

Original Article - Year 2011 - Volume 26 -
Congenital melanocytic nevi: a retrospective study of epidemiological and therapeutic aspects in a series of 45 patients
Nevo melanocítico congênito: estudo retrospectivo dos aspectos epidemiológicos e terapêuticos em uma série de 45 pacientes
ABSTRACT
Introduction: Although relatively rare, congenital melanocytic nevi are lesions that require monitoring for a long period in order to prevent psychological sequelae and complications inherent to this pathology such as psychological stress, neurocutaneous melanosis, and malignant degeneration. Methods: This study presents epidemiological therapeutic data from a series of 45 cases treated at the Divisão de Cirurgia Plástica e Queimaduras, Hospital das Clínicas, Faculdade de Medicina, Universidade de São Paulo.
Keywords: Nevi and Melanomas. Nevus. Melanoma. Melanosis.
RESUMO
Introdução: Os nevos melanocíticos congênitos, apesar de apresentarem uma incidência relativamente rara, são lesões que necessitam acompanhamento por longo período, a fim de se evitar sequelas psicológicas e complicações inerentes a essa patologia, como estresse psicológico, melanose neurocutânea e degeneração maligna. Método: O presente estudo demonstra os dados epidemiológicos e terapêuticos em uma série de 45 casos atendidos na Divisão de Cirurgia Plástica e Queimaduras do Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo.
Palavras-chave: Nevos e Melanomas. Nevo. Melanoma. Melanose.
Congenital melanocytic nevi (CMN) are pigmented lesions present from the time of birth, and they result from the accumulation of melanocytes of neuroectodermal origin in ectopic locations1. Some peculiar histological features define the congenital etiology of the nevi, such as the presence of nevus cell clusters and the presence of such cellular elements in glandular structures, blood vessels, and subcutaneous tissue2.
The CMN incidence in the general population is 1 in 100 live births; when considering only giant nevi, the incidence is reduced to 1 in 20,000 live births3. Its body distribution is broad, being found more frequently on the trunk and face4,5.
CMN are classified according to their size in different ways. The 3 most cited classifications take into account the nevus' larger diameter, its area, or the percentage of body surface it occupies; according to each classification method, nevi are considered giant when they are more than 20 cm in diameter, cover more than 100 cm2 of the body surface, or cover more than 2% of the body surface, respectively6-8.
The presence of neurocutaneous melanosis, malignant degeneration into melanoma, and the psychological stress for families and children are the major motivations for CMN treatment9.
The actual incidence of melanoma in CMN remains controversial in the literature, and most recent case selections report the incidence to range from 2.8-8.5% of patients with congenital nevi4. A nevus that is >20 cm2, the number of lesions, and age of <3 years are the clinical characteristics associated with the risk of malignant transformation7,10-14.
Surgical treatment is the preferred option when appropriate. The following surgical modalities are used: resection followed by skin grafting, partial resection, and the use of tissue expanders in the adjacent healthy areas followed by resection of the lesions and advancement or rotation of the expanded flaps.
This paper aims to demonstrate the experience of a referral service for CMN treatment from the perspectives of epidemiology, clinical presentation, the employed surgical modality, histopathological reports, and the rate of malignancy in a series of 45 patients.
METHODS
This study is a retrospective study conducted by a medical history review of patients treated at the Divisão de Cirurgia Plástica e Queimaduras, Hospital das Clínicas, Faculdade de Medicina, between 1998 and 2010. CNM was diagnosed considering the related ICD-10 codes: D-28, I-78.1, and Q-82.5. The medical records of 45 patients were located and reviewed.
Epidemiological, clinical, and anatomopathological data were assessed. Epidemiological data included age, gender, ethnicity, and family history. The lesion location, size, and the presence of satellite lesions were reviewed clinically. The employed surgical modality as well as the number of required surgical approaches and complications were also assessed.
The anatomopathological studies of the resected specimens were reviewed to observe the histological types, macroscopic size, and incidence of neoplastic foci.
RESULTS
Of the 45 patients who were assessed, 62% were female and only 13% had a family history of congenital nevus. The ethnic distribution was as follows: 75% of patients were white; 16%, brown; 5%, black; and 4%, Asian (Table 1).
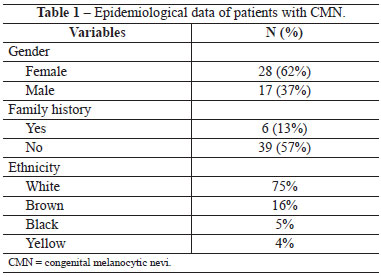
The most frequent location of the CMN was the head and neck region in 35% of cases, followed by the trunk in 27%, and the limbs in 9%. The simultaneous occurrence of nevi in 2 body segments occurred in 20% of patients and occurrence in 3 or more in 9% (Figure 1).
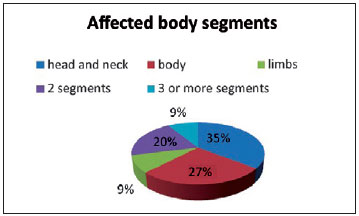
Figure 1 - Distribution of patients with CMN according to affected body segment.
With respect to size, in 69% of the cases the nevi covered 2% or more of the body surface, indicating a giant congenital nevus. Excluding nevi covering less than 1% of the body surface, the arithmetic mean of lesion occurrence was 9.65% (2-50%).
Satellite lesions were present in 30% of the studied cases.
In this case series, 29 (64%) patients underwent some type of surgical treatment. The remaining 16 patients were either under clinical observation, refused surgical treatment, or were awaiting a surgical program.
In absolute numbers, in these 29 patients, 66 partial resections, 13 resections followed by skin grafting, and 18 tissue expansions followed by rotation of the extended flaps were performed (Table 2). Each child received an average of 2.57 surgical approaches (range, 1-9), and only 7 patients were discharged after completion of treatment. The other patients were either awaiting minor resections, refused to continue the treatment due to the high exposure of patients to surgical procedures, or were lost to follow-up.
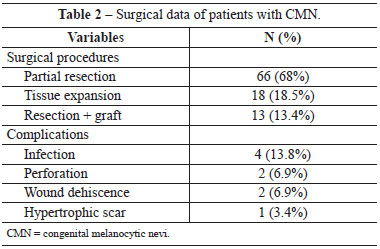
Surgical wound or expander infections, small dehiscence, and wound healing disorders among others were considered as treatment complications. Of the 31% cases that had complications, most were infections (4/29); other complications were small wound dehiscence (2/29), perforation of expanders (2/29), and hypertrophic scarring (1/29) (Table 2).
The analysis of anatomopathological results showed that in 20 cases, there were reports of compound nevi, 12 intradermal nevi, and 2 junctional nevi. Of these, 16 reports mention pathognomonic characteristics of a congenital pattern. Patients with a diagnosis of blue nevus, nevus sebaceous of Jadassohn, and hamartoma of neural cyst as well as isolated cases of proliferative nodules in congenital nevi, typical hyperplasia, and Spitzoid melanoma were evidenced.
In this study, only 1 (2.2%) patient was diagnosed with melanoma. Although the revised report hypothesizes a differential diagnosis of Spitzoid pattern, a melanoma was considered for treatment and follow-up in this case. The patient was a female and had lesions on the trunk that occupied 24% of the body surface, without satellite lesions or neurocutaneous melanosis. As treatment, 7 surgical approaches had already been performed, including 5 partial resections and 2 tissue expansions.
Neurocutaneous melanosis was present in 3 (6.67%) cases, with lesions in meningeal and subcutaneous locations as well as the nails. All of these patients were white and had nevi in multiple segments; only 1 of them had satellite lesions.
With regard to the resected specimens, it was noticed that on an average 147.4 cm2 of skin was resected in each patient, and the largest resection involved resection of 560 cm2 of skin.
Figures 2 to 5 illustrate some cases in this case selection.
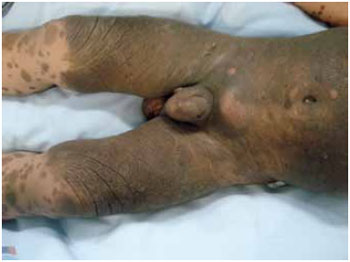
Figure 2 - A nevus occupying 50% of the body surface in a patient with CMN and neurocutaneous melanosis.
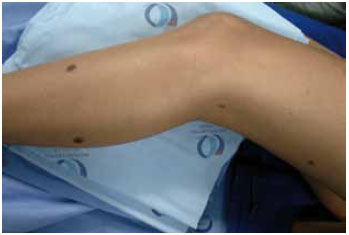
Figure 3 - Photo illustrating satellite lesions in CMN.

Figure 4 - Example of a patient with a congenital nevus on the face who underwent partial resections.
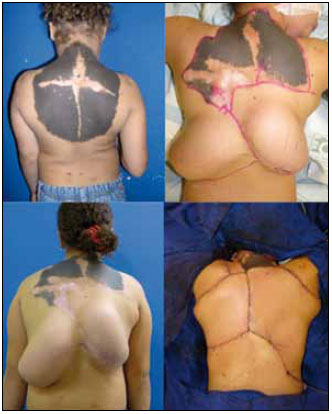
Figure 5 - Example of tissue expansion and flap rotation for a giant congenital nevus on the back.
DISCUSSION
Although relatively rare, congenital nevi are lesions that require multidisciplinary monitoring for a long period in order to avoid psychological sequelae and complications inherent to its pathology.
In the presented case selection, female pediatric patients were predominant, although no consensus was found in the literature regarding gender dominance. With regard to ethnicity, the data corroborate a higher prevalence in white individuals.
Our results identify the head and neck regions as the most frequent areas of CMN, which corroborates the results published by Turkmen et al.5. However, other studies consider the trunk to be the most frequently affected area15. It is important to highlight that in this study, in 29% of the cases, a large proportion of nevi attained dimensions that exceeded the boundaries of body segments and affected more than 2 segments.
Arneja and Gosain2 published data showing the prevalence of satellite lesions to be 80%, which contradicts the present results that show that only 30% of the cases had satellite lesions.
There is a consensus that surgical treatment is most effective when its indication is relevant, but the surgical procedure depends on the characteristics of the lesion, the service complexity, and the plastic surgeon's personal experience. The approaches used in this study were not in line with those proposed in the international literature and predominantly comprised of partial resection followed by tissue expansion5; the reason for this may be the ease of implementing partial resection because of its lower costs and morbidity rates. However, partial resection requires a greater number of approaches to properly resolve each case as compared to tissue expansion.
Infectious complications are more frequent in pediatric patients, and infection remains the major type of complication, especially with the use of expanders. The incidence varies from 4.4% to 9.3% of cases16-21. The incidence was 13% in our case selection; furthermore, as already proposed in the literature, the occurrence of infection is not indicative of the interruption of the expansion program or its failure. In contrast, extrusion is another important complication and treatment involves early removal of the expander21.
Malignant degeneration into melanoma is considered an important complication of CMN because of the high morbidity and mortality associated with the pathology of CMN. Early published works present varying incidences of melanoma, ranging from 0-42%22- 24. Recent studies show that the incidence is lower (around 2.8-8.5% of CMN cases) than what was previously believed7,9. The present results are concordant with this since we obtained an incidence of only 2.2%. Although we identified only 1 case of malignant degeneration in our case selection, we observed that this patient presented with the risk factors proposed in the literature, i.e., large size (24% of the body surface) and age <3 years4,25.
According to the latest diagnostic criteria for neurocutaneous melanosis, 3 patients presented with neurocutaneous melanosis in our case selection. Although all patients had large nevi, only 1 had satellite nevi, which are considered an important risk factor for neurocutaneous melanosis, especially when more than 20 satellite lesions are present26. The occurrence of nevi on the midline, head, and neck is also considered an important risk factor for neurocutaneous melanosis27, but this was not observed in our case selection. In our cases, the lesion was too large to be considered as restricted to one body segment or the midline of the body.
Depending on the age of incidence, neurocutaneous melanosis may be associated with clinical manifestations such as varying degrees of neuropsychomotor development delay, seizures, and weight and height growth restriction.
In this retrospective study, a preference for the treatment of patients with large nevi was noticed; these patients require repeated surgeries and follow-up. Clinical follow-up is critical for these patients in order to diagnose early malignancies and other complications.
Patients with giant CMN should receive special attention from multi-disciplinary staff, including occupational therapists, psychotherapists, and plastic surgeons. In these patients, psychological sequelae occur due to both the stigma associated with the pigmented lesions and the numerous hospitalizations and procedures required for total lesion excision. In order to prevent psychological sequelae and malignant degeneration in children with CMN, surgical treatment should be initiated as early as possible with the aim to complete treatment, if possible, when the child is of preschool age.
REFERENCES
1. Heffek DF, Thaller S. Congenital melanosis: an update. J Craniofac Surg. 2005;16(5):940-4.
2. Bentz ML, Bauer BS, Zuker RM. Principles and practice of pediatric plastic surgery. St Louis:Quality Medical Publishing;2007.
3. Castilla EE, Dutra MG, Oriolo-Parreiras IM. Epidemiology of congenital pigmented naevi: incidence rates relative frequencies. Br J Dermatol. 1981;104(3):307-15.
4. Arneja JS, Gosain AK. Giant congenital melanocytic nevi. Plast Reconstr Surg. 2007;120(2):26e-40e.
5. Turkmen A, Isik D, Bekerecioglu M. Comparison of classification systems for congenital melanocytic nevi. Dermatol Surg. 2010; 36(10):1554-62.
6. Kaplan E, Nickoloff BJ. Clinical and histologic features of nevi with emphasis on treatment approaches. Clin Plast Surg. 1987;14(2):277-300.
7. Quaba AA, Wallace AF. The incidence of malignant melanoma (0 to 15 years of age) arising in "large" congenital nevocellular nevi. Plast Reconstr Surg 1986;78(2):174-81.
8. Bauer BS, Corcoran J. Treatment of large and giant nevi. Clin Plast Surg. 2005;32(1):11-8.
9. Chung C, Forte AJ, Narayan D, Persing J. Giant nevi: a review. J Craniofac Surg. 2006;17(6):1210-5.
10. Solomon LM. The management of congenital melanocytic nevi. Arch Dermatol. 1980;116(9):1017.
11. Kaplan EN. The risk of malignancy in large congenital nevi. Plast Reconstr Surg. 1974;53(4):421-8.
12. Watt AJ, Kotsis SV, Chung KC. Risk of melanoma arising in large congenital melanocytic nevi: a systematic review. Plast Reconstr Surg. 2004;113(7):1968-74.
13. Rhodes AR, Wood WC, Sober AJ, Mihm MC Jr. Nonepidermal origin of malignant melanoma associated with a giant congenital nevocellular nevus. Plast Reconstr Surg. 1981;67(6):782-90.
14. Trozak DJ, Rowland WR, Hu F. Metastatic malignant melanoma in prepubertal children. Pediatrics. 1975;55(2):191-204.
15. Arneja JS, Gosain AK. Giant congenital melanocytic nevi of the trunk and an algorithm for treatment. J Craniofac Surg. 2005;16(5):886-93.
16. Elias DL, Baird WL, Zubowicz VN. Applications and complications of tissue expansion in pediatric patients. J Pediatr Surg. 1991;26(1):15-21.
17. Iconomou TG, Michelow BJ, Zuker RM. Tissue expansion in the pediatric patient. Ann Plast Surg. 1993;31(2):134-40.
18. Gibstein LA, Abramson DL, Bartlett RA, Orgill DP, Upton J, Mulliken JB. Tissue expansion in children: a retrospective study of complications. Ann Plast Surg. 1997;38(4):358-64.
19. Neale HW, High RM, Billmire DA, Carey JP, Smith D, Warden G. Complications of controlled tissue expansion in the pediatric burn patient. Plast Reconstr Surg. 1998;82(5):840-8.
20. Hurvitz KA, Rosen H, Meara JG. Pediatric cervicofacial tissue expansion. Int J Pediatr Otorhinolaryngol. 2005;69(11):1509-13.
21. Adler N, Dorafshar AH, Bauer BS, Hoadley S, Tournell M. Tissue expander infections in pediatric patients: management and outcomes. Plast Reconstr Surg. 2009;124(2):484-9.
22. Danarti R, Konig A, Happle R. Large congenital melanocytic nevi may reflect paradominant inheritance implying allelic loss. Eur J Dermatol. 2003;13(5):430-2.
23. Keipert JA. Giant pigmented naevus: the frequency of malignant change and indications for treatment in prepubertal children. Australas J Dermatol. 1985;26(2):81-5.
24. Bergfeld W, Helwig WB. Risk of developing melanoma in congenital nevi. Presented at the Annual Meeting of the American Academy of Dermatology, Miami, FL, December 27, 1972.
25. Shah KN. The risk of melanoma and neurocutaneous melanosis associated with congenital melanocytic nevi. Semin Cutan Med Surg. 2010;29(3):159-64.
26. Marghoob AA, Dusza S, Oliveria S, Halpern AC. Number of satellite nevi as a correlate for neurocutaneous melanocytosis in patients with large congenital melanocytic nevi. Arch Dermatol. 2004;140(2):171-5.
27. Kadonaga JN, Frieden IJ. Neurocutaneous melanosis: definition and review of literature. J Am Acad Dermatol. 1991;24(5 Pt 1):747-55.
1. Post-graduate in Plastic Surgery in progress at the School of Medicine, Universidade de São Paulo (USP); Resident Physician in Plastic Surgery in the Division of Plastic Surgery and Burns of Hospital das Clínicas, School of Medicine of Universidade de São Paulo (FMUSP), São Paulo, SP, Brazil.
2. Professor at the Department of Surgery, FMUSP; Assistant Physician at the Division of Plastic Surgery and Burns of HCFMUSP, São Paulo, SP, Brazil.
3. Master in Plastic Surgery; Fellow Doctor at the Division of Plastic Surgery and Burns of HCFMUSP, São Paulo, SP, Brazil.
4. PhD in Plastic Surgery; Assistant Physician at the Division of Plastic Surgery and Burns of HCFMUSP, São Paulo, SP, Brazil.
5. Specialist in Plastic Surgery; Fellow Doctor at the Division of Plastic Surgery and Burns of HCFMUSP, São Paulo, SP, Brazil.
6. Resident Physician in General Surgery at HCFMUSP, São Paulo, SP, Brazil.
7. Medical student, FMUSP, São Paulo, SP, Brazil.
8. Full Professor at the Division of Plastic Surgery and Burns HCFMUSP, São Paulo, SP, Brazil.
Correspondence to:
Dov Charles Goldenberg
Rua Pedro de Toledo, 980 Cj. 124
São Paulo, SP, Brazil - CEP 04039-002
E-mail: drdov@terra.com.br
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Received: January 24, 2011
Accepted: March 23, 201
Work performed at the Divisão de Cirurgia Plástica e Queimaduras do Hospital das Clínicas da Faculdade de Medicina da Universidade de São Paulo - (Division of Plastic Surgery and Burns of Hospital das Clínicas, School of Medicine, Universidade de São Paulo), São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter