

Case Reports - Year 2011 - Volume 26 -
Surgical treatment of cicatricial alopecia of the scalp and cranial bone defect in a child without blood transfusion
Tratamento cirúrgico de alopecia cicatricial no couro cabeludo e defeito da calota craniana em criança, sem transfusões de sangue
ABSTRACT
Introduction: Jehovah's Witnesses is a religious group that does not accept blood transfusions. Case Report: The authors describe the case of a child, a Jehovah's Witness, who had cicatricial alopecia of the scalp and a cranial bone defect caused by multiple injuries. Treatment was started with the placement of the expander in the scalp and administration of erythropoietin and ferrous sulfate to increase hemoglobin levels. The expander was successfully withdrawn and the cranial bone defect was successfully corrected with an acrylic plate without resorting to blood transfusions.
Keywords: Jehovah's witnesses. Alopecia. Tissue expansion. Blood transfusion. Erythropoietin.
RESUMO
Introdução: As Testemunhas de Jeová são um grupo religioso que não aceita as transfusões de sangue. Relato do Caso: Os autores descrevem o caso de uma criança, Testemunha de Jeová, que apresentava alopecia cicatricial no couro cabeludo e defeito da calota craniana, resultantes de politraumatismo. O tratamento foi iniciado com a colocação de expansor no couro cabeludo e uso de eritropoetina e sulfato-ferroso para aumento dos níveis de hemoglobina. A retirada do expansor e a correção do defeito da calota craniana com placa de acrílico foram realizadas com sucesso e sem o emprego de transfusões de sangue.
Palavras-chave: Testemunhas de Jeová. Alopecia. Expansão de tecido. Transfusão de sangue. Eritropoetina.
Successful surgical correction of cicatricial alopecia of the scalp and a cranial bone defect in a child without resorting to blood transfusions can be challenging.
Although blood transfusions have been used for decades, in recent times, their role in the management of anemia has been questioned1. Surgeons are not only aware of the risks of blood transfusions, but also of the associated cost and limited supply of blood. Some patients do not accept blood transfusions for various reasons, including religious views2. These factors have given rise to alternative therapeutic strategies to blood transfusion, which are commonly called "blood conservation and management" strategies1. Such strategies should not be limited to the treatment of Jehovah's Witnesses, for whom blood transfusions are not a therapeutic option, but should form an integral part of the surgeon's routine.
Reconstruction of scalp defects is required in cases of acute trauma, tumor resection, and to treat unsightly scars and alopecia. Tissue expansion should be considered, especially in the case of more extensive lesions for which the use of local flaps is inadequate because of the extent of the defect or the traumatized tissue3.
This case report describes surgical treatment of cicatricial alopecia of the scalp with tissue expansion and correction of the cranial bone defect in a child, a Jehovah's Witness, by applying alternative strategies to blood transfusion.
CASE REPORT
B.L.A., a 9-year-old male Jehovah's Witness, was a victim of a car accident at 6 years of age and suffered multiple trauma, resulting in scalping and skull fracture in the right orbitofrontal region. His injuries were treated with suturing of the dura mater at the edges of the bone, debridement of devitalized tissues, and local rotation of the flap of the scalp. Alternative treatments to blood transfusions were used at the time, and the case report was published4.
Three years later, the patient presented with extensive cicatricial alopecia in the right temporoparietal and frontal region, sequelae of his injuries. A computed tomography scan revealed small areas of cortical-subcortical hypoattenuation in the right orbitofrontal region with the appearance of sequelae (Figure 1A). Because the extent of cicatricial alopecia was large, we opted to correct it by scalp tissue expansion. Alternative strategies to blood transfusion were used to comply with the patient's religious views.
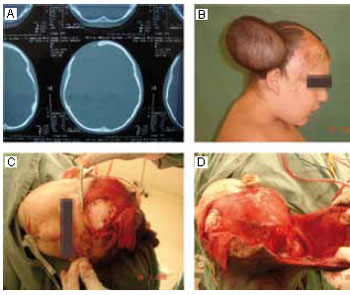
Figure 1 - A: Computed tomography scan showing right orbitofrontal sequelae. B: Picture of the patient after completion of scalp expansion. C: Intraoperative view after placement of the acrylic plate. D: Expanded scalp flap before advancement.
Epinephrine solution (1:250,000) was infused under general anesthesia to reduce intraoperative bleeding. A 480 ml kidney-shaped expander was placed in the subgaleal plane through a right temporal and parietal incision. The preoperative hemoglobin level was 12.5 g/dl, and decreased to 12.4 g/dl on the first postoperative day.
The patient underwent conventional scalp tissue expansion on a weekly basis, attaining a final volume of 660 ml (Figure 1B). In order to increase hemoglobin levels in preparation for the second surgery, during which there was a greater risk of bleeding, the patient received 10,000 IU (300 IU/kg) alpha-erythropoietin subcutaneously 3 times a week, commencing 4 weeks before the surgery, for a total of 9 applications. The patient's serum ferritin level before the erythropoietin treatment was 107.7 ng/ml, which is within the reference range for his age, indicating adequate iron storage. The hemoglobin level was 12.4 g/dl. Oral elemental iron (5 mg/kg daily) and a multivitamin supplement were prescribed. An increase in the number of reticulocytes was observed 7 days after the start of erythropoietin treatment and iron supplementation. At the end of the treatment, the hemoglobin level had increased to 15.5 g/dl.
The second surgery was performed under general anesthesia, starting with the infiltration of 1:250,000 adrenaline solution. The expander and the areas affected by cicatricial alopecia were removed. The neurosurgical team repaired the defect of the right orbitofrontal cranial bone by placing an acrylic plate (Figure 1C). The flap of expanded scalp was advanced fully to cover the defect (Figure 1D). One day after the surgery, the hemoglobin level was 12.1 g/dl. The patient was discharged on the second postoperative day, and the outcome was excellent (Figure 2).
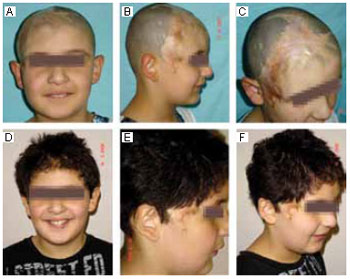
Figure 2 - A, B, and C: Preoperative images. D, E, and F: 4 months after the surgery.
DISCUSSION
Blood conservation and management has been defined as "the appropriate use of blood and blood components with a goal of minimizing their use"1. The exposure of patients to blood transfusions can be minimized by the systematic use of multiple blood conservation techniques that involve the proper use of drugs, equipment, and surgical techniques either separately or in combination. Several surgical and anesthetic techniques have been developed to reduce blood loss, such as endoscopy and laparoscopy; hemostatic surgical instruments (e.g., electrocautery and the argon beam coagulator); biological materials such as fibrin glue that reduce bleeding; techniques that increase vasoconstrictor solution infiltration; the use of drugs that stimulate the production of platelets (e.g., recombinant IL-11); drugs that reduce blood loss during surgery, such as prothrombin complex concentrates and antifibrinolytics (tranexamic acid); desmopressin (which reduces bleeding in patients treated with NSAIDs before surgery); drugs that help reduce acute bleeding (e.g., Factor VIIa); anesthesia with controlled hypotension; acute normovolemic hemodilution; intraoperative cell recovery; and maintenance of normothermia (hypothermia reduces platelet function, thereby increasing bleeding)5,6.
In this case, the frontal skull bone defect was corrected with an acrylic plate, although the use of an external parietal plate removed without craniotomy is the preferred treatment for this type of defect. Cranioplasty with the placement of an acrylic plate in the frontal region may result in osteomyelitis in the long term owing to the proximity of the frontal sinus when it is fully developed. Moreover, plaque destabilization may occur as the child's skull develops. The infiltration of vasoconstrictor solution into the scalp during both surgical placement of the expander and its retreat reduced bleeding from soft tissues.
The knowledge of the compensatory mechanisms (i.e., increased cardiac output and displacement of the hemoglobin dissociation curve to the right) that are activated in the human body in the anemic state to allow greater tolerance to anemia has also contributed to reducing the use of blood transfusions. Previous studies have shown that hematocrit levels as low as 20% are well tolerated in hemodynamically stable children4. However, the use of erythropoietin is recognized as the most significant advance in alternative strategies to blood transfusions.
Erythropoietin is a glycoprotein produced by the kidneys that acts directly on the progenitors of red blood cells in bone marrow, stimulating proliferation, differentiation, and erythrocyte maturation4. Several erythropoiesis-stimulating factors have been approved for clinical trials, each one with different pharmacokinetic and pharmacodynamic properties. Administered subcutaneously, erythropoietin can promote an increase in the number of reticulocytes in 3 days. The equivalent of a blood bag is produced in seven days and that of five bags in 28 days. The stimulation of erythropoiesis by erythropoietin is independent of age or gender. The time required for adequate preoperative stimulation of erythropoiesis is about 4 weeks7.
Several different dosage recommendations of erythropoietin, including 300 IU/kg 3 times per week for 3-4 weeks and 200-600 IU/kg once a week for 4 weeks, have been successfully used to avoid blood transfusions during surgical procedures7. Serum ferritin levels should be monitored to determine body iron stores; a serum ferritin level of 100 ng/ml is adequate. Supplementation of iron; folate; and vitamins B6, B12, and C are recommended during erythropoietin therapy in order to accelerate the erythropoietic response and prevent iron depletion. Although in this case report, the erythropoietic response was stimulated with oral elemental iron and erythropoietin, previous studies have reported that intravenous iron in combination with erythropoietin (intravenous iron 200 mg twice a week) results in greater stimulation8. For the administration of intravenous iron, a dilution of 100 mg iron per 100 ml normal saline and a slow infusion rate (1 ml per minute) are recommended.
Among the possible side effects of erythropoietin, there are rare reports of self-limited flu-like symptoms, hypertension, anaphylactic reaction, hypercalcemia, thrombocytosis, and thrombosis4. Erythropoietin is contraindicated in cases of hypersensitivity, malignant hypertension, pregnancy, and lactation. Erythropoietin has no clinically significant drug interactions, and its safety in patients undergoing surgical procedures has been demonstrated by a similar distribution of adverse effects, including thrombotic events, in over 1,000 patients treated with either erythropoietin or placebo in clinical trials7.
There are several complications associated with blood transfusions; further, their benefits have been questioned. A systematic review and meta-analysis evaluating the efficacy of blood transfusions in critically ill patients was recently published9. As compared to other study types, systematic reviews and meta-analyses present the most scientific evidence10. The systematic review referred to above9 comprised 45 studies including more than 270,000 patients divided into different groups such as trauma victims and general and cardiac surgery patients. Of the 45 studies analyzed, 42 show that the risks of blood transfusions (i.e., increased infection, increased mortality) outweigh the benefits, 2 studies present neutral results, and only 1 reports on benefits in a specific subgroup of patients. The authors of the systematic review conclude that blood transfusions are associated with increased morbidity and mortality rates, and, therefore, the current practice of blood transfusion must be reassessed9.
CONCLUSION
The literature indicates that surgical procedures can be safely performed without blood transfusions with appropriate preoperative preparation. In light of the concern among surgeons regarding the efficacy, risks, costs, and limited supplies of blood, blood management and conservation principles should form an integral part of the surgeon's routine. There are several alternative strategies to blood transfusion that may be used for any patient, including the treatment of Jehovah's Witnesses, and the use of recombinant human erythropoietin is one of the most important, as demonstrated in this case.
ACKNOWLEDGEMENTS
The authors thank Dr. Fernando José Cabral, a member of the Society for the Advancement of Blood Management (SABM), for the thoroughly reviewing this study.
REFERENCES
1. Goodnough LT, Shander A. Blood management. Arch Pathol Lab Med. 2007;131(5):695-701.
2. Gohel MS, Bulbulia RA, Slim FJ, Poskitt KR, Whyman MR. How to approach major surgery where patients refuse blood transfusion (including Jehovah's Witnesses). Ann R Coll Surg Engl. 2005;87(1):3-14.
3. Radwanski HN, Almeida MWR, Aguiar LFS, Altenhofen MS, Pitanguy I. Algoritmo para as alopécias cicatriciais e suas opções de tratamento. Rev Bras Cir Plást. 2009;24(2):170-5.
4. Digieri LA, Pistelli IP, Carvalho CE. The care of a child with multiple trauma and severe anemia who was a Jehovah's Witness. Hematology. 2006;11(3):187-91.
5. Centeno RF, Long CD, Granick MS. Elective plastic surgery in a Jehovah's Witness: a case series and review of the literature. Ann Plast Surg. 2000;45(3):244-51.
6. Shander A. Surgery without blood. Crit Care Med. 2003;31(12 Suppl):S708-14.
7. Goodnough LT, Monk TG. Erythropoietin therapy in the perioperative setting. Clin Orthop Relat Res. 1998;(357):82-8.
8. Rohling RG, Zimmermann AP, Breymann C. Intravenous versus oral iron supplementation for preoperative stimulation of hemoglobin synthesis using recombinant human erythropoietin. J Hematother Stem Cell Res. 2000;9(4):497-500.
9. Marik PE, Corwin HL. Efficacy of red blood cell transfusion in the critically ill: a systematic review of the literature. Crit Care Med. 2008;36(9):2667-74.
10. Goldenberg DC, Baroudi R. Medicina baseada em evidências: como e quando em cirurgia plástica? Rev Bras Cir Plást. 2009;24(1):I.
1. Plastic Surgeon, specialist degree in plastic surgery from the Brazilian Society of Plastic Surgery (SBCP), Associate Member of the SBCP, São Paulo, SP, Brazil.
2. Master in Medicine, Faculty of Medical Sciences of Santa Casa de São Paulo, Member of the SBCP, Physician Volunteer of the Santa Casa de São Paulo, SP, São Paulo, Brazil.
3. PhD in Medicine, Faculty of Medical Sciences of Santa Casa de São Paulo, Full Member of the SBCP, Chief of Plastic Surgery, Santa Casa de São Paulo, SP, São Paulo, Brazil.
Corresponding author:
Juan Carlos Montano Pedroso
Av. Bem-te-vi, 362 - 84A - Moema
São Paulo, SP, Brazil - CEP 04524-030
E-mail: juancmontano@hotmail.com
Submitted to SGP (Sistema de Gestão de Publicações/Manager Publications System) of RBCP (Revista Brasileira de Cirurgia Plástica/Brazilian Journal of Plastic Surgery).
Received: July 25, 2009
Accepted: September 22, 2009
Work performed at the Disciplina de Cirurgia Plástica do Departamento de Cirurgia da Irmandade da Santa Casa de Misericórdia de São Paulo - (Plastic Surgery Discipline of the Department of Surgery of the Irmandade da Santa Casa de Misericórdia de São Paulo), São Paulo, SP, Brazil.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter