

Original Article - Year 2026 - Volume 41Issue 1
Integration of New Biological Therapies into the Brazilian Unified Health System: Logistics Assessment and First Clinical Applications of Legalized Amniotic Membrane Transplantation
Integração de novas terapias biológicas ao Sistema Único de Saúde: Avaliação logística e primeiras aplicações clínicas do transplante legalizado de membrana amniótica
ABSTRACT
Introduction The human amniotic membrane (AM) possesses immunomodulatory and regenerative properties, making it valuable in the treatment of burns and complex skin lesions. In Brazil, Ordinance No. 8,244, issued in September 2025, regulates the processing and clinical use of AM by multi-tissue banks.
Objective To present the initial operational results from implementing this new therapeutic resource in an accredited tissue bank.
Materials and Methods This descriptive study, a preliminary note, evaluated the period from October 7 to November 26, 2025, spanning 50 days. We analyzed both operational (recruitment, processing, and distribution) and clinical data (initial transplants). The processing involved thorough donor screening, antibiotic treatment, handling in a cleanroom with International Organization for Standardization (ISO) 5 laminar flow hood qualification, and microbiological and serological quality control. Tissue release occurred only after consistently negative test results.
Results Dr. Roberto Corrêa Chem Tissue Bank (BTDRCC) harvested 13 AMs and achieved 100% compliance with microbiological and serological standards. The total volume released was 4,157 cm2 (60% distribution), with an average interval from harvesting to release of 12.5 days. The first legally authorized AM transplant in Brazil occurred during this period.
Conclusion Preliminary results validate the operational feasibility and safety of the protocol established by BTDRCC, ensuring 100% microbiological compliance and rapid tissue release. These data form the basis for future investigations into the clinical outcomes and cost analysis of AM use in the Brazilian setting.
Keywords: amniotic membrane; transplants; tissue banks; burns; graft
RESUMO
Introdução A membrana amniótica humana (MA) possui propriedades imunomoduladoras e regenerativas que a tornam valiosa no tratamento de queimaduras e lesões cutâneas complexas. No Brasil, a Portaria n° 8.244, de setembro de 2025, regulamentou seu processamento e uso clínico por bancos de multitecidos.
Objetivo Apresentar os resultados operacionais iniciais da implementação deste novo recurso terapêutico em um banco de tecidos credenciado.
Materiais e Métodos Trata-se de um estudo descritivo, do tipo nota prévia, que avaliou o período de 07 de outubro a 26 de novembro de 2025, ao longo de 50 dias. Os dados operacionais (captação, processamento, distribuição) e clínicos (primeiros transplantes) foram analisados. O processamento envolveu triagem rigorosa de doadoras, tratamento antibiótico, manipulação em sala limpa com fluxo laminar com qualificação 5 da Organização Internacional de Normalização (ISO) e controle de qualidade microbiológico e sorológico. O tecido foi liberado após resultados negativos consistentes.
Resultados O Banco de Tecidos Dr. Roberto Corrêa Chem (BTDRCC) captou 13 MA e alcançou 100% de conformidade microbiológica e sorológica. O volume total liberado foi de 4.157 cm2 (60% distribuídos), com um intervalo médio de 12,5 dias entre captação e liberação. O primeiro transplante legalizado de MA no Brasil foi realizado no período avaliado.
Conclusão Os resultados preliminares validam a viabilidade operacional e a segurança do protocolo estabelecido pelo BTDRCC, garantindo 100% de conformidade microbiológica e liberação rápida de tecido. Estes dados estabelecem a base para futuras investigações sobre os desfechos clínicos e a análise de custos do uso da MA no cenário brasileiro.
Palavras-chave: membrana amniótica; transplantes; bancos de tecidos; queimaduras; enxerto
Introduction
The amniotic membrane (AM), an avascular structure derived from the placenta, has been extensively studied due to its immunobiological and regenerative properties. It contains an extracellular matrix that is rich in collagen types I, III, IV, V, and VII, as well as fibronectin, laminin, and proteoglycans. These components contribute to its anti-inflammatory, immunomodulatory, antimicrobial, and epithelizing properties.1 The AM also has epithelial and mesenchymal cells with a pluripotency profile and low immunogenicity, allowing its allogeneic use with minimal riskof rejection. Clinical and experimental studies demonstrated that AM accelerates reepithelialization, reduces pain, minimizes the formation of hypertrophic scars, and acts as a temporary biological barrier in complex wounds, especially burns and extensive skin lesions.2
In Brazil, clinical AM use was restricted to experimental protocols or import on demand until September 2025. The publication of Ordinance GM/MS No. 8,244, on September 25, 2025, enabled the regulation of AM processing and clinical application by multitissue banks.3 This ordinance, by explicitly including AM transplantation in the Brazilian Unified Health System (Sistema Único de Saúde, SUS) procedure table, enabled the legalized and nonexperimental activity of institutions accredited in the Brazilian National Transplant System. This regulation is a determining factor, as it elevates this therapy as an established treatment standard, with defined procurement and quality control protocols.
In light of the new regulations, Dr. Roberto Corrêa Chem Tissue Bank (BTDRCC) at the Santa Casa de Porto Alegre, adapted its infrastructure to incorporate the processing of AM. This article presents the initial operational results of this implementation, evaluating the immediate logistical and clinical impact of this new therapeutic resource.
Materials and Methods
This is a descriptive study, characterized as a preliminary note, covering the period from October 7, 2025, to November 26, 2025, spanning 50 days. The data analyzed were operational (collection, processing, quality control, distribution) and clinical (recording of the first transplants). This study is in accordance with the opinion approved by the Research Ethics Committee of the institution, with the CAAE: 59854922.5.0000.5335. The AM donors signed the informed consent form before delivery.
The inclusion criteria encompassed pregnant women aged 18 to 35 years, undergoing elective cesarean sections, at a gestational age of 34 to 40 weeks, with negative serology testing, no obstetric complications, and no active infectious or autoimmune comorbidities.
The exclusion criteria were premature rupture of membranes for over 12 hours, labor longer than 24 hours, presence of chorioamnionitis, endometritis, fever or signs of infection, illicit drug use, history of hematological malignancies, systemic autoimmune diseases, suspected or confirmed congenital infections, fetal distress, and any placental abnormality that compromised tissue integrity.
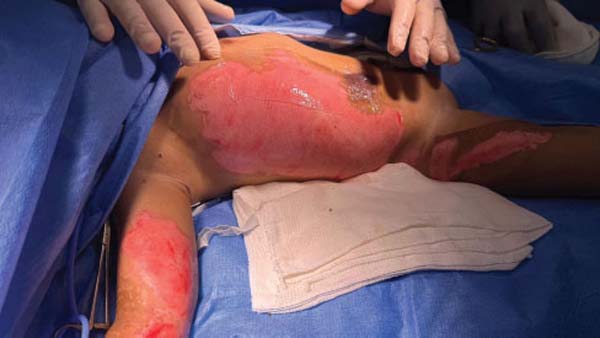
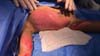
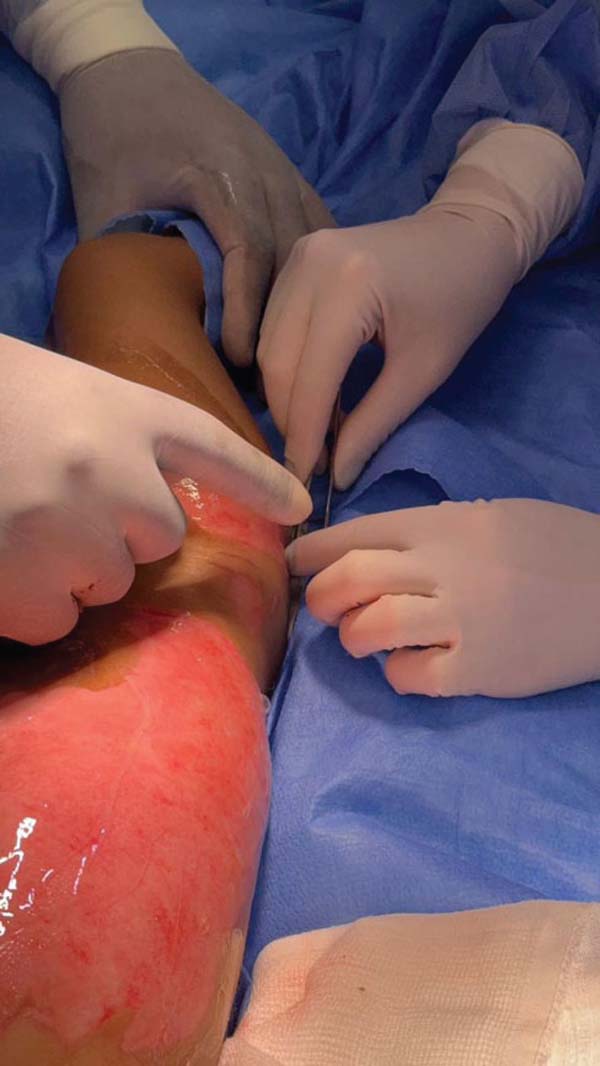
The entire amniotic membrane harvesting process was performed in an operating room. Clean rooms, hand and forearm degerming, surgical gowning, and the use of sterile materials were part of the tissue harvesting routines. After the cesarean section, the amniotic membrane was aseptically separated from the placenta using a finger-like technique, properly packaged, and transported to the Biovascular Rehabilitation Center at BTDRCC under refrigeration (2–8 ° C). ►Fig. 1 illustrates these processes.
To process AM, there are three phases. First, for prophylactic treatment, AM is placed in a solution containing a mixture of antibiotics and kept refrigerated (2 to 8 °C) for at least 8 hours. The day after antibiotic treatment, the AM tissue undergoes phase 1 processing, during which the tissue is washed with sterile 0.9% saline solution to remove excess antibiotics and placed in glycerol solution with a higher concentration (95%). Phase 2 involves trimming the edges of the membrane tissue sheets, packaging, and measuring the tissue in cm2. The tissue storage temperature is 2 to 8 °C. All materials and instruments used in the processing and contacting with AM were sterile. The handling and exposure of tissues throughout processing occurred in a laminar flow hood with an International Organization for Standardization (ISO) 5 air quality qualification.
Processed AM was released and made available for therapeutic use after macroscopic evaluation and negative microbiological test results. At each stage of processing, fragments were collected for microbiological analysis. The fragments were submerged in liquid culture media, including tryptone soya broth (TSB) and thioglycolate, to provide growth and retrieval of aerobic bacteria, anaerobic bacteria, and fungi potentially present in the tissue.
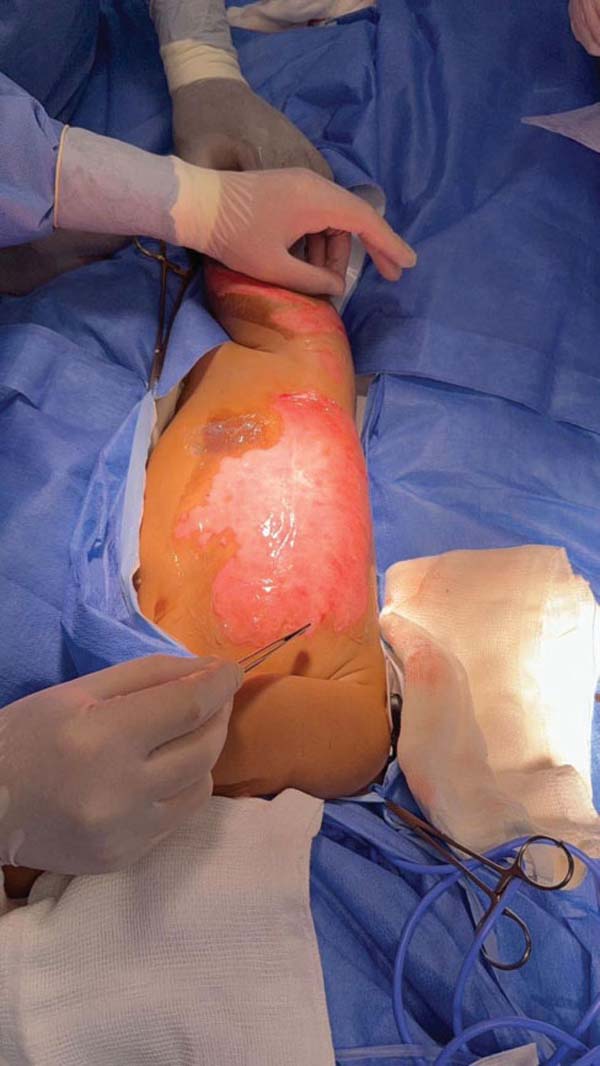
Additionally, serological tests were performed for cytomegalovirus, toxoplasmosis, human immunodeficiency virus (HIV), hepatitis C virus (HCV), hepatitis B virus (HBV, HBc, and HBsAg), human T-cell lymphotropic virus (HTLV I and II), syphilis, and Chagas disease. The presence of HIV, HCV, and HBV were confirmed by molecular analysis. Tissue release for clinical use was contingent upon obtaining consistently negative results in all contamination and serological tests. The approved tissues were made available for distribution through the Brazilian National Transplant System. ►Fig. 2 shows the biomaterial available for transplantation.
The production data (collection, area processed, area distributed) have a descriptive quantitative nature. Absolute and relative frequencies for categorical variables (quality control results) and descriptive statistics (mean and range) for the time interval between collection and release were calculated. No statistical inference tests were applied, given the initial, descriptive nature of this preliminary note.
Results
The operational capacity of BTDRCC over the analyzed 50 days is shown in ►Table 1.
| Operational variable | Index value |
|---|---|
| AM harvesting | 13 |
| Microbiological and serological compliance | 100% |
| Total volume of released tissue | 4,157 cm2 |
| Total volume of distributed tissue | 2,485 cm2 |
| Percentage of distribution | 60% |
| Average interval between harvesting and release | 12.5 days |
| Supported institutions | 4 |
Abbreviation: AM, amniotic membrane.
The BTDRCC achieved a 100% microbiological and serological compliance rate in the 13 AM samples collected. This result was obtained within an average interval of 12.5 days between collection and release.
The total volume of tissue released was 4,157 cm2, of which 2,485 were allocated to four different hospitals.
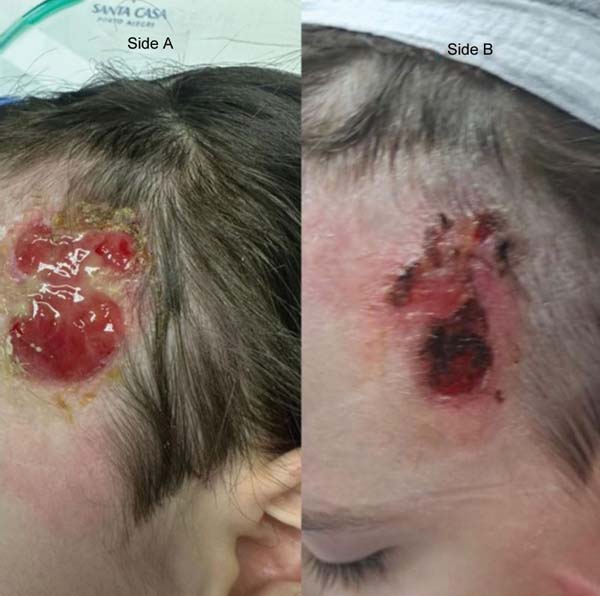
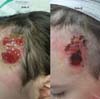
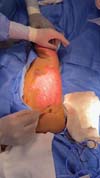
The first legally sanctioned AM transplant occurred on November 5, 2025, at Dom Vicente Scherer Hospital, in an adult patient with a complex skin lesion. On the same date, the biomaterial was applied successfully to a pediatric burn victim at the Santo Antônio Children’s Hospital. ►Figs. 3–4 to 5 show AM use in these two initial cases.
Discussion
The use of AM as a legalized transplant in Brazil represents a significant advance in the incorporation of biological therapies into the national surgical arsenal. Ordinance GM/MS No. 8,244/2025 permits authorized tissue banks to collect, process, and distribute AM for therapeutic purposes, beyond experimental use.3 As such, the tissue can be employed as a biological graft in standard clinical procedures, given that technical and legal support is available and the criteria for microbiological quality, traceability, and documentation compliance are satisfied.
Unlike previous uses restricted to research protocols or occasional imports, legalized transplantation integrates AM into regular healthcare practice, expanding access to a biomaterial with anti-inflammatory, epithelizing, and immunomodulatory properties, especially useful in burns, complex wounds, and skin reconstructions.4 This regulatory change not only strengthens patient safety and procedural standardization but also opens up opportunities for multicenter studies and large-scale cost-effectiveness analyses.
Harvesting 13 samples and releasing over 4,157 cm2 in just 50 days demonstrates the robustness of the collection and processing protocol, as well as the high unmet clinical demand for AM. The high utilization rate (60% of the distributed stock) corroborates the current need for biological dressings with its properties. Studies show that its early application in partial-thickness burns can reduce the depth of the lesion, modulate the scarring response, and decrease the need for surgical debridement and autologous grafting, generating economic and morbidity benefits for patients.5
Additionally, AM provides a covering that relieves pain and provides an ideal environment for regeneration.6
The 100% success rate in quality control (absence of contamination) in this study attests to the effectiveness of the protocols, from donor screening to final processing. Microbiological safety is a cornerstone of transplant therapy and a prerequisite for the widespread clinical use of AM.
The pioneering application in adult and pediatric patients highlights the clinical versatility of the tissue. The pediatric case particularly underscores the importance of AM in the management of burns in children, where minimizing pain and accelerating healing are critical to preventing contracture and optimizing long-term functional and aesthetic outcomes.
Previous studies have consistently demonstrated that AM applications accelerate the healing process and reduce the need for more invasive procedures.7 As a biological dressing, AM has proven cost-effective in the treatment of burns, promoting angiogenesis, collagen organization, and epithelial regeneration.8
A comparative analysis between initial data from AM and consolidated indicators for allogeneic skin at BTDRCC suggests a potentially relevant operational difference. While the average processing and release time for skin is around 45 days, the observed interval for AM was approximately 12.5 days. Furthermore, the discard rate due to microbiological contamination in skin is historically close to 25%, whereas in initial AM procurements, the discard rate was 0%. Although these data do not yet allow for definitive conclusions, they point to a potentially greater microbiological and operational efficiency, which could represent a promising path to expanding the supply of biological tissues in contexts of scarcity. Prospective and multicenter studies will be fundamental to validate these observations and establish more robust comparative parameters.
Limitations of this preliminary note include the absence of clinical follow-up data (healing time, grafting rate, scar quality, complication rate). However, the objective of validating the logistics implementation and production capacity has been achieved successfully. Our team’s next steps include prospective monitoring of clinical outcomes and a rigorous economic analysis. Cost-benefit analysis is key to justifying the expansion and sustainability of the program within the Brazilian National Transplant System. It is expected that the reduction in morbidity, length of hospital stays, need for reinterventions, and cost reduction will consolidate AM as an efficient therapeutic option in the Brazilian context.
Conclusion
During the initial 50 days of implementation, the established protocol resulted in the release of 4,157 cm2 of AM suitable for grafting, with 100% microbiological compliance. These preliminary results demonstrate the operational feasibilityandsafety of the process, while also highlighting the innovative use of fetal adnexal tissue that is often discarded after birth.
The BTDRCC’s experience demonstrates that AM can be a strategic resource in the care of patients with complex wounds, especially burns, allowing for integrated action between plastic surgery, obstetrics, burn teams, and other specialties involved in these high-morbidity care situations. This interprofessional collaboration strengthens the care pathway from pregnancy and childbirth, with planned and safe tissue procurement, to its clinical application in highly complex scenarios.
The widespread dissemination of this program, both among healthcare professionals and the general population, is fundamental to expanding knowledge about the safety and benefits of AM and, consequently, stimulating an increase in the number of donors. By transforming a tissue with no frequent use into a biological graft of high therapeutic value, the healthcare system incorporates an effective and socially relevant alternative, with a potential impact on reducing morbidity, length of hospital stays, and the need for additional surgical procedures. These preliminary data validate the operational feasibility and safety of the therapy, establishing the basis for future investigations into clinical outcomes and cost analysis of AM use by plastic surgeons in Brazil.
References
1. https://doi.org/10.3390/pharmaceutics15030748
2. https://doi.org/10.1097/TA.0000000000003050
3. Brasil. Ministério da Saúde. Portaria n° 8.244, de 25 de setembro de 2025. Atualiza as Tabelas de Serviço Especializado e habilitações do Cadastro Nacional de Estabelecimentos de Saúde (CNES) e inclui e altera Procedimentos, relacionados a Transplante Pediátrico e Banco de Multitecidos na Tabela de Procedimentos, Medicamentos, Órteses, Próteses e Materiais Especiais do SUS (Tabela de Procedimentos do SUS) Brasília: Diário Oficial da União; 2025. Available from: https://www.in.gov.br/web/dou/-/portaria-gm/ms-n-8.244-25-de-setembro-de-2025-658712781
4. https://doi.org/10.1016/j.bjps.2022.08.028
5. https://doi.org/10.1007/s12015-025-10892-x
6. https://doi.org/10.3390/jcm14124272
7. https://doi.org/10.1111/srt.13860
8. https://doi.org/10.1016/j.burns.2025.107522
1. Dr. Roberto Corrêa ChemHumanTissue Bank, Hospital Santa Casa de Porto Alegre, Porto
Alegre, RS, Brazil
2. Hospital Santa Casa de Porto Alegre, Centro Histórico, Porto Alegre, RS, Brazil
3. Maternity Ward, Hospital Santa Casa de Porto Alegre, Centro Histórico, Porto Alegre,
RS, Brazil
Financial Support
Address for correspondence Eduardo Mainieri Chem, Hospital Santa Casa de Porto Alegre, Banco de Tecidos Humanos Dr. Roberto Corrêa Chem, Rua Professor Annes Dias, 295, Centro Histórico, Porto Alegre – RS, Brazil. CEP: 90020-090 (e-mail: eduardo@chem.com.br).
Artigo submetido: 10/12/2025.
Artigo aceito: 16/12/2025.
Conflict of Interests The authors have no conflict of interests to declare.
Editor-in-Chief: Dov Charles Goldenberg.




 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter