ADMs. They are available in absorbable materials: Vicryl® (Ethicon Inc., USA), SeraGyn® BR (Serag Wiessner, Germany) and TIGR® Matrix(Novus Scientific, Uppsala, Sweden); or partially absorbable: ULTRAPRO® (Ethicon Inc., USA) and TiLOOP® Bra (Pfm Medicalis, Germany).
Bibliographic update
Twenty articles were analyzed in the PubMed and VHL last five years databases (preferably). They allowed an updated view of the use of synthetic mesh and acellular dermal matrix in immediate mammary reconstructions and evidence of the need for a careful selection of patients who are candidates for their use. The incidence of infection, hematoma, necrosis of the skin flap, seroma, extrusion or loss of the implant, capsular contracture were influenced by pre- and/or intraoperative factors that included: age (over 65 years), large breasts (more than 600g), extensive skin resections and inverted T-incisions, obesity (BMI greater than 30kg/m2), smoking, diabetes, hypertension, length of removal time in the postoperative period (PO), pre-postoperative radiotherapy.
DISCUSSION
The use of mesh or matrix in breast reconstruction is associated with some complications such as infection, seroma, hematoma, capsular contracture, necrosis of the skin flap, extrusion or implant loss. It is clear that the characteristics inherent to patients are associated with increased rates of complications, with or without a mesh/matrix. Patient selection is essential to avoid complications. Well-identified risk factors for complications are well known as the history of previous radiotherapy or indication of postoperative radiotherapy, active smoking, age group over 65 years, body mass index (BMI) >30kg/m2, resection of large breast volumes (>600g)1-3.
The presence of significant mammary ptosis (grade III and IV), the need for large skin resections and/or the choice of an inverted T-skin incision promotes a higher risk of skin necrosis. The implant volume should be adapted to avoid any tension in the closure of the surgical wound. The proper vascularization of the cutaneous flaps of the mastectomy should be strictly observed. During a mastectomy, the dissection technique should be careful in the cutaneous-glandular separation, preserving a flap, without irregularities, with a minimum of 1cm thickness4-6.
Spear et al. (2008) 7 found a higher rate of complications: 42.1% (p=0.002) among irradiated and non-irradiated patients, showing that in patients to whom radiotherapy was applied, there were higher rates of contracture and implant loss (when replacing the expander).
Nyame et al. (2011) 8 conducted a resazurin® which can be reliably used to quantify bacterial adhesion to acellular dermal matrix material or synthetic material. The S. aureus SC-1 and UAMS-1 strains adhered better to acellular dermal matrix materials(AlloDerm® versus FlexHD® than synthetic material: Prolene®. The S. aureus strain also adhered better to Vicryl® than to Prolene® .
Jacobs et al. (2015) 9 presented a review of the use of ADM and synthetic screens in breast reconstruction with silicone prosthesis that allowed a decrease in the number of surgical procedures (in relation to the provisional expander) secondary to mastectomy. They pointed out the low cost of synthetic fabrics (absorbable or not) as a safe alternative to ADMs despite the regenerative and integration potential to adjacent tissues of ADMs. They discussed some of the main complications in the late postoperative period, such as capsular contracture, poor implant position, rippling, symastia and animation deformities (migration of the implant superiorly or laterally when contraction of the pectoralis major muscle occurs).
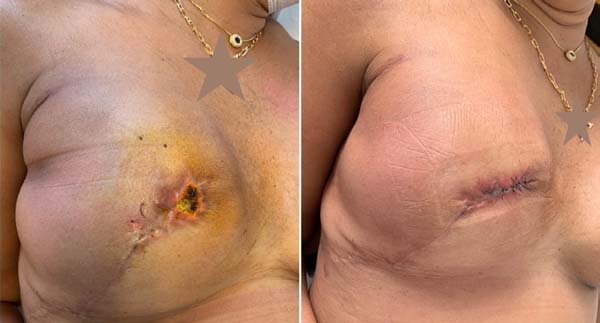
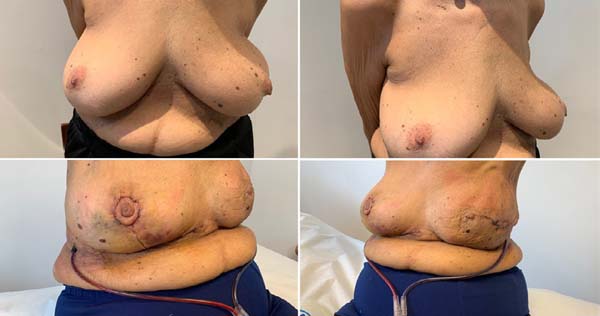
Gschwantler-Kaulich et al. (2016) 10 conducted a prospective study with 48 patients, divided into two groups; 25 patients submitted to mammary reconstruction with a nonabsorbable synthetic mesh(Tiloop® and 23 patients used porcine ADM (Protexa®. The mean age was 48 years, and body mass index (BMI): 24kg/m2. Different surgical incisions were used: inframammary:14 patients, inverted T: 16 patients, periareolar or tennis racket: 18 patients. There were nine complications in the ADM group (three seromas, three infections, two dehiscences in surgical scars: inverted T and one patient with red breast syndrome) (Figure 3).
There were six complications in the group that used the synthetic mesh, one seroma, one infection, 2 hematomas, and 2 dehiscences in surgical scars (inverted T). These complications caused implant loss in 7 patients in the ADM group (30.4%) and 2 patients in the synthetic mesh group (7.7%). In the evaluation after 6 months postoperatively, there was a better aesthetic result in the synthetic mesh group.
Bertozzi et al. (2017) 4 emphasized the requirement (after mastectomy) of a skin flap with a minimum thickness of 1 cm, well-vascularized, thus, reducing the incidence of skin necrosis. They recommended immediate reconstruction with breast prosthesis up to 600g and/or mild ptoses. They stated absolute contraindication for breasts that will be irradiated later.
Pukancsik et al. (2017) 11 carried out a retrospective study, from 2013 to 2016, with 102 patients who underwent immediate breast reconstruction using a silicone prosthesis and partially absorbable Ultrapro® synthetic mesh. The mean age of the patients was 43 years, with a BMI of 23 kg/m2. The average volume of surgical implants was 480cc. The average rate of complications was 18.3%, with a rate of surgical reintervention of 11.4%. Seroma was the most common complication (5.1%), and skin infection was noted in 8 patients (4.5%) without removing the silicone implant or synthetic mesh. There were complications in 50% of the patients who had undergone preoperative radiotherapy. There was removing the silicone implant in 7 patients (4%) due to skin necrosis and/or nipple-areola complex. The authors revealed the great difference in costs of the synthetic canvas (30 euros) in relation to ADM ($ 1,825- $ 4,856 - depending on the size, thickness and manufacturer).
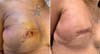
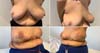
Machleidt et al. (2018) 12 reported the increasing use of biological or synthetic materials in breast reconstruction with silicone prosthesis. They conducted a prospective study with 148 breasts using a partially absorbable synthetic mesh(Seragyn® with a mean follow-up of seven months, patients with a mean age of 49, and BMI of 24kg/m2. In 38 patients (25.7%), postoperative seroma was attributed to the greater number of patients submitted to enlarged skin resections and/or surgical approach of the armpit. In 21 patients (14.2%), there were hemorrhagic complications. In 21 patients, there were skin infections. There were changes in skin flap vascularization in 20 patients (13.5%) (Figure 4).
The synthetic mesh had to be removed in 11 patients (7.4%) due to skin infection and five patients (3.4%) due to changes in surgical healing. These patients with complications had an average dry breast weight of 440g and an average drain permanence period of 10 days.
Gschwantler-Kaulich et al (2018)13 conducted a retrospective study from 2008 to 2013, with 180 patients undergoing immediate mammary reconstruction - IMR (n=148, 82.2%) (n=32, 17.8%), with BMI=23.4 kg/m2. The reconstruction with silicone prosthesis (without autologous flaps at a distance) corresponded to 62 patients (34.4%), of which 33 patients (18.4%) were used: synthetic mesh (Tiloop®) in 5 patients and 28 patients, porcine ROM (Strattice®) was used. There were findings of a higher incidence of complications (hematoma/skin necrosis) in this IMR group (37.1%), in relation to the permanent expander group (10.5%) and the two-stage reconstruction/temporary expander group (22.9%).
After 46 months of follow-up, there was no statistically significant difference in the 3 groups regarding the loss of silicone implant or severe capsular contracture. Patients submitted to papilla-sparing mastectomy and immediate breast reconstruction with silicone prosthesis showed better positive results in the questionnaire on quality of life concerning sexual interest/activities and aesthetic results.
Pompei et al. (2018) 15 conducted a retrospective study, from 2014 to 2016, with 49 patients undergoing immediate breast reconstruction using TIGR® synthetic absorbable mesh. The mean age was 51 years, and only eight patients (16.3%) had a BMI above 30kg/m2. Of a total of 60 screens used, 2 patients had seroma, one patient had skin infection (requiring removal of the screen), 3 patients had skin necrosis (resulting in the removal of 1 screen), 3 patients had dehiscence of surgical scar and hematoma was present in 4 patients. They found evidence that the complication rate in obese patients was 22%, compared to patients with normal BMI (2%).
Hallberg et al. (2018) 16 carried out a retrospective study (2015 to 2016) with 49 patients (65 breasts) who used TIGR® synthetic mesh in breast reconstruction with silicone prosthesis. The mean age of the patients was 46 years old, BMI 23kg/m2. Sixty tissue expanders (92.3%) were used. There were 15 breasts (23%) with complications in the first 30 postoperative days. Four patients with severe complications, which caused the loss of 2 silicone implants (skin necrosis and skin infection) ), and an urgent reoperation (hematoma followed by venous thromboembolism, in a patient with a history of previous valve heart disease). The most used skin incision was in the breast crease: 39 breasts (61.9%). The mean weight of the resected breasts was 255g, attributed to the lower BMI of the patients, with a low seroma index (3.1%) and skin infection (1.5%).
Remington et al. (2019)14 reported a retrospective study of 288 reconstructed breasts with silicone expander positioned sub-pectoral in the upper half and used ADM (Alloderm®) in the lower and lateral half. The mean age of the patients was 46 years old, with a BMI of 28 kg/m2. Papilla-sparing mastectomy was performed in 124 patients (74.7%), and skin-sparing mastectomy was performed in 24 patients (14.5%). The average volume of intraoperative expansion was 81ml, requiring an average of 4 outpatient expansions to reach a maximum volume of 520ml (in the average of patients). The suction drain was removed, on average, after 15 days. The rate of postoperative infection was 16.9%, and cutaneous necrosis of mastectomy was 6.6%. The loss of reconstruction (early removal of the expander) occurred in 14 patients (8.4%). A BMI greater than 27kg/m2 was correlated as a predisposing factor for the need to use larger expanders and a consequent greater chance of complications in the postoperative period.
Hallberg et al. (2019) 17 conducted a prospective study of patients submitted to immediate breast reconstruction with silicone prosthesis. One group (1) studied from 2005 to 2014, with 116 breasts, using a biological matrix (Surgisis®) as a complement of the submuscular pocket and another group (2) studied from 2015 to 2016, with 65 breasts, using synthetic mesh (TIGR®). There was a similarity in the groups concerning age group and BMI. There were similar capsular contracture indices, but group 1 had lower cosmetic satisfaction. They concluded that there was evidence of good long-term results using both materials.
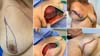
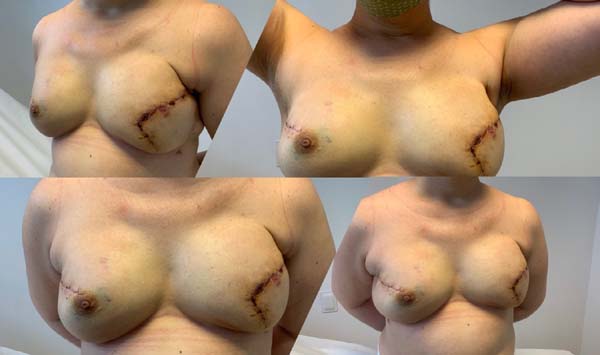
Bonomi et al. (2019) 18 conducted a retrospective study (2014 to 2016) with 56 patients who presented medium to marked hypertrophy and breast ptosis and underwent immediate breast reconstruction (after mastectomy) through an inverted T-skin incision in Italy. They used a de-epithelialized dermal flap on the submuscular pocket and Vicryl® absorbable synthetic mesh to protect them from possible local skin necrosis. Reconstruction was unilateral in 50 patients with opposite mammoplasty in 45 patients. The mean age of the patients was 47 years, BMI was 23.8kg/m2. The average volume of silicone prostheses was 520g, and there was an average of 20 days of use of suction drain in the postoperative period. Figures 5, 6 and 7. Sixteen (16) patients presented higher complications, and eight patients had partial necrosis of the skin flap of the mastectomy. There were no reports of loss of silicone implant.
Potter et al. (2019) 19 conducted a retrospective study (2014 to 2016) with 2,081 patients who underwent immediate mammary reconstruction in the United Kingdom. The average age was 49 years. The average weight of the resecting breast was 390g, with BMI<30 kg/m2 in 1,613 patients (77%). Skin-sparing mastectomy was observed in 1,161 patients (55%), papilla-sparing mastectomy in 486 patients (23%), and another 398 patients (19%) underwent a modified radical mastectomy. A total of 1,638 patients (78%) underwent mammary reconstruction in a single time, and another 453 patients (21%) had the use of a temporary expander. ADM was used in 1,121 patients. In 236 patients, there was a synthetic mesh, and in 436 patients, a de-epithelialized dermal flap was used to make up the submuscular pocket. A 90-day postoperative follow-up revealed that 182 patients (9%) had a loss of silicone implant, 522 patients (25%) were treated for skin infection, and 370 patients (18%) returned to the operating room to correct complications. The study revealed evidence that high BMI and smoking increase the chance of surgical complications in breast reconstructions with silicone implants.
Hanssona et al. (2020)20 concluded that there were more myofibroblasts and neovascularization in biological matrices than synthetic fibers. In biological matrices, the collagen fibers appeared to be aligned in an irregular pattern with parallel and vertical fibers. More giant cells and foreign body reaction and the collagen fibers were loose, well-aligned, and oriented parallel to the implant surface in the synthetic meshes. Synovial metaplasia was seen in most biological and synthetic meshes.
CONCLUSION
The consensus of the data presented showed that the IBRP with synthetic meshes produced aesthetic results comparable to the ADMs, with lower costs and complication rates.
REFERENCES
1. Vardanian AJ, Clayton JL, Roostaeian J, Shirvanian V, Lio A, Lipa JE, et al. Comparison of implant-based immediate breast reconstruction with and without acellular dermal matrix. Plast Reconstr Surg. 2011 Nov;128(5):403e-10e. DOI: https://doi.org/10.1097/PRS.0b013e31822b6637
2. Billon R, Hersant B, Bosc R, Meningaud JP. Acellular dermal matrix and synthetic mesh in implant-based immediate breast reconstruction: current concepts. Gynecol Obstet Fertil Senol. 2019 Mar;47(3):311-6. DOI: https://doi.org/10.1016/j.gofs.2019.01.010
3. Gómez-Modet S, Tejedor L. Synthetic mesh in immediate breast reconstruction. London: IntechOpen; 2020. DOI: http://dx.doi.org/10.5772/intechopen.90884
4. Bertozzi N, Pesce M, Santi P, Raposio E. One-stage immediate breast reconstruction: a concise review. Biomed Res Int. 2017;2017:6486859. DOI: https://doi.org/10.1155/2017/6486859
5. Leal DG. Reconstrução mamária com implantes e tela sintética. Rev Bras Cir Plást. 2018 Jul/Set;33(3):281-92. DOI: https://doi.org/10.5935/2177-1235.2018RBCP0139
6. Ellis HL, Asaolu O, Nebo V, Kasem A. Biological and synthetic mesh use in breast reconstructive surgery: a literature review. World J Surg Oncol. 2016 Abr;14:121. DOI: https://doi.org/10.1186/s12957-016-0874-9
7. Spear SL, Parikh PM, Reisin E, Menon NG. Acellular dermis-assisted breast reconstruction. Aesthetic Plastic Surg. 2008 Mai;32(3):418-25.
8. Nyame TT, Lemon KP, Kolter R, Liao EC. High-throughput assay for bacterial adhesion on acellular dermal matrices and synthetic surgical materials. Plast Reconstr Surg. 2011 Nov;128(5):1061-8.
9. Jacobs J, Salzberg CA. Implant-based breast reconstruction with meshes and matrices: biological vs synthetic. Br J Hosp Med (Lond). 2015 Abr;76(4):211-6.
10. Gschwantler-Kaulich D, Schrenk P, Bjelic-Radisic V, Unterrieder K, Leser C, Fink-Retter A, et al. Mesh versus acellular dermal matrix in immediate implant based breast reconstruction - a prospective randomized trial. Eur J Surg Oncol. 2016 Mai;42(5):665-71. DOI: http://dx.doi.org/10.1016/j.ejso.2016.02.007
11. Pukancsik D, Kelemen P, Gulyás G, Újhelyi M, Kovács E, Éles K, et al. Clinical experiences with the use of ULTRAPRO® mesh in single-stage direct-to-implant immediate postmastectomy breast reconstruction in 102 patients: a retrospective cohort study. Eur J Surg Oncol. 2017 Jul;43(7):1244-51. DOI: http://dx.doi.org/10.1016/j.ejso.2017.01.236
12. Machleidt A, Schmidt-Feuerheerd N, Blohmer JU, Ohlinger R, Kueper J, Von Waldenfels G, et al. Reconstructive breast surgery with partially absorbable bi-component Seragyn® BR soft mesh: an outcome analysis. Arch Gynecol Obstet. 2018 Out;298(4):755-61. DOI: https://doi.org/10.1007/s00404-018-4859-5
13. Gschwantler-Kaulich D, Leser C, Salama M, Singer CF. Direct-to-implant breast reconstruction: higher complication rate vs cosmetic benefits. Breast J. 2018 Set;24(6):957-64. DOI: https://doi.org/10.1111/tbj.13113
14. Remington AC, Gurtner GC, Wan DC, Nguyen D, Momeni A. Identifying risk factors for postoperative major complications staged implant-based breast reconstruction with AlloDerm. Breast J. 2017 Mai;25(4):597-603. DOI: https://doi.org/10.1111/tbj.13299
15. Pompei S, Evangelidou D, Arelli F, Ferrante G. The use of TIGR Matrix in breast aesthetic and reconstructive surgery: is a resorbable synthetic mesh a viable alternative to acellular dermal matrices?. Clin Plast Surg. 2018 Jan;45(1):65-73. DOI: http://dx.doi.org/10.1016/j.cps.2017.08.005
16. Hallberg H, Rafnsdottir S, Selvaggi G, Strandell A, Samuelsson O, Stadig I, et al. Benefits and risks with acellular dermal matrix (ADM) and mesh support in immediate breast reconstruction: a systematic review and meta-analysis. J Plast Surg Hand Surg. 2018 Jun;52(3):130-47. DOI: https://doi.org/10.1080/2000656X.2017.1419141
17. Hallberg H, Elander A, Kölby L, Hansson E. A biological or a synthetic mesh in immediate breast reconstruction? A cohort-study of long-term Health related Quality of Life (HrQoL). Eur J Surg Oncol. 2019 Out;45(10):1812-6. DOI: https://doi.org/10.1016/j.ejso.2019.03.013
18. Bonomi S, Sala L, Gennaro M, Ricci C, Cortinovis U. Skin-reducing mastectomy and direct-to-implant breast reconstruction with submuscular-dermal-mesh pocket. Ann Plast Surg. 2019 Jan;82(1):19-27. DOI: https://doi.org/10.1097/SAP.0000000000001614
19. Potter S, Conroy EJ, Cutress RI, Williamson PR, Whisker L, Thrush S, et al. Short-term safety outcomes of mastectomy and immediate implant-based breast reconstruction with and without mesh (iBRA): a multicentre, prospective cohort study. Lancet Oncol. 2019 Fev;20(2):254-66. DOI: http://dx.doi.org/10.1016/S1470-2045(18)30781-2
20. Hansson E, Burian P, Hallberg H. Comparison of inflammatory response and synovial metaplasia in immediate breast reconstruction with a synthetic and a biological mesh: a randomized controlled clinical trial. J Plast Surg Hand Surg. 2020;54(3):131-6. DOI: https://doi.org/10.1080/2000656X.2019.1704766
1. Private Clinic, Plastic Surgery, Fortaleza, CE, Brazil.
2. Fortaleza General Hospital, Plastic Surgery, Fortaleza, CE, Brazil.
Corresponding author: Francisco Jose Fontenele Bezerra, Rua Barão de Aracati, 444/900, Meireles, Fortaleza, CE, Brazil. Zip Code: 60115-080 E-mail: fco.fontenele@yahoo.com.br
Article received: July 03, 2020.
Article accepted: April 23, 2021.
Conflicts of interest: none
COLLABORATIONS
FJFB Analysis and/or data interpretation, Conception and design study, Conceptualization, Final manuscript approval, Formal Analysis, Methodology, Project Administration, Realization of operations and/or trials, Supervision, Writing - Original Draft Preparation, Writing - Review & Editing
RMGM Conception and design study, Conceptualization, Final manuscript approval, Validation





 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter