

Original Article - Year 2017 - Volume 32 -
Volumetric assessment of breasts in patients undergoing fat grafting with pre-operative expansion (BRAVA®): experience of Santa Casa de São Paulo
Avaliação volumétrica das mamas em pacientes submetidas à lipoenxertia com uso de expansão pré-operatória (BRAVA®): experiência da Santa Casa de São Paulo
ABSTRACT
INTRODUCTION: Autologous fat graft possesses the desired qualities of a filling material. However, the largest barriers to its further development are unpredictable results and high rate of absorption of the grafted fat over time. The objective is to perform a volumetric assessment of the breasts in patients undergoing fat grafting using pre-operative external expansion (BRAVA®).
METHODS: Nineteen patients were operated between March 2012 and June 2015 in the Service of Plastic Surgery, Santa Casa de São Paulo. The parameters evaluated using nuclear magnetic resonance (pre-operatively and 6 months post-operatively) were the following: augmented volume=post-operative volume - pre-operative volume; percentage of augmentation=augmented volume/pre-operative volume x 100; percentage of integration=augmented volume/volume grafted x 100.
RESULTS: The average volume of the breasts in the pre- and post-operative periods were 294.73 ml and 458.42 ml, respectively. The average graft volume was 274.4 ml (150-350 ml). The following parameters were assessed: average volume augmentation, 168.42 ml (90-270 ml); average percentage of augmentation, 58.98% (23-90%); and average percentage of integration, 62.36% (30-80%).
CONCLUSION: The use of external expansion (BRAVA®) in breasts was effective in increasing the rates of integration of the fat grafts and the overall augmentation in breast volumes.
Keywords: Graft Survival; Tissue expansion devices; Mammoplasty; Autografts.
RESUMO
INTRODUÇÃO: O enxerto autólogo de tecido adiposo exibe qualidades desejáveis para utilização como material de preenchimento. Contudo, a grande barreira para o seu maior desenvolvimento foram os resultados pouco previsíveis e o alto índice de absorção da gordura enxertada ao longo do tempo. O objetivo é realizar avaliação volumétrica das mamas de pacientes submetidas à lipoenxertia com uso de expansão pré-operatória (BRAVA®).
MÉTODOS: Foram operadas 19 pacientes no período entre março de 2012 e junho de 2015 na disciplina de Cirurgia Plástica da Santa Casa de São Paulo. Os parâmetros avaliados por ressonância nuclear magnética (pré e 6 meses pós-operatório) foram: Volume Aumentado=Volume Pós-operatório - Volume Pré operatório; Percentual de Aumento=Volume Aumentado/Volume Pré operatório x100; Percentual de Integração=Volume Aumentado/Volume Enxertado x 100.
RESULTADOS: O volume médio das mamas no pré-operatório foi de 294,73 ml e de 458,42 ml no pós-operatório. O volume médio de enxerto foi de 274,4 ml (150-350 ml). Em relação aos parâmetros avaliados, o volume médio de aumento foi de 168,42 ml (90-270 ml), o percentual médio de aumento foi de 58,98% (23-90%) e o percentual médio de integração foi de 62,36% (30-80%).
CONCLUSÃO: A utilização da expansão externa (BRAVA®) na mama mostrou-se um método efetivo nas taxas de integração do enxerto de gordura, bem como no aumento global do volume mamário.
Palavras-chave: Sobrevivência de enxerto; Dispositivos para expansão de tecidos; Mamoplastia; Autoenxertos.
Autologous fat graft possesses the desired qualities of a filling material.
It is autologous, biocompatible, present in sufficient quantities as a donor tissue in most individuals, and is easily obtained, processed, and applied1,2. However, an analysis of the historical evolution of this technique indicates that the largest barriers to its further development were unpredictable results and a high rate of absorption of the grafted fat over time3,4.
The first report on the use of fat for breast filling was in 1895, a milestone indicating the first attempt at breast reconstruction.
The largest impetus for fat grafting occurred in the 1980s with the introduction of the technique known as liposuction by Illouz5-7.
In 1987, the American Society of Plastic Surgeons (ASPS) criticized the use of fat grafting in breasts due to the high associated complication rates (oily cysts, steatonecrosis, infection, risk of cancer, and difficulty in radiological assessment of breasts)8.
In 2007, the ASPS recanted and considered the possibility of fat grafting in breasts; however, they recommended the need for further studies to evaluate the effectiveness and safety of this technique.
Despite the uncertainties, surgeons from Europe and North America published a few series of cases with good results and low complication rates.
The major obstacle that remains is the rate of resorption of the graft and the lack of scientific proof regarding the best methods for harvesting, processing, and placing the tissue.
Numerous procedures have been used for more predictable long-term results. Three categories of treatments can corroborate this process: 1 - treatment of the recipient site (BRAVA®), 2 - injection of additives (hormones, growth factors, platelet-rich plasma, etc.), and 3 - cell culture and expansion.
Treatment of the recipient site with negative pressure (Brava System®), developed by the North American surgeon Roger Khouri, gained great prominence in the recent years and is being used in several centers worldwide9,10.
This technique promotes 5 major effects:
• Increasing the potential of grafting (expanded receptor area);
• Allowing the expansion of areas with fibrosis and constriction;
• Reducing the demand on adipocytes to function as expanders, as the resultant expansion will reduce the tension of the tissue;
• Promoting angiogenesis;
• Optimizing the surgical time.
Khouri et al.11, in 2014, presented the results of a multicenter study with 476 patients who underwent the expansion. They observed an integration rate of 76.9% over an average follow-up period of 3.5 years. This is in contrast with the integration rate of 58.3% reported by Coleman and Saboeiro 12 in 2007, without the use of external expansion. Kosowski et al.13, in 2015, in a study involving 488 patients and a follow-up period of 7 years, also reported a higher rate of integration with the use of external expansion (BRAVA®).
The effects of fat grafting are not restricted to volumetric expansion alone; the regenerating power of the tissue is also increased because of the transferred mesenchymal cells.
It is a fact that fat grafting in breasts is gaining popularity worldwide and is an extremely useful tool, both in reconstruction and esthetic surgery. Newer scientific studies should establish the best methods for harvesting, processing, and placing the adipose tissue into the breasts, in addition to radiation and oncological safety.
OBJECTIVE
The objective of this study was to conduct a volumetric assessment of the breasts in patients undergoing fat grafting using pre-operative expansion (BRAVA®).
METHODS
A total of 19 patients were operated between March 2012 and June 2015 in the Service of Plastic Surgery, Santa Casa de São Paulo, São Paulo, SP, in strict accordance with the guidelines of the Declaration of Helsinki.
All patients were female, with an average age of 33.2 years. The calculation of body mass index (BMI) was performed pre- and post-operatively.
All patients underwent nuclear magnetic resonance pre-operatively and at 6 months post-operatively.
Of the 19 patients, 15 underwent fat grafting for breast reconstruction and 4 underwent correction of esthetic deformities.
All patients used the external expander device (BRAVA®) pre-operatively for 10-12 hours per day for a total duration of 30 days.
At the end of this period, the patients were operated at the central hospital of Santa Casa de São Paulo, under general anesthesia. Fat was collected using 0.5-cm incisions and multiple-hole 3-mm calibre cannulas. The donor areas were the abdomen, flanks, and inner surface of the thighs. Aspiration was performed with an appliance using a pressure of 300 mm Hg.
The fat obtained was centrifuged manually for 2 minutes to separate the oil, fat, and blood fractions (Figures 1 and 2).
Figure 1. Manual centrifugation.
Figure 2. Separation of components (oil, fat, and blood) after centrifugation.
The oil and liquid fractions were discarded, retaining only the fat. This fat was transferred to the grafting gun used to perform the grafting in multiple planes in the shape of a fan. Retro injection was performed and 1 ml grafted with each shot, avoiding grafting in bolus. The grafting cannula was 2.0 mm in diameter.
Areas subjected to radiotherapy or those with fibrosis were released using a procedure called rigotomy, which consisted of the use of a 40 x 12 mm needle to create a multiperforated mesh that would allow expansion and grafting under the scarred or fibrotic area.
The dressing consisted of Zobec® and non-compressive bandages for 1 week.
The parameters evaluated using nuclear magnetic resonance were as follows:
• Augmented volume=post-operative volume - pre-operative volume;
• Percentage augmentation in volume=augmented volume/pre-operative volume x 100;
• Percentage of integration=augmented volume/grafted volume x 100.
RESULTS
The data analysis confirmed that there was no statistically significant difference in the BMI pre- and post-operatively.
All patients were female with an average age of 33.2 years.
The average breast volumes were 294.73 ml pre-operatively and 458.42 ml post-operatively.
The average graft volume was of 274.4 ml (150-350 ml). The following parameters were assessed: average increase in volume, 168.42 ml (90-270 ml); average percentage increase, 58.98% (23-90%); and average percentage of integration, 62.36% (30-80%) (Figures 3 and 4).
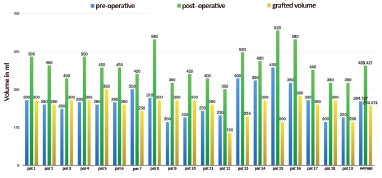
Figure 3. Pre- and post-operative breast volumes and grafted volume per patient.
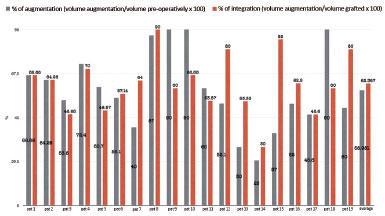
Figure 4. Percentage of breast augmentation and integration of the graft.
Of the 19 patients, 15 underwent fat grafting for breast reconstruction and 4 underwent correction of esthetic deformities (Figures 5 to 11).
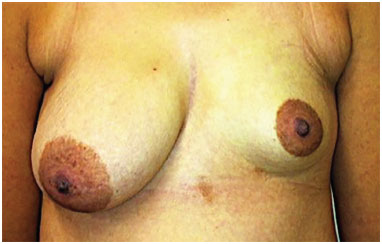
Figure 5. Pre-operative aspect.
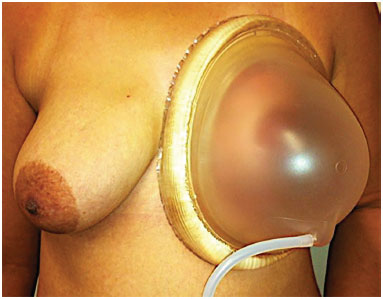
Figure 6. Use of external expansion (BRAVA®).
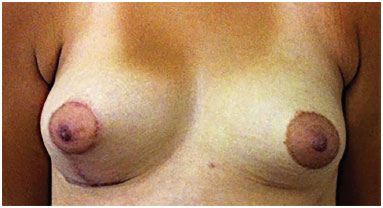
Figure 7. Fat graft in left breast (280 ml) at 6 months post-operatively and right mastopexy.
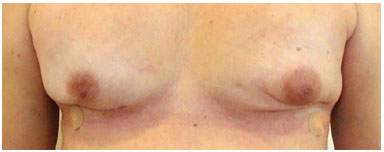
Figure 8. Bilateral adenectomy post-operatively.
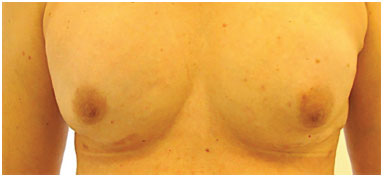
Figure 9. Fat graft for correction of scar retractions (150 ml in each breast) postoperatively; BRAVA® was used pre-operatively.
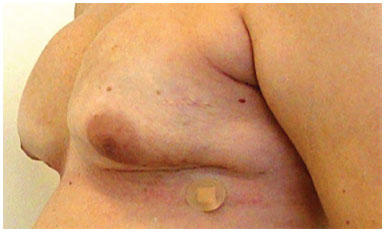
Figure 10. Bilateral adenectomy post-operatively.
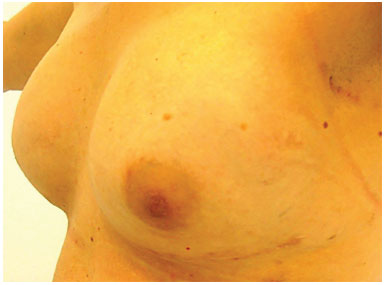
Figure 11. Fat grafts in both breasts (150 ml in each breast) at 1 year post-operatively.
The average surgical time was 2 hours and 17 minutes. All patients had some degree of ecchymosis in the breasts post-operatively and 1 patient had an allergic reaction to the silicone present in the expansion device pre-operatively.
All patients reported improvement in the esthetic appearance of the breast, although the rate of absorption was around 38%, and expressed the desire to undergo more grafting sessions.
On imaging post-operatively, we observed the presence of small oily cysts and areas of sparse steatonecrosis in 7 of the 19 patients. None of the images were considered suspicious enough by the radiology team to warrant a biopsy.
DISCUSSION
There are several factors that interfere with the long-term performance of transplanted tissue. The most important of these are the method of tissue harvest, tissue-processing, grafting, and the quality of the receptor bed. Each of these factors contributes to the final result independently.
Unfortunately, there is no consensus in the literature regarding the best technique, but some guidelines are listed below.
For graft harvesting, cannulae with 2.5-3 mm diameter are used. These result in smaller grafted fat lobules that are more easily revascularized, and cause less injury to the donor area and less trauma during grafting.
Regarding the number of aspiration holes, cannulae with 6-12 side holes (2 x 1 mm) are recommended. Thus, it is possible to achieve a high flow of fat, even at low pressures.
Regarding the grafting method, holding with a blunt tip cannula of 1.5 to 2 mm calibre is recommended, with the lateral orifice diameter equalized in relation to the suction cannulae to entail less trauma.
One can use syringes of 1, 3, or 5 cc, or 'gun' devices that allow the user control over the quantity of graft injected per shot (0.5, 1, or 1.5 cc). The graft should not be injected as a bolus or in cavities. The graft must be in contact with the recipient site for adequate integration.
Dispersion of the graft should be performed in tunnels, multiple layers, and retro injections.
Grafting within the mammary parenchyma should be avoided. Grafts are most frequently placed in the subdermal, subcutaneous, supramuscular, or even the intramuscular planes.
If the patient has an implant or expander in situ, the grafting may be performed between the capsule and the underlying tissue (muscle).
Regarding the aspiration method, the adequate suction pressure is theoretically the lowest possible pressure that can detach the fat from the donor area, thus obtaining a highly viable grafting material, entailing low levels of cellular injury.
Thus, either with a syringe or a device, the negative pressure should remain below 300 mm Hg to obtain adipocytes with a high degree of viability.
When we analyzed the factors related to donor area selection, a consensus was not found. In an experimental study, Padoin et al.14 in 2008 observed that the lower abdomen and inner thighs had a larger number of mesenchymal cells in the adipose tissue when compared to other regions. However, clinically, there was no difference in the outcome with different donor areas.
The area of choice depends largely on the amount of tissue required and the available harvest areas.
Although there is no scientific evidence, it is recommended not to expose the tissue to the external environment. It is preferable to use a closed system to avoid contamination and loss of viability.
When we analyzed the method of processing the graft, we found disparate results.
Smith et al.15, in 2006, performed a clinical study comparing 6 different adipose tissue processing methods before placement (1 - without preparation, 2 - centrifugation only, 3 - washing with Ringer's lactate solution, 4 - washing with 0.9% saline solution, 5 - ringer lactate and centrifugation, 6 - 0.9% saline with centrifugation) and found no statistical differences among the methods.
The 3 main methods of processing the graft prior to its placement are as follows:
• Washing the graft with 0.9% saline solution;
• decantation; and
• centrifugation.
Condé-Green et al.16, in 2010, compared these 3 methods of graft processing and concluded that centrifugation at 3000 rpm (1286 g) was the most deleterious for adipocytes. In contrast, with regard to the concentration of mesenchymal cells, centrifugation had better results in comparison to the others on pellet (deepest layer resulting from the separation of components after centrifugation) analysis.
Washing was associated with the highest cellular viability when a moderate number of mesenchymal cells were used.
There is no consensus regarding the best method as all related studies were non-randomized, and therefore, not comparable.
Nevertheless, the methods that are currently more frequently used are decantation and manual centrifugation, with the intention of causing least injury to the adipocytes.
There are other methods to process the adipose tissue to be grafted that have been used in experimental studies.
Yoshimura et al.17, in 2008, showed high rates of graft integration and low rates of complications when the graft was enriched with mesenchymal cells extracted from the patient's own adipose tissue.
Fraga et al.18, in 2008, compared the integration and viability of autologous grafts of autonomous and non-autonomous adipose tissue; they observed a higher number of viable adipocytes, and a lower rate of fibrosis and necrosis in the graft containing autonomous tissue.
The use of substances that increase the integration of grafted tissue is also an area of constant study.
Pires Fraga et al.19, in 2010, observed that the use of platelet-rich plasma in autologous free fat grafts led to a lower rate of absorption and better integration of the transplanted tissue.
One of the most promising areas in relation to the search for best results is in the manipulation of the recipient site. An external expansion device (BRAVA®) has been used for this purpose.
In the early 1990s, Ilizarov demonstrated that cells, when placed under tension, responded with proliferation and expansion, filling the injury with new tissue20,21. Scientists named this phenomenon mechanotransduction22.
Lancerotto et al.23 showed that, besides mechanotransduction, the tension forces caused a transient ischemia that activates neoangiogenesis. This phenomenon is the basis for the development of the Brava external expansion device (Brava, LLC, Miami, Fl.).
Tom Baker presented the preliminary results of this device at the congress of the ASPS in 1999. Initially, the device was used alone, without a fat graft. It was believed that breast enlargement was solely due to the edema caused in the tissue subjected to expansion. The effects lasted a few weeks but were dependent on the intensive use of the device. The major breakthrough occurred on analyzing the resonance imaging results before and after use of the BRAVA®.
An intense increase in the vascular flow to the breast was observed, in addition to tissue expansion, which could function as a scaffold to receive a greater volume of graft.
This is the basis for the method used to prepare the recipient site.
Two main reasons for fat grafting to the breast are the graft-receptor bed interface and the threshold pressure of the interstitial fluid.
A large volume graft can lead to impaired revascularization because of inadequate contact with the receptor bed; a significant increase in the interstitial pressure may also result, which can lead to ischemia and cell death.
Pre-operative external expansion allows augmentation of the recipient site, together with an increase in the number of blood vessels; these allow the placement of a graft with larger volume without the occurrence of ischemia and loss of the transferred adipose tissue.
Khouri et al.24, in 2014, observed that an optimal graft radius did not exceed 2 mm. A small-sized graft increases the chance of integration and the success of the procedure.
Khouri et al.11, in 2014, presented the results of a multicenter study that involved 476 patients who had undergone expansion; they observed an integration rate of 76.9% over an average follow-up period of 3.5 years.
In this study, we observed an integration rate of 62.36% and augmentation of 58.98%. Although we had a slightly lower integration rate than that of Khouri et al., the percentage of breast augmentation was very high in our study.
Percentage of breast augmentation is the main parameter to be analyzed, as already shown by Khouri and Khouri25 in 2015. The percentage of integration is not the most important clinically relevant parameter. It is not related to the quality of the graft, the recipient site, or the surgical technique used; instead, it is associated with the relationship between the graft volume and the whole receptor area. For example, if we place a 10 ml graft into a 100 ml recipient site (breast) using the appropriate technique, we will probably achieve an integration of 90%. Clinically, this translates to an increase of only 9% in the final volume of the breast.
In contrast, if the same surgeon, using the same technique, grafts 100 ml into the same breast (100 ml recipient site) and achieves an of integration of 60%, this will translate to a final breast augmentation of 60%. Hence, despite a lower rate of integration, the augmentation achieved becomes clinically relevant.
With the external expansion of the tissue, we could increase the graft area, attaining a higher percentage increase in the final volume. Another important point is that with an increase in the complacency of the receptor area, it becomes possible to perform a greater transfer of fat without building up a very large interstitial pressure. This is one of the main predictors of a good outcome.
Numerous tactics have been employed to improve the treatment of breast disorders through fat grafting.
Manipulation of the recipient site using pre-operative expansion has shown good results, despite some degree of discomfort for the patients who use the device. Perhaps this is one of the major obstacles to its more frequent use. There is a cultural and climactic (temperature) difference in our country compared to the United States and European countries, which makes the use of the pre-operative expansion easier in these countries.
Nevertheless, we observed good results in this study that involved 19 patients, which encouraged us to continue using this approach.
CONCLUSION
The use of external expansion (BRAVA®) in the breast as an auxiliary method increased the rates of integration of fat grafts and the overall volume of breasts.
COLLABORATIONS
MFPF Analysis and/or interpretation of data; final approval of the manuscript.
LFP Writing the manuscript or critical review of its contents.
AHJ Final approval of the manuscript.
NCA Statistical analyses.
REFERENCES
1. Billings E Jr, May JW Jr. Historical review and present status of free fat graft autotransplantation in plastic and reconstructive surgery. Plast Reconstr Surg. 1989;83(2):368-81. PMID: 2643129 DOI: http://dx.doi.org/10.1097/00006534-198902000-00033
2. Coleman SR. Structural fat grafting: more than a permanent filler. Plast Reconstr Surg. 2006;118(3 Suppl):108S-20S. DOI: http://dx.doi.org/10.1097/01.prs.0000234610.81672.e7
3. Schorcher F. Fettgewebsverpflanzung bei zu kleiner. Brust Munchen Med Wochenschr. 1957;99:489.
4. Fischer A, Fischer GM. Revised techniques for cellulitis fat: reduction in riding breeches deformity. Bull Int Acad Cosm Surg. 1977;2(4):40-3.
5. Illouz YG. Body contouring by lipolysis: a 5-year experience with over 3000 cases. Plast Reconstr Surg. 1983;72(5):591-7. PMID: 6622564 DOI: http://dx.doi.org/10.1097/00006534-198311000-00001
6. Illouz YG. De l'útilization de la graisse aspirée pour combler les defects cutanés. Rev Chir Esthet Lang Fr. 1985;10:13-20.
7. Illouz YG. The fat cell "graft": a new technique to fill depressions. Plast Reconstr Surg. 1986;78(1):122-3. DOI: http://dx.doi.org/10.1097/00006534-198607000-00028
8. American Society of Plastic and Reconstructive Surgeons. Report on autologous fat transplantation by the ASPRS ad hoc committee on new procedures. Chicago: American Society of Plastic and Reconstructive Surgeons; 1987. 174 p.
9. Khouri R, Del Vecchio D. Breast reconstruction and augmentation using pre-expansion and autologous fat transplantation. Clin Plast Surg. 2009;36(2):269-80. DOI: http://dx.doi.org/10.1016/j.cps.2008.11.009
10. Smith CJ, Khouri RK, Baker TJ. Initial experience with the Brava nonsurgical system of breast enhancement. Plast Reconstr Surg. 2002;110(6):1593-5. DOI: http://dx.doi.org/10.1097/00006534-200211000-00036
11. Khouri RK, Khouri RK Jr, Rigotti G, Marchi A, Cardoso E, Rotemberg SC, et al. Aesthetic applications of Brava-assisted megavolume fat grafting to the breasts: a 9-year, 476-patient, multicenter experience. Plast Reconstr Surg. 2014;133(4):796-807. DOI: http://dx.doi.org/10.1097/PRS.0000000000000053
12. Coleman SR, Saboeiro AP. Fat grafting to the breast revisited: safety and efficacy. Plast Reconstr Surg. 2007;119(3):775-85. DOI: http://dx.doi.org/10.1097/01.prs.0000252001.59162.c9
13. Kosowski TR, Rigotti G, Khouri RK. Tissue-Engineered Autologous Breast Regeneration with Brava®-Assisted Fat Grafting. Clin Plast Surg. 2015;42(3):325-37. DOI: http://dx.doi.org/10.1016/j.cps.2015.03.001
14. Padoin AV, Braga-Silva J, Martins P, Rezende K, Rezende AR, Grechi B, et al. Sources of processed lipoaspirate cells: influence of donor site on cell concentration. Plast Reconstr Surg. 2008;122(2):614-8. PMID: 18626381 DOI: http://dx.doi.org/10.1097/PRS.0b013e31817d5476
15. Smith P, Adams WP Jr, Lipschitz AH, Chau B, Sorokin E, Rohrich RJ, et al. Autologous human fat grafting: effect of harvesting and preparation techniques on adipocyte graft survival. Plast Reconstr Surg. 2006;117(6):1836-44. PMID: 16651956 DOI: http://dx.doi.org/10.1097/01.prs.0000218825.77014.78
16. Condé-Green A, Baptista LS, de Amorin NF, de Oliveira ED, da Silva KR, Pedrosa Cda S, et al. Effects of centrifugation on cell composition and viability of aspirated adipose tissue processed for transplantation. Aesthet Surg J. 2010;30(2):249-55. DOI: http://dx.doi.org/10.1177/1090820X10369512
17. Yoshimura K, Sato K, Aoi N, Kurita M, Hirohi T, Harii K. Cell-assisted lipotransfer for cosmetic breast augmentation: supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg. 2008;32(1):48-55. DOI: http://dx.doi.org/10.1007/s00266-007-9019-4
18. Fraga MF, Helene A Jr, Nakamura F, Lellis RF, Kikuchi W, Esteban D. Comparative study of the integration and viability of autonomised and nonautonomised autologous fat tissue grafts-experimental model in rabbits. J Plast Reconstr Aesthet Surg. 2008;61(9):1044-8. DOI: http://dx.doi.org/10.1016/j.bjps.2007.06.034
19. Pires Fraga MF, Nishio RT, Ishikawa RS, Perin LF, Helene A Jr, Malheiros CA. Increased survival of free fat grafts with platelet-rich plasma in rabbits. J Plast Reconstr Aesthet Surg. 2010;63(12):e818-22. DOI: http://dx.doi.org/10.1016/j.bjps.2010.07.003
20. Ilizarov GA, Devyatov AA, Kamerin VK. Plastic reconstruction of longitudinal bone defects by means of compression and subsequent distraction. Acta Chir Plast. 1980;22(1):32-41.
21. Ilizarov GA, Deviatov AA. Clinical and experimental data on bloodless lengthening of lower extremities. Eksp Khir Anesteziol. 1969;14:27-32.
22. French AS. Mechanotransduction. Annu Rev Physiol. 1992;54:135-52. PMID: 1373277 DOI: http://dx.doi.org/10.1146/annurev.ph.54.030192.001031
23. Lancerotto L, Chin MS, Freniere B, Lujan-Hernandez JR, Li Q, Valderrama Vasquez A, et al. Mechanisms of action of external volume expansion devices. Plast Reconstr Surg. 2013;132(3):569-78. PMID: 23676966 DOI: http://dx.doi.org/10.1097/PRS.0b013e31829ace30
24. Khouri RK, Rigotti G, Cardoso E, Khouri RK Jr, Biggs TM. Megavolume autologous fat transfer: part I. Theory and principles. Plast Reconstr Surg. 2014;133(3):550-7. PMID: 24572848
25. Khouri RK Jr, Khouri RK. Percentage augmentation: the more meaningful index of success in fat grafting. Plast Reconstr Surg. 2015;135(5):933e-935e. PMID: 25700295 DOI: http://dx.doi.org/10.1097/PRS.0000000000001191
Faculdade de Ciências Médicas da Santa Casa de São Paulo, SP, Brazil
Institution: Faculdade de Ciências Médicas da Santa Casa de São Paulo, SP, Brazil.
Corresponding author:
Murillo Francisco Pires Fraga
Rua Cesário Motta Jr, s/n - Vila Buarque
São Paulo, SP, Brazil Zip Code 01221-0100
E-mail: murifraga@ig.com.br
Article received: February 18, 2016.
Article accepted: February 21, 2017.
Conflicts of interest: none.


 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter