

Original Article - Year 2011 - Volume 26 -
Results of keloid treatment with surgery and imiquimod 5% cream: a prospective study
Resultados do tratamento das cicatrizes queloideanas com cirurgia e imiquimode 5% creme: um estudo prospectivo
ABSTRACT
Introduction: Keloid treatment represents a therapeutic challenge, often with disappointing results. More effective adjuvant therapies are necessary to reduce the high rates of recurrence. This research is aimed at assessing the treatment and recurrence of keloid scars after the use of a combined treatment modality comprising the application of imiquimod 5% cream and surgery. Methods: A prospective study that assessed 14 patients with a total of 25 keloids, with a minimum follow-up of 6 months, was carried out. Intralesional excision was performed, and the application of imiquimod 5% cream was initiated on the day of surgery and was continued for two months. Scar assessment was carried out by means of an assessment scale (patient and an observer - Head of the Plastic Surgery Service) and by observing signs of recurrence and scar size during the pre- and postoperative periods. Results: At the 6-month follow-up, there was an improvement of the scar as rated by patients in 72% of the cases and as rated by the observer in 88% of the cases, with a recurrence rate of 8% and significant scar area reduction (p=0.0053). Conclusion: The use of imiquimod 5% cream after keloid resection surgery significantly reduced recurrence in the 6-month postoperative assessment.
Keywords: Keloid. Recurrence. Aminoquinolinaes.
RESUMO
Introdução: O tratamento do queloide representa um desafio terapêutico, com resultados muitas vezes frustrantes. Terapias adjuvantes mais eficazes são necessárias para reduzir as altas taxas de recidivas. Essa pesquisa é baseada em uma modalidade conjunta com a aplicação de imiquimode 5% creme associada à cirurgia, tendo como objetivo a avaliação do tratamento e recidiva da cicatriz queloidiana. Método: Estudo prospectivo que avaliou 14 pacientes em um total de 25 queloides, com um seguimento mínimo de 6 meses. Foi realizada excisão intralesional e iniciada a aplicação do imiquimode 5% creme no dia da cirurgia e mantido por dois meses. A avaliação da cicatriz foi analisada por meio de uma Escala de Avaliação (paciente e um observador - regente de serviço de Cirurgia Plástica), sinais de recidiva e também do tamanho da cicatriz no pré e pós-operatório. Resultados: Após o acompanhamento de 6 meses, houve melhora da cicatriz por parte do paciente em 72% e para o observador em 88%, tendo uma taxa de recidiva de 8% e redução da área cicatricial (p=0,0053). Conclusão: O uso do Imiquimode 5% creme no pós-operatório da cirurgia de ressecção do queloide diminuiu significativamente sua recidiva na avaliação de 6 meses.
Palavras-chave: Queloide. Recidiva. Aminoquinolinas.
Hypertrophic and keloid scars are fibroproliferative disorders arising from abnormal wound healing due to alteration of cell migration and proliferation, inflammation, increased synthesis of cytokines and extracellular matrix proteins, and remodeling of the synthesized matrix1. Keloids differ from hypertrophic scars because the scar tissue exceeds the limits of the causative lesions, without regression or with a tendency to continue progressing after 6 months of development2,3. Commonly encountered in surgical practice, keloids have a reported incidence that varies from 5% to 16% for at-risk populations, including blacks, Hispanics, and Asians4.
Many treatment regimens such as intralesional corticosteroid therapy, compression, laser therapy, and cryosurgery have been used; however, the outcomes are inconsistent5. Isolated surgical excision may be associated with recurrence rates of 45% to 100%4,6. In combination with radiotherapy, isolated surgical excision is considered to be the most effective treatment for severe keloids, according to international clinical recommendations for the management of scars7. However, van de Kar et al.5 suggest that surgical excision combined with radiotherapy should be used as a last resource in the treatment of keloids resistant to therapy.
When topically applied, imiquimod 5% cream acts an immune response modifier that, with the induction and activation of natural killer cells, macrophages and Langerhans cells, induces the synthesis and local release of cytokines, including interferon (IFN)-α; IFN-γ; tumor necrosis factor-α; and interleukins 1, 6, 8, and 12. IFN-α and IFN-γ fibroblasts dose-dependently inhibit the production of collagen. IFN-γ decreases both collagen production and glycosaminoglycans. Based on the ability of IFN-α2b to reduce the recurrence of keloids, this study aims to assess the outcomes of combined treatment of keloids with surgery and imiquimod 5% cream.
METHODS
Fourteen patients with 25 keloids who were treated at the medical facilities of the Plastic Surgery Service were included in the study if they met the following criteria: presence of a wound that exceeded the scar edges and that did not regress spontaneously or progressed after 6 months of treatment; patients who did not receive treatment for two months; presence of a scar for at least one year; scars that could be primarily excised and sutured; and vulnerable individuals, such as children and adolescents, accompanied by their legal representatives (Table 1).
All patients signed a consent form. The study excluded patients who had not complied with the treatment protocol; patients for whom histological analysis revealed a diagnosis other than keloid scar; pregnant and breastfeeding women; heart disease patients; transplant patients; immunosuppressive drug users; patients with infection close to the scar; or those with scars requiring more than one surgical procedure for total scar excision.
All keloids were intralesionally excised under local anesthesia (Xylestesin 2% with adrenaline 1:200,000; Cristália). Hemostasis control was maintained with electrocautery, followed by primary closure with colorless nonabsorbable thread subdermally in order to reduce tension and cutaneous suture with nonabsorbable black thread (Ethilon nylon; Ethicon). For ear defects, only Ethilon skin sutures were applied. The skin suture was removed after 14 days.
Application of imiquimod 5% cream was initiated in all patients on the day of surgical excision and continued for two months. At the 6 month follow-up, the patient and one observer who did not participate in any surgery (the Head of the Plastic Surgery Service from BSPS) subjectively assessed the scar by means of pre- and postoperative photographs (Figures 1 to 3) by using an assessment scale. Both the patients' and the observer's scales contained items that were rated on a scale of 1 to 10, where 1 indicated "resembling normal skin" and 10, "the worst injury possible". For the observer, the scale items were vascularization, pigmentation, surface, and overall impression; for patients, the items were pain, itching, color, hardness, thickness, roughness, and overall impression. The patients were questioned about the scar's appearance with the options of "better", "the same", "or worse", and whether the treatment was considered successful or not.
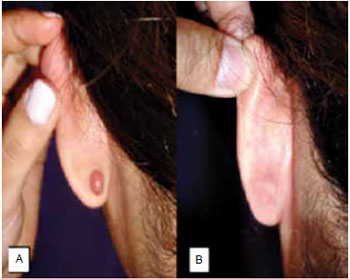
Figure 1 - A: Pre-operative image of a patient with a keloid on the left earlobe; B: After treatment with surgical excision and imiquimod 5% cream at 24 weeks.
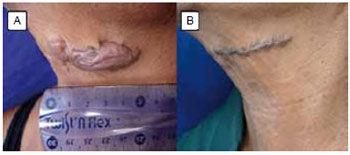
Figure 2 - A: Pre-operative image of a patient with a keloid in the cervical region; B: After treatment with surgical excision and imiquimod 5% cream at 24 weeks.
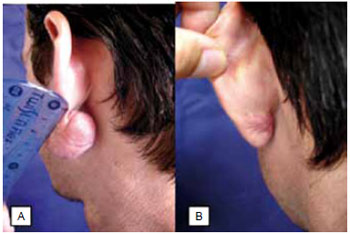
Figure 3 - A: Pre-operative image of a patient with a keloid on the left earlobe; B: After treatment with surgical excision and imiquimod 5% cream at 24 weeks.
We conducted a descriptive analysis of patient data, keloid characteristics during the postoperative period, and scar assessment on week 24 after the surgery. A paired t-test was applied to assess scar areas pre- and postoperatively, setting the significance level at 5%. The signs of recurrence were assessed by the observer by using a scale of crosses (+/++/+++ or ++++) (1-4), based on the criteria of scar elevation, extension beyond the original surgical edges, and absence of recurrence (zero), i.e., a scar without elevation or signs of extension.
RESULTS
Sixteen patients and 27 keloids were studied; however, in 2 cases, the histological confirmation of keloid scar was not obtained, and these patients were excluded from the study. Therefore, 14 patients with a total of 25 keloids were assessed during follow-up.
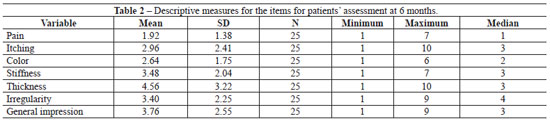
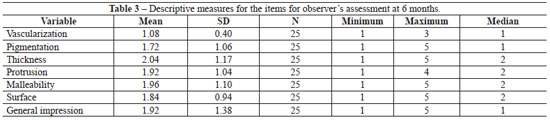
In 100% of the cases, adverse reactions, ranging from redness, burning sensation, pain, itching, swelling, desquamation, diarrhea, dizziness, and even ulceration were observed in the first 2 weeks. The postoperative results for the 25 keloids were assessed by the patient and an observer on week 24. The descriptive results are shown in Tables 2 and 3.
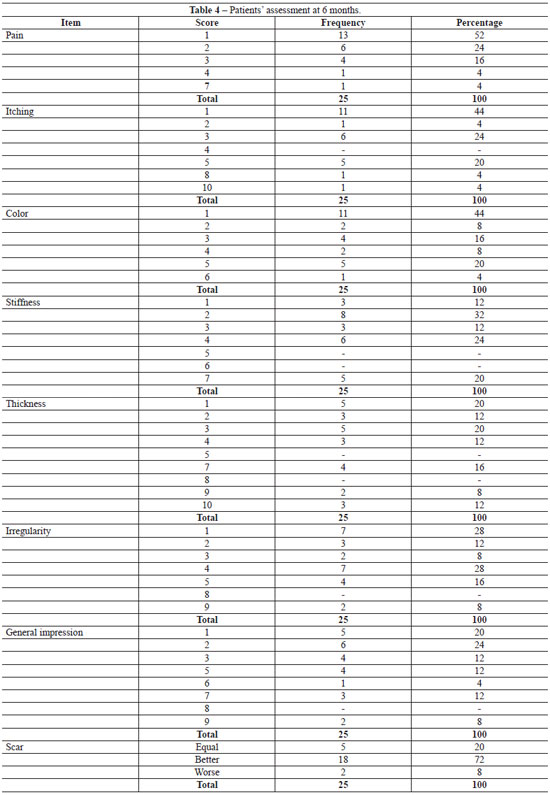
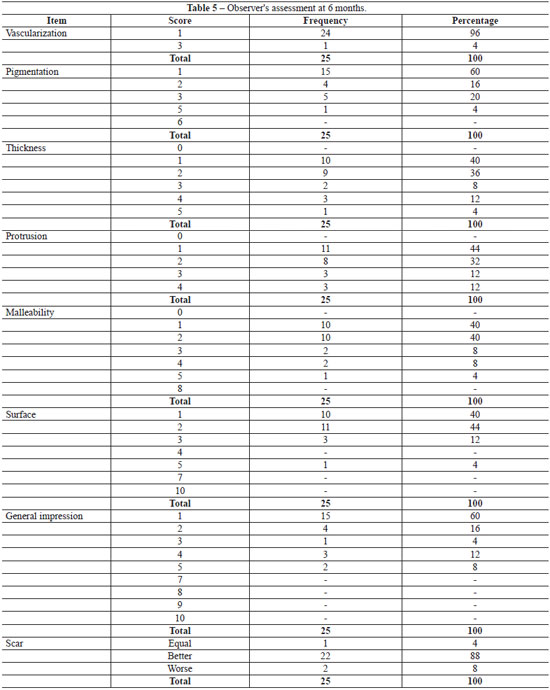
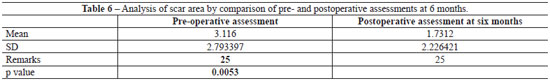
According to the patients' assessment, the scar improved in 72% (18/25) of the cases, was similar to the previous scar in 20% (5/25), and worsened in 8% (2/25). According to the observer's assessment, there was improvement in 88% (22/25) of the cases (Tables 4 and 5). Regarding the signs of recurrence, the observer gave the following ratings: absence of recurrence in 16% (4/25) of cases; +/4+ in 48%, ++/4+ in 24%, +++/4+ in 4%, and ++++/4+ in 8%. The area of the scars was assessed in 2 stages (pre-operative and postoperative period at 24 weeks), and a significant reduction (p=0.0053; Table 6) was observed.
DISCUSSION
Imiquimod 5% cream was applied on the scar for 6 to 8 hours on the night before the surgery and washed off the next morning before surgical excision. This was followed with the use of topical ointment (neomycin/bacitracin) for 2 months. This study focuses on the ability of IFN-α2b to reduce the recurrence of keloids when used immediately after excision, as demonstrated by Berman & Flores8 in 1997. Kaufman & Berman9 examined the effects of imiquimod 5% cream, and reported no recurrence within 24 weeks among the 11 keloids of 10 patients who completed the study, a rate lower than that reported in the literature4,6. Similar results were reported by Arellano and others at the World Congress of Dermatology (Paris, 2002)10. In our study, we considered a recurrence rate of 8% (++++/4+), since the other assessments may change with later evolution to a hypertrophic or even enlarged scar. In our study, the treated keloids were in various locations, unlike the aforementioned studies that assessed keloids only in one location9. Some authors are of the opinion that prognosis is independent of location11,12, but others reported significant clinical differences13,14, such as a greater recurrence of chest and shoulder keloids due to the increased tension in these areas. This finding was confirmed in the current study, with 100% of the recurrences located in the chest region.
The keloids included in the study were histologically confirmed after excision; this was done because Cosman13 noted differences in the recurrence rates of scars without histological confirmation, with the possibility of the inclusion of hypertrophic scars.
There was significant variation in the follow-up period among previous studies on keloids, with many recommending a minimum of 1 year11,13 while others postulating a period of 2 years12,15. The current study presents results of a 6-month follow-up, which could explain the reduced percentage of recurrence. Many authors failed to define how the results were deemed successful or how recurrence was identified4,9,12. In the current study, besides patients using an assessment scale to rate postoperative results, an observer also assessed signs of recurrence and calculated the scar area during the pre- and postoperative periods; both these parameters showed a significant reduction.
CONCLUSION
At 24 weeks, the rate of recurrence after excision of keloids and adjuvant treatment with imiquimod 5% cream indicated that this combined therapy is promising, although a longer follow-up is required.
REFERENCES
1. Alves JCRR, Silva Filho AF, Pereira NA. Cicatrização patológica e seu tratamento. In: Mélega JM, ed. Cirurgia plástica fundamentos e arte - princípios gerais. Rio de Janeiro:Medsi;2002. p.15-24.
2. Figueiredo JCA, Junior FCO, Zampar AG, Mélega JM. Quelóide: fatores de influência prognóstica. Rev Bras Cir Plást. 2008;23(4):274-80.
3. Ogawa R, Mitsuhashi K, Hyakusoku H, Miyashita T. Postoperative electron-beam irradiation therapy for keloids and hypertrophic scars: retrospective study of 147 cases followed for more than 18 months. Plast Reconstr Surg. 2003;111(2):547-53.
4. Berman B, Villa A. Imiquimod 5% cream for keloid management. Dermatol Surg. 2003;29(10):1050-1.
5. van de Kar AL, Kreulen M, van Zuijlen PP, Oldenburger F. The results of surgical excision and adjuvant irradiation for therapy-resistant keloids: a prospective clinical outcome study. Plast Reconstr Surg. 2007; 119(7):2248-54.
6. Rosen DJ, Patel MK, Freeman K, Weiss PR. A primary protocol for the management of ear keloids: results of excision combined with intraoperative and potoperative steroid injections. Plast Reconstr Surg. 2007;120(5):1395-400.
7. Mustoe TA, Cooter RD, Gold MH, Hobbs FD, Ramelet AA, Shakespeare PG, et al; International Advisory Panel on Scar Management. International clinical recommendations on scar management. Plast Reconstr Surg. 2002;110(2):560-71.
8. Berman B, Flores F. Recurrence rates of excised keloids treated with postoperative triamcinolone acetonide injections or interferon alpha-2b injections. J Am Acad Dermatol. 1997;37(5 Pt 1):755-7.
9. Kaufman J, Berman B. Topical application of imiquimod 5% cream to excision sites is safe and effective in reducing keloid recurrences. J Am Acad Dermatol. 2002;47:S209-11.
10. Clark J. Imiquimod vs. interferon: cream matches injection in terms of eliminating keloid recurrence while showing no adverse events. Dermatol Times. 2002:15-6.
11. Ollstein RN, Siegel HW, Gillooley JF, Barsa JM. Treatment of keloids by combined surgical excision and immediate postoperative X-ray therapy. Ann Plast Surg. 1981;7(4):281-5.
12. Kovalic JJ, Peres CA. Radiation therapy following keloidectomy: a 20- year experience. Int J Radiat Oncol Biol Phys. 1989;17(1):77-80.
13. Cosman B. The surgical treatment of keloids. Plast Reconstr Surg. 1961;27:335.
14. Enhamre A, Hammar H. Treatment of keloids with excision and postoperative X-ray irradiation. Dermatologica. 1983;167(2):90-3.
15. Norris JE. Superficial x-ray therapy in keloid management: a retrospective study of 24 cases and literature review. Plast Reconstr Surg. 1995;95(6):1051-5.
1. Plastic Surgeon, Specialist Member of the Brazilian Society of Plastic Surgery (BSPS), Plastic Surgery Department Prof. Linneu Mattos Silveira PUCSorocaba, Sorocaba, SP, Brazil.
2. Plastic Surgeon, Full Member of the (BSPS), Plastic Surgery Department Prof. Linneu Mattos Silveira PUC - Sorocaba, Sorocaba, SP, Brazil.
3. Trainee Doctor in the Plastic Surgery Department Prof. Linneu Mattos Silveira PUC - Sorocaba, Sorocaba, SP, Brazil.
4. Plastic Surgeon, Full Member of the Brazilian Society of Plastic Surgery (BSPS), Head of the Plastic Surgery Department Prof. Linneu Mattos Silveira PUC - Sorocaba, Sorocaba, SP, Brazil.
Corresponding author:
Álisson Yoshiharu Umemura
Rua Benedito Ferreira Telles, 341, apto 82, bloco B - Jd. Simus
Sorocaba, SP, Brazil - CEP 18055-270
E-mail: alissonumemura@ig.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Received: January 9, 2010
Accepted: February 25, 2011
Work performed at Conjunto Hospitalar de Sorocaba, Serviço de Cirurgia Plástica do Departamento de Cirurgia do Centro de Ciências Médicas e Biologicas, PUC - SP - (Plastic Surgery Service of the Department of Surgery of the Biological and Medical Center of PUC - SP), Sorocaba, SP, Brazil.
This work was honored with the Victor Spina Award 2010.
Article submitted for promotion to status of full member of the BSPS.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter