

Articles - Year 2002 - Volume 17 -
Effect of Ginkgo Biloba Extract on Skin Flap Survival in Rats
Efeito do Extrato de Ginkgo Biloba na Sobrevida de Retalhos Cutâneos em Ratos
ABSTRACT
The partial loss of skin flaps is still responsible for a substantial amount of the morbidity in aesthetic and reconstruction surgeries. Many treatments hare been proposed in order to increase flap survival rates. The present study aims to assess if Ginkgo Biloba Extract (GBE) is effective in improving the survival rate of randomized skin flaps. To that purpose, an experimental model of a flap from the back of a rat, described by McEarlane and modified by Hammond, was used. The animals were treated for 5 days with GBE (100 mg/kg, ip, 1 x day) or normal saline solution (Sodium Chloride 0.9%, ip, 1 x day) -control group - and sacrificed on the sixth day in order to assess results. The average flap survival area in the control group was 2.702 ± 0.195 cm2 (n=9), and 5.490 ± 0.283 cm2 (n=8) in the group treated with GBE. Our results showed that animals treated with GBE had a significant increase (p<0.00l) in the survival rate of randomized skin flaps, suggesting that the drug may play a role on skin flaps with distal vascular suffering.
Keywords: Ginkgo Biloba; surgical flaps; tissue survival; skin; ischemia.
RESUMO
A perda parcial de retalhos cutâneos continua sendo responsável por uma parcela significativa de morbidade nas cirurgias estéticas e reconstrutoras. Várias terapias já foram propostas com o objetivo de aumentar a taxa de sobrevida dos retalhos. O presente trabalho tem como objetivo avaliar se o Extrato de Ginkgo Biloba (EGB) é efetivo na melhoria da taxa de sobrevida de retalhos cutâneos randomizados. Para tanto, utilizou-se o modelo experimental de retalho de dorso de rato descrito por McFarlane e modificado por Hammond. Os animais foram tratados por 5 dias com EGB (100 mg/kg, ip, 1x dia) ou solução salina (SF 0,9%, ip, 1x dia) - grupo controle - e sacrificados no sexto dia para a avaliação dos resultados. A média da área de sobrevida do retalho no grupo controle foi de 2,702 + 0,195 cm2 (n=9) e no grupo tratado com EGB foi de 5,490 + 0,283 cm2 (n=8). Nossos resultados mostraram que o tratamento dos animais com EGB aumentou de forma significativa (p< 0,001) a taxa de sobrevida dos retalhos cutâneos randomizados, sugerindo um possível papel para essa droga no manuseio de retalhos cutâneos com sofrimento vascular distal.
Palavras-chave: Ginkgo Biloba; retalhos cirúrgicos; sobrevivência de tecidos; pele; isquemia
Ischemia and subsequent necrosis of flaps remain an important clinical problem that increases morbidity in surgical reconstruction procedures. Despite extensive research on the utilization of different strategies to improve flap survival(1), the pathophysiological process of flap ischemia has not been totally explained, and therefore requires both new clinical and experimental studies.
The various treatments used for improving the survival of randomized skin flaps have focused on improving blood flow and tissue oA)Tgenation, therefore decreasing metabolic demands and blocking the reperfusion lesion induced by ischemia(2, 3).
Various inflammation mediators have been thought to be associated to the process lesion induced by ischemia, anl0ng which: prostaglandins, leucotrienes, platelet activating factor - PAF, nitric oxide, free radicals, growth factors and various other cytokines(4, 5, 6, 7, 8). During the process, endothelial cells seem to play an important role because of their location on the interface between blood and tissues, and they are anlong the first elements to take action when the partial pressure of oxygen blood varies. These cells have various major functions: regulating the release of anti-thrombotic molecules, synthesizing vasoactive molecules and compounds that regulate the activity of leucocytes, platelets and smooth muscle cells, in addition to working as a selective barrier and secreting various metabolites(9) .
Ginko Biloba Extract (GBE) has been widely used for treating many vascular conditions comprising ischemic events such as cerebrovascular failure and peripheral vascular failure(1O). It has also been used experimentally for treating intestine(11), cardiac(12) and retina(13) ischemic lesions. GBE protects the endothelial from the decrease in ATP induced by hypoxia(14), works as an important free radical depurator(l5) and is an antagonist of Platelet Activating Factor - PAF(l6). It also seems to play an important role in inhibiting the production of itric Oxide(17).
OBJECTIVES
The objective of the present study was to investigate the possible role of GBE on the survival of randomized skin flaps in rats, by comparing it with the control group.
MATERIALS AND METHODS
EXPERIMENTAL MODEL
Twenty Wistar rats (180-200 g) were divided into two groups of 10 animals each, from which a back skin flap was randomly performed. The model described by McFarlane(18) and modified by Hanlmond(l9) was used. All procedures were performed under anesthesia and afterwards all animals were put in separate cages and received water and food ad libitum. The caudal base of all flaps measured 2 x 7 cm (Fig. 1). A triangular 2 cm skin segment was made on the top of the flap (Fig. 2). A panniculus carnosus was taken from this skin segment in order to make its grafting at the base of the flap feasible (Fig. 3). The surgical wound was closed (Fig. 4) and the flap sutured over the closed area, remaining totally isolated from the subjacent bed (Fig. 5).
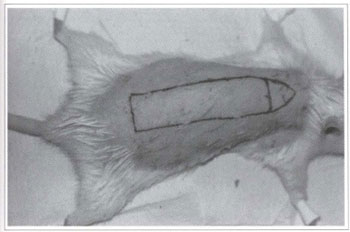
Fig. I - Marking of the randomized rat back skin flap.
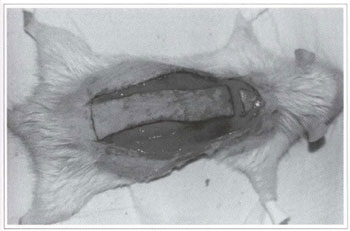
Fig. 2 - Skin incision of the randomized rat back skin flap.
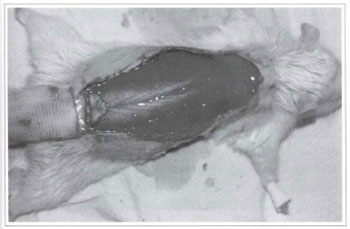
Fig. 3 - Grafting of the triangular segment on the base of the randomized rat back skin flap.
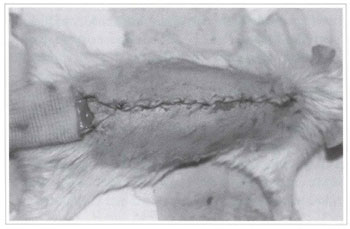
Fig. 4 - Closing of the donor area of the randomized rat back skin flap.
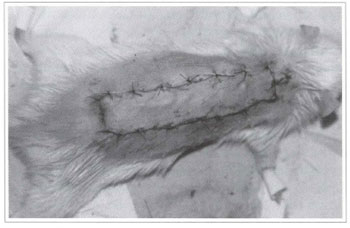
Fig. 5 - Fixation of the randomized rat back skin flap on the closed donor area.
EXPERIMENTAL GROUPS
Treatments began 24 hours after the flaps were made and continued for 5 days. Animals were sacrificed on the sL'::th day in order to assess results.
- Group I (control): animals received 5 ml of 0.9%, intra-peritoneal saline, once a day, for 5 days.
Group II: animals received intra-peritoneal GBE (l00 mg/kg), once a da); for 5 days.
RESULTS ASSESSMENT
The feasible flap area was measured on measuring wax paper and results were presented in cm2 of feasible tissue.
STATISTICAL ANALYSIS
Results were presented as average ± average standard error (ASE). The averages of the control group and the group treated with GBE were assessed using analysis of variance, followed by Student's t test, considering p
RESULTS
None of the flaps presented any dehiscence, inflammation, infection or hematoma that could affect the final result. There was one surgical death in group I and two in group II, all of which occurred before beginning treatments.
Ater 24 hours, flaps presented a purplish color on their distal end, although without a well-defined limit between the feasible and non-feasible area. Three days after treatment, flaps had a more clearly defined limit of the level of necrosis, most of which on the total width. On the sixth day after flaps were made, and when the animals were sacrificed to check results, the degree of necrosis was totally defined, and there was a clear limit between the feasible and non-feasible area (Fig. 6).
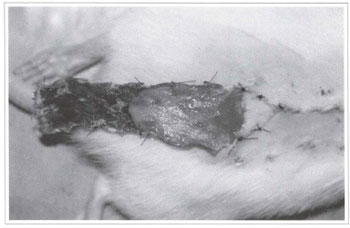
Fig. 6 - Clear limit between the feasible and non feasible area, with total width necrosis of the distal portion of the randomized rat back skin flap.
Treating rats with GBE for 5 days significantly increased the flap survival area when compared to the control group (p <0.001) (Figs. 7 and 8). The average flap survival in the control group was 2.702 ± 0.195 cm2 (n=9), and 5.490 ± 0.283 cm2 (n=8) in the group treated with GBE (Table I) (Fig. 9).
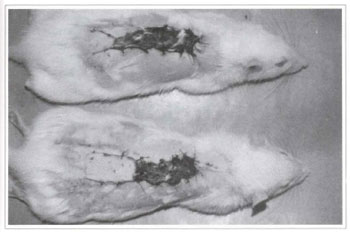
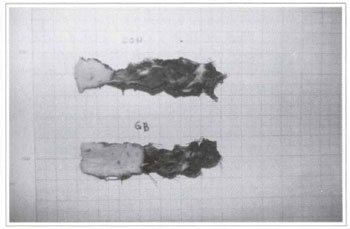

DISCUSSION
In the present study, we replicated the experimental model on acute loss of randomized skin flap described by McFarlane(l8) and modified by Hammond(l9). The study verified tlut Gingko Biloba Extract is efficacious for preventing flap necrosis, since its intra-peritoneal administration for 5 days significantly increased (p
The modified experimental model was chosen due to the following facts i) the direct comparison ofthe standard of necrosis and flap survival in rats showed that when the distal portion of the flap is fastened directly on the donating bed, it is significantly influenced by the grafting effect of the receptor bed; ii) as flaps are generally thicker in human beings, a recently elevated flap depends initially on the metabolic support of its pedicle, and the grafting effect is not as important on flap survival(l8); iii) the strategy of separating the flap from the bed with plastic or silicone strips used by some authors(l) is an inadeguate solution for the problem, and cases of intense inflammatory process, exudate, infection, dehiscence and flap loss have been observed(l9).
As the surgical wound closes below the flap, metabolic support is limited to the pedicle. This allows for a controlled model of acute flap loss, and may be considered a strict flap survival test, similar to those mat occur clinically when a flap is inserted in an area severely irradiated or with excessive scar tissue, in which revascularization may be minimum.
Ginkgo Biloba (Ginkgoaceae) is an ancient Chinese plant that has been cultivated for centuries because it is considered sacred due to its medicinal effects. The extract is prepared with dry leaves from tlle plant. The Ginko Biloba Extract (GBE) is standardized so as to contain 24% of ginkgoflavanoids, 6% of terpenes (ginkgolides and bilobalides) and other uncharacterized compounds(1O).
The effect of GBE on increasing flap survival may be attributed to a series of factors. We believe the most important to be: i) the powerful depurating role of the free radicals of the flavanoids present in GBE, in addition to the inhibiting effect on the formation of free radicals attributed to its terpenic portion(ll, 12, 13, 15); ii) ginkgolide B, present in the terpenic portion, acting as an important Platelet Activating Factor inhibitor - PAF(l6); iii) the protecting effect of the terpenic portion, mainly of bilobalides, on the mortality of endothelial cells induced by hypoxia, as well as the prevention of the decrease of ATP induced by hypoxia(14); and iv) a inhibition of messenger RNA, expression of the O-synthase enzyme by GBE, blocking the synthesis of Nitric Oxide(17).
GBE may act as a sum of tlle actions of its various components, and one component may augment the effect of another. Similar to other inflammatory processes(20), Nitric Oxide formation may be the final event in the distal loss of the flap, which is stimulated by a series of cytokines and growth factors, PAF among them, strongly inhibited by ginkgolide B present in me terpenic portion of GBE.
We believe that GBE may be a useful drug for handling skin flaps with distal suffering, although me mechanism by which it works needs furmer explanation, so we may define its ideal indication more safely.
CONCLUSION
Treating animals with Gtnko Biloba Extract was capable of Significantly increasing (p<0.001) the survival area of randomized skin flaps in the experimental model of rat back flap, described by McFarlane(18) and modified by Hammond(19). Further experimental studies should be carried out to better explain the mechanism by which GBF protects flap suffering.
REFERENCES
1. Knight KR. Review of postoperative pharmacological infusions in ischemic skin flaps. Microsurgery 1994; 15:675-84.
2. Emery EM, Kodey TR, Bomberger RA, et al. The effect of nifedipine on skin flap survival. Plast Reconstr Surg. 1990; 85:61-4.
3. Hawkes IS, Young CMA, Cleland LG. Ischemia reperfusion injury in pedicle skin flaps in the pig: lack of protective effect of SOD and allopurinol. Br J Plast Surg. 1989; 42:668-71.
4. Kirshner RE, Evfe BS, Hoffman LA, et al. Ischemia reperfusion injury in myocutancous flaps: role of leukocytes and leukotrienes. Plast Reconstr Surg. 1997; 99:1485-95.
5. Stotland MA, Kerrigan CL. The role of Platelet-Activating Factor in Musculocutaneous flap reperfusion injury. Plast Reconstr Surg. 1997; 99:1989-99.
6. Urn SC, Suzuki S, Tovokuni S, et al. Involvemente of Nitric Oxide in survival of random pattern skin flap. Plast Reconstr Surg. 1998; 101:785-92.
7. Banbury I, Siemionow M, Porvasnik S, et al. Improved perfusion after subcritical ischemia in muscle flaps treated with vascular endothelial growth factor. Plast Reconstr Surg. 2000; 7:1541-6.
8. Most D, Hovt J, Sibley RK, et al. Parenchymal cytokine expression precedes clinically observed ischemia in dorsal flaps in the rat. Plast Reconstr SUrg. 1996; 5:856-61.
9. Palombo JD, Blackburn GL and Forse RA. Endothelial cell factors and response to injury. Surgery. 1991; 173:505-18.
10. De Feudis FV. Ginkgo biloba extract (Egb 761): Pharmaccological activities and clinical applications. Paris: Editions Scientifiques Elsevier, 1991.
11. Colak O, Sahin A, Alatas O, et al. The effect of Ginkgo biloba on the activity of catalase and lipid peroxidation in experimental strangulation ileus. Int J Clin Fab Res. 1998; 28:69-71.
12. Pietri S, Seguin JR, d'Arbignv P, et al. Ginkgo biloba extract (Egb 761) pretreatment limits free radical-induced oxidative stress in patients under-going coronary bypass surgery. Cardiovasc Drugs Thcr. 1997; 11:121-31.
13. Szabo MF, Droy-Lefaix MT, Doly M, et al. Free radical-mediated effects in re perfusion injury: a histologic study with superoxide dismutase and EGB 761 in rat retina. Ophthalmic Res. 1991; 23:225-34.
14. Janssens D, Michiels C, Delaive F, et al. Protection of hypoxia-induced ATP decrease in endothelial cells by ginkgo biloba extract and bilobalide. Biochcm Pharmacol. 1995; 50:991-9.
15. Seif-El-Nasr M, El-Fattah AA. Lipid peroxide, phospholipids, glutathione levels and superoxide dismutase activity in rat brain after ischaemia: effect of ginkgo biloba extract. Pharmacol Res. 1995; 32:273-8.
16. Guinor P, Caffrey E, Lambe R, et al. Tanakan inhibits platelet-activating-factor-induced platelet aggregation in healthy male volunteers, Haemostasis. 1989; 19:219-23.
17. Varga F, Bodi A, Ferdinandy P, et al. The protective effect of EGb 761 in isolated ischemic/reperfused rat hearts: a link between cardiac function and nitric oxide production. J Cardiovasc Pharmacol. 1999; 34:711-7.
18. McFarlane RM, He Young G, and Heniy RA. The design of a pedicle flap in the rat to study necrosis and its prevention. Plast Reconstr Surg. 1965; 35:177-9.
19. Hammond DC, Brooksher RD, Mann R), et al. The dorsal skin-flap model in the rat: factors in-ftucncinn survival. Plast Reconstr Surg. 1993: 91:316-21.
20. Souza-Filho MVP, Lima MVA, Pompeii NLV1L, Ballejo G, Cunha FQ, Ribeiro RA. Involvenient of Nitric Oxide in the Pathogenesis of Cydophos-phamide-Induced Hemorrhagic Ostitis. Am J Pathol 1997; 150:247-56.
1. Medical Resident - Specialist by SBCP; Microsurgery and Reconstruction Plastic Surgen Service - Instituto National do Câncer - INCA.
2. Senior Member of SBCP; Head of the Microsurgery and Reconstruction Plastic Surgery Service - INCA.
3. Assistant Physician - Head of SBCP; Head of the INCA Microsurgery and Experimental Surgery Training Center.
4. Medical Resident; Microsurgen and Reconstruction Plastic Surgery Service - Instituto National do Câncer-INCA.
Address for correspondence:
Marcus Vinicius Ponte de Souza Filho, MD
Pça. da Cruz Vermelha, 23 - 8º and. - Centro
22230-130 - Rio de Janeiro - R]
Brazil
Phone: (55 211 2506-6087
e-mail: mvponte@ig.com.br
INCA - Servico de Cirurgia Plástica Reconstrutora c Microcirurgia


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter