

Original Article - Year 2011 - Volume 26 -
Scar quality assessment following transareolomammillary and inframammary augmentation mammaplasty: a prospective longitudinal study
Avaliação da qualidade de cicatrizes em mamoplastia de aumento por via submamária e transareolomamilar: um estudo longitudinal prospectivo
ABSTRACT
Introduction: Breast augmentation surgery has become one of the most common procedures in plastic surgery. It is almost entirely determined by 3 variables: location of the incision, pocket plane for implant placement, and type of implant. There is no evidence supporting the superiority of one combination of options over another. The aim of this prospective study was to compare the quality of the scars caused by inframammary and transareolomammillary augmentation mammaplasty in terms of surgeon and patient assessments. Methods: The study included women who underwent cosmetic breast augmentation surgery via inframammary and transareolomammillary incisions. The Patient and Observer Scar Assessment Scale was used for comparing the scar quality 1 year after the surgery. Results: Of 49 patients, 22 and 27 patients underwent primary augmentation mammaplasty via inframammary and transareolomammillary incisions, respectively. Their average follow-up period was 13 months. The transareolomammillary group had better patient assessment scores. However, the surgeon assessment scores of both groups were similar. Conclusions: According to the patients, transareolomammillary augmentation mammaplasty yielded better scar quality, but the surgeons noted no significant difference in scar quality between the inframammary and the transareolomammillary incisions. Additional studies involving a higher number of patients are required to confirm these results.
Keywords: Mammaplasty. Breast implants. Breast implantation. Silicone gels.
RESUMO
Introdução: O aumento das mamas se tornou um dos procedimentos mais realizados em Cirurgia Plástica. A cirurgia de aumento dos seios é quase inteiramente determinada por três variáveis: localização da incisão, plano de alocação da prótese e tipo de implante. Não há provas incontestáveis que apóiem a superioridade de uma combinação de opções sobre outra. O objetivo deste estudo é comparar, através da análise do cirurgião e das pacientes submetidas ao procedimento, a qualidade da cicatriz originada pela cirurgia de aumento mamário utilizando a via submamária e transareolomamilar. Método: Estudo longitudinal prospectivo para investigar pacientes submetidas a aumento mamário estético. Foi utilizada a Escala Paciente e Observador de Avaliação da Cicatriz para mensuração da qualidade cicatricial na revisão de um ano. Resultados: Quarenta e nove mulheres foram submetidas a mamoplastia de aumento primária por via inframamária (22 pessoas) ou abordagem transareolomamilar (27 pessoas), em um seguimento médio de 13 meses. O grupo transareolomamilar apresentou melhores resultados na avaliação do paciente. Entre os cirurgiões, a avaliação de ambos os grupos apresentou resultados semelhantes. Conclusões: Concluímos, com base nas escalas utilizadas e nos dados clínicos obtidos, que a via transareolomamilar demonstrou qualidade superior na avaliação de pacientes. Não houve diferença estatisticamente significativa entre as duas incisões na análise dos cirurgiões. Estudos adicionais envolvendo maior número de pacientes são necessários para comparar o resultado de cada uma das incisões avaliadas.
Palavras-chave: Mamoplastia. Implantes de mama. Implante mamário. Géis de silicone.
Breast augmentation surgery currently is one of the most common cosmetic surgeries. Its purpose is to increase the breast volume to improve the patient's self-image, reducing dissatisfaction related to size, shape, and appearance of the breasts. Several techniques have been used to achieve this goal, and the most common procedure nowadays is silicone breast implantation.
When planning breast augmentation with silicone implants, the 3 key variables are location of the incision, pocket plane for implant placement, and type of implant1-3. There is no evidence to sustain the superiority of one combination of options over another. However, some anatomical configurations that may differ from the surgeon's preferred approach, in both primary cases and reinterventions, are better treated with a specific combination among the available options. Therefore, knowledge and flexibility in the face of different diagnoses and treatments are essential for achieving good results after this surgical intervention4.
The aim of this study was to compare the quality of the scar caused by inframammary and transareolomammillary augmentation mammaplasty in terms of surgeon and patient assessments.
METHODS
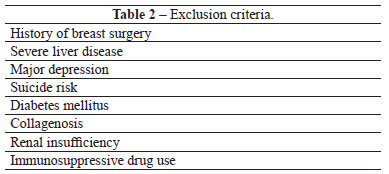
Women who sought breast enlargement surgery were selected for this prospective longitudinal study on the basis of the inclusion and exclusion criteria listed in Tables 1 and 2, respectively.

The choice of access route was based on the areola size. In patients with a transverse areolar diameter equal to or greater than 4 cm, the preferred approach was via a transareolomammillary incision. When the areolar diameter was less than 4 cm or when the prosthesis volume was larger than 250 ml, an inframammary incision was indicated. However, the patients made the final decision on the incision location, based on the surgeon's advice, without influence of this study.
All the procedures were performed under epidural anesthesia with intravenous sedation. Pre-incision infiltration was performed with a solution of 200 ml of 0.9% saline and 0.5 mg epinephrine for local vasoconstriction.
The skin was incised with a 15 blade in the inframammary approach and an 11 blade in the transareolomammillary approach. The pocket plane was detached with an electric scalpel, digital maneuvers, and scissors. By using the nipple as a reference, cranial, caudal, and medial detachments were performed, about 1 cm greater than the diameter of the prosthetic base. The inframammary crease was frequently surpassed, regardless of the incision, so that the prosthesis could be accommodated without folds. Lateral detachments wider than half the base diameter were not made to avoid nerve damage.
Hemostasis was achieved by electrocoagulation as the surgery progressed, and after detaching both sides, minor bleeding was controlled by using a fiber-optic retractor. Surgical gloves were replaced, washed with distilled water, and dried with sterile swabs before each prosthesis introduction. The prosthesis was washed with saline, touched only by the surgeon, and introduced by digital maneuvers, so that its largest projection was placed in the nipple location at the end of the surgery.
In the inframammary approach, 5-0 individual monofilament nylon sutures were placed for the mammary plane and 4-0 continuous monofilament nylon sutures were used for the intradermal plane. In the transareolomammillary approach, only simple 5-0 nylon sutures were used in both planes. Micropore tape and a bra shaper that produces gentle compression were applied at the end of the procedure. The sutures were removed after 14 days. No drains were used.
For postoperative analgesia, 8 mg/kg intravenous dipyrone every 6 h and 0.3 mg/kg intravenous tenoxicam every 12 h were used; if necessary, 0.5 mg/kg intravenous meperidine was administered up to every 3 h. The procedures were performed at a hospital surgical center, and the patients spent the immediate postoperative period at the unit's recovery room. No antibiotics or corticosteroids were used postoperatively.
The patients were divided into 2 groups. In group I, the patients underwent inframammary augmentation mammaplasty. In group II, the patients underwent transareolomammillary augmentation mammaplasty. All data regarding the procedures and the preoperative examinations and postoperative follow-ups were recorded in a specific protocol.
When the patients returned for follow-up 12 months postoperatively, the Patient and Observer Scar Assessment Scale (EPOAC) was used7. This scale consists of 2 numeric scales: the patient scale includes 6 items (pain, itching, color, hardness, thickness, and irregularity) and the observer scale includes 5 items (vascularization, thickness, relief, malleability, and pigmentation). All the items are scored from 1 to 10, where 10 reflects the worst imaginable scar or sensation and 1 reflects normal skin. The lowest overall scores (6 and 5, respectively) reflect normal skin, whereas the highest overall scores (60 and 50, respectively) reflect the worst imaginable scar7,8.
The sample size calculation was based on α = 0.05 with an estimation power of 0.80 by using the Power and Sample Size Calculation software (Vanderbilt University Medical Center, Nashville, TN, USA). The minimum calculated sample value was considered the sample size for this study9. It resulted in 14 individuals in each group.
After entering the data, statistical analysis was performed by means of nonparametric tests for independent samples (chi-square test, t-test, and Mann-Whitney U-test) with SPSS 11.0 (SPSS-IBM, Inc., Chicago, IL, USA). A value lower than 95% was regarded as significant, indicated by p<0.05. The results were assessed after stratification of the cases by patient age, follow-up time, distance from the nipple to the inframammary crease, smoking status, ethnicity, body mass index (BMI), surgical time, prosthesis volume, and implant plane.
RESULTS
Of 49 patients who participated in this study, 22 were allocated to group I and 27 were included in group II. The implants used were from the Allergan, Mentor, and Eurosilicone brands.
As shown in Table 3, the groups showed no significant difference in terms of patient age, distance from the nipple to the inframammary crease, smoking status, surgical time, BMI, implant plane, and prosthesis volume.
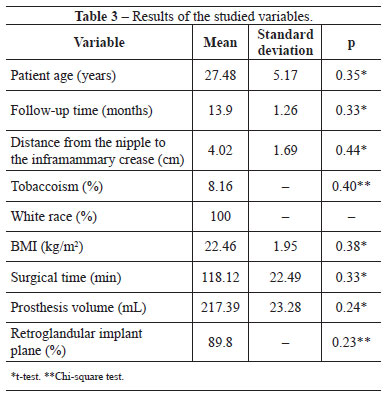
Regarding the scar quality, group II had significantly lower patient assessment scores than group I. However, the groups showed no significant differences in terms of the observer assessment and EPOAC scores (Table 4). Figures 1 and 2 show the results of breast augmentation surgery with inframammary and transareolomammillary incisions, respectively.
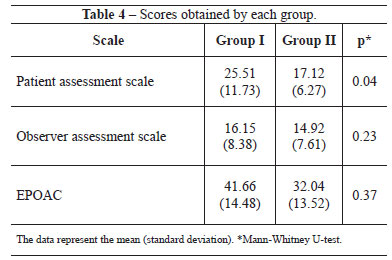
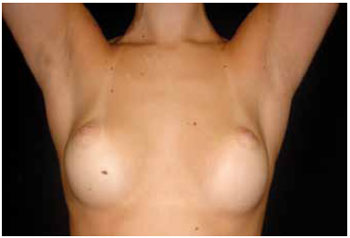
Figure 1 - Thirteen months after inframammary augmentation mammaplasty, with 250 ml prostheses.
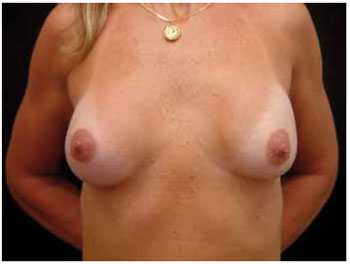
Figure 2 - Thirteen months after transareolomammillary augmentation mammaplasty, with 250 ml prostheses.
There was no surgical wound infection, extrusion, capsular contracture, reoperation, or drainage during the study period. Two cases of superficial cutaneous suture dehiscence, one in each group, were treated with bandages and ointment based on fibrinolysin, deoxyribonuclease, and chloramphenicol. At 12 months postoperatively, 1 patient in each group wished to repair their scar for cosmetic reasons (Figure 3).
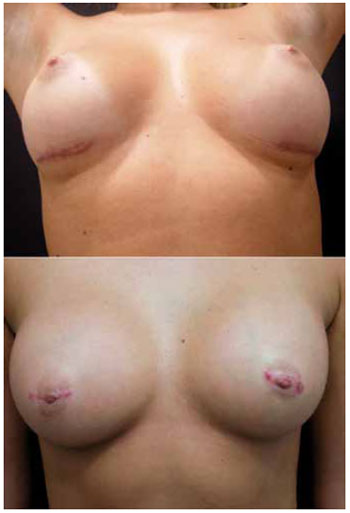
Figure 3 - Inframammary (above) and transareolomammillary (below) scars requiring correction.
DISCUSSION
Czerny of Heidelberg is considered to be the pioneer of breast augmentation surgery, and in the first surgery in 1895, the dorsal lipoma was resected and grafted back in the breast. During the development of breast augmentation surgery, autologous adipose tissue was used, as well as injection of substances such as beads of glass, ivory, rubber, bovine cartilage, polyethylene microplaques, and polyester foam sponge; however, none of these procedures were scientifically acceptable. In the 1960s, 2 surgeons, Gerow Frank and Thomas Cronin, proposed an implant with a solid cover filled with silicone gel. This new prosthesis was an immediate success, making other methods of breast augmentation obsolete. Implant coatings were regularly changed, and several incision alternatives, careful insertion methods, and changes in positioning of the implants were developed10.
The incision for breast prosthesis placement produces a scar, which can lead to functional, cosmetic, and psychological changes. Scar tissue is generally different from healthy skin in terms of aberrant color, increased thickness, irregular surface area, and loss of elasticity. The characteristics of the scar depend on its size and position, suture technique, and wound management, as well as age, ethnicity, diet, associated morbidity, medication use, and genetic predisposition11.
For an evidence-based study, it is necessary to convert the qualitative aspect of scar assessment into a quantitative one12. The EPOAC was employed because it is a reliable tool to quantify the scar quality, with good internal consistency, and because it has been tested and approved for linear scars8. In this study, the scale was easy to apply, and although it did not directly represent satisfaction with the scar, it was an excellent tool for comparisons between the groups.
At present, smaller incisions are made to improve the aesthetic results, while the volume of breast implants has increased. The incision location depends on the surgeon's preference. It is a determining factor for the degree of tissue trauma, implant exposure to endogenous bacteria, potential sensory damage, visibility, and hemostasis1,3,13. In this study, breast augmentation surgeries with inframammary and transareolomammillary incisions were compared because these are surgeon-preferred techniques. It is also important to highlight that the literature lacks studies comparing these 2 scars by using a numerical scale.
The inframammary incision enables direct access to the area of implant placement, without penetrating the breast parenchyma. It is about 4.0 cm long, parallel to the crease, and located about 1 cm above it. Two-thirds of its length is laterally positioned to the breast vertical axis. It is a standard approach, showing no limitation to any type of breast and leaves a scar that is well accepted by most patients. It offers excellent exposure of all detachment areas and for creating the retromammary or retropectoral space. Through this route, additional trauma to tissue other than skin and subcutaneous tissue is avoided. Its major criticism is related to its aesthetic appearance3,14. The outcomes of this study are consistent with such drawbacks, as described in several studies; the patients assessed this scar as having inferior quality to the transareolomammillary scar. Figure 4 shows a good result in this type of scar.
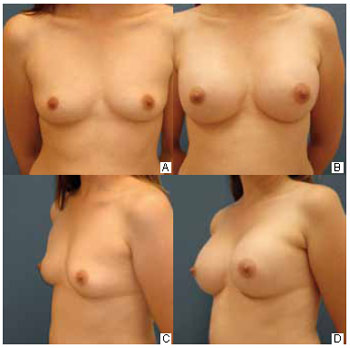
Figure 4 - Inframammary scars showing good results 12 months after 200 ml prosthesis insertion.
The transareolomammillary incision has a transverse location in the center of the mammillary-areolar complex (MAC) and is performed up to 2 mm before its edges. There is, however, greater difficulty in preparing the area and placing the prosthesis. The literature indicates that this type of incision could be associated with increased risk of infection by incision of milk ducts, because the secretion found inside these ducts contains microorganisms capable of causing postoperative infection. This incision could also hinder lactation, generate capsular fibrosis due to increased contamination, and cause additional scars in the case of breast ptosis correction. However, there are no studies demonstrating any of these possible effects, and the literature suggests that, besides preserving MAC vascularization and innervation, it produces good aesthetic results2,5,15. In the patients' assessment, this incision produced a better aesthetic appearance than the inframammary incision. Figure 5 shows a good result of this scar.
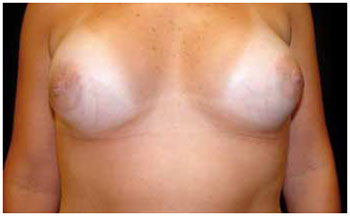
Figure 5 - Transareolomammillary scars showing good results 12 months after 200 ml prosthesis insertion.
Patients and surgeons independently assessed wound healing by using an assessment scale, and all observers recorded an improvement in inframammary scars. However, only the patients' assessment was significant. In the surgeons' assessment, and therefore, in the EPOAC, transareolomammillary scars tended to demonstrate better quality; however, no significant differences were recorded.
Both the study groups had comparable results in the analysis of variables inherent to the healing process. The p value below 0.05 obtained in the patients' assessment suggests that such a difference occurred by chance. However, no test was used to define the degree of expectation and demand in relation to the scars, and the results of this study may have been derived from changes in these factors between the groups.
No one knows for sure why the patients considered the inframammary scar as having worse quality. It may be explained by the fact that the patient assessment scale includes 2 questions on pain and itching. These symptoms, which were not assessed by the surgeons, could be responsible for the difference between the group scores. However, further studies are required to confirm these results and define in detail the reasons for this difference.
CONCLUSION
In the patients' opinion, transareolomammillary augmentation mammaplasty yielded better scar quality than inframammary augmentation mammaplasty. However, the surgeons' assessment showed no significant difference in scar quality between the approaches. Additional studies involving a higher number of patients are needed to confirm these results.
REFERENCES
1. Barbosa MV, Nahas FX, Ferreira LM. A safe way to insert breast implants. Plast Reconstr Surg. 2005;116(6):1824-5.
2. Pitanguy I. Evaluation of body contouring surgery today: a 30-year perspective. Plast Reconstr Surg. 2000;105(4):1499-514.
3. Amaral PB, Rezende KL, Valente DS. Mamaplastia de aumento. In: Carreirão S, Cardim V, Goldenberg D, orgs. Cirurgia plástica. Rio de Janeiro:Atheneu;2005. p.597-605.
4. Hidalgo DA. Breast augmentation: choosing the optimal incision, implant, and pocket plane. Plast Reconstr Surg. 2000;105(6):2202-16.
5. Pitanguy I. Transareolar incision for breast augmentation. Aesthetic Plast Surg. 1978;2:363-72.
6. Regnault P. Breast ptosis. Definition and treatment. Clin Plast Surg. 1976;3(2):193-203.
7. Draaijers LJ, Tempelman FR, Botman YA, Tuinebreijer WE, Middelkoop E, Kreis RW, et al. The patient and observer scar assessment scale: a reliable and feasible tool for scar evaluation. Plast Reconstr Surg. 2004;113(7):1960-5.
8. van de Kar AL, Corion LU, Smeulders MJ, Draaijers LJ, van der Horst CM, van Zuijlen PP. Reliable and feasible evaluation of linear scars by the Patient and Observer Scar Assessment Scale. Plast Reconstr Surg. 2005;116(2):514-22.
9. Dupont WD, Plummer WD Jr. Power and sample size calculations. A review and computer program. Control Clin Trials. 1990;11(2):116-28.
10. Glicenstein J. History of augmentation mammaplasty. Ann Chir Plast Esthet. 2005;50(5):337-49.
11. Lemchem HF, Irigaray JH. Ferida pós-operatória: conduta na evolução normal e nas complicações. In: Pitrez FAB, Pioner SR, eds. Pré e pós-operatório em cirurgia geral e especializada. Porto Alegre: Artmed;1999. p.163-71.
12. Valente DS, Borile G, Tostes F, Chem E, Leonardi DF, Chem RC. Medicina baseada em evidências e conceitos de epidemiologia clínica. In: Maciel E, Serra MC, eds. Tratado de queimaduras. São Paulo: Atheneu; 2004. p.23-7.
13. Tebbetts JB, Adams WP. Five critical decisions in breast augmentation using five measurements in 5 minutes: the high five decision support process. Plast Reconstr Surg. 2005;116(7):2005-16.
14. Doncatto L, Ribeiro L, Graf R, Neves RI. Debate sobre uso de implantes de silicone. Rev Soc Bras Cir Plast. 2006;21(4):246-50.
15. Kompatscher P, Schuler C, Beer GM. The transareolar incision for breast augmentation revisited. Aesthetic Plast Surg. 2004;28(2):70-4.
Plastic Surgeon and Member, Plastic Surgery Service, Hospital Mãe de Deus, Porto Alegre, RS, Brazil.
Corresponding author:
Denis Souto Valente
Rua Veríssimo do Amaral, 580-1706 - Jardim Europa
Porto Alegre, RS, Brazil - CEP 91360-470
E-mail: denisvalente@hotmail.com
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Received: January 24, 2011
Accepted: March 23, 2011
Work performed at the Sistema de Saúde Mãe de Deus de Porto Alegre - (Health System Mãe de Deus de Porto Alegre), Porto Alegre, RS, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter