

Special Article - Year 2026 - Volume 41Issue 1
Lei Rights of Breast Cancer Patients: Development of the Mama Lei App
Direitos de pacientes com câncer de mama: desenvolvimento do aplicativo Mama Lei
ABSTRACT
Introduction Although breast cancer is the second most prevalent cancer among women, the lack of information leads patients not to exercise their rights under Brazilian law. The present study describes the development ofMama Lei, an application (app) to help patients and the general population understand and use the benefits granted by law.
Materials and Methods We conducted a review of the literature and legislation on the subject to obtain a theoretical foundation to create an algorithm, which was validated using the Delphi technique. In total, 20 evaluators - plastic surgeons, mastologists, or lawyers - were invited to evaluate the content of the algorithm and suggest adjustments. The Cronbach's alpha coefficient and the Content Validity Index (CVI) were calculated to determine the reliability of the assessment tool. After the necessary adaptations, the algorithm was transformed into an app.
Results In total, 18 evaluators with an average age of 39.5 years answered within the established timeframe. The average length of professional activity was of 15.5 years, and 44.4% of the evaluators were plastic surgeons. The overall CVI was of 97.2%, showing that the instrument is reliable. The overall Cronbach's alpha coefficient was of 0.754, revealing acceptable internal consistency. The Mama Lei app was based on an algorithm (https://mama-lei.netlify.app/), and it was registered with the Brazilian National Institute of Industrial Property (Instituto Nacional da Propriedade Industrial, INPI, in Portuguese) under number BR512023001996-8.
Conclusion The Mama Lei app was successfully developed and is now available free of charge to breast cancer patients and the general public.
Keywords: patient rights; breast neoplasms; mobile applications; smartphone
RESUMO
Introdução O câncer de mama é o segundo tumor maligno mais prevalente entre as mulheres, mas a falta de informações faz com que as pacientes não exerçam por completo seus direitos assegurados pela lei brasileira. Este estudo descreve o desenvolvimento do Mama Lei, um aplicativo (app) para ajudar as pacientes e a sociedade em geral a entender e aproveitar os benefícios garantidos por lei.
Materiais e Métodos Conduziu-se uma revisão da literatura e da legislação sobre o assunto para formar uma base teórica e criar umalgoritmo, que foi validado segundo a técnica de Delphi. No total, 20 avaliadores - cirurgiões plásticos, mastologistas ou advogados - foram convidados a analisar o conteúdo do algoritmo e sugerir ajustes. O alfa de Cronbach e o Índice de Validade do Conteúdo (IVC) foram calculados para determinar a confiabilidade da ferramenta de avaliação. Após as modificações necessárias, o algoritmo foi transformado em um app.
Resultados Ao todo, 18 avaliadores com idade média de 39,5 anos responderam dentro do período estabelecido. O tempo médio de atividade profissional foi de 15,5 anos, e 44,4% dos avaliadores eram cirurgiões plásticos. O IVC geral foi de 97,2%, o que mostra que o instrumento é confiável. O coeficiente alfa de Cronbach geral foi de 0,754, o que revela consistência interna aceitável. O app Mama Lei foi desenvolvido a partir de um algoritmo (https://mama-lei.netlify.app/) e registrado no Instituto Nacional da Propriedade Industrial (INPI) sob o número BR512023001996-8.
Conclusão O app Mama Lei foi desenvolvido com sucesso e está disponível gratuitamente para pacientes com câncer de mama e o público geral.
Palavras-chave: direitos do paciente; neoplasias de mama; aplicativos móveis; smartphone
Introduction
In Brazil, breast cancer is the second most common type of cancer among women after non-melanoma skin cancers. It represents 1 in 10 new cases of neoplasia diagnosed world-wide each year.1 The Brazilian National Cancer Institute (Instituto Nacional de Câncer, INCA, in Portuguese) estimat-ed an incidence of 74 thousand new cases of breast cancer annually in Brazil until 2025, making it the leading cause of cancer death in women, at a rate of 15.5%.2
Breast cancer treatment is lengthy and multidisciplinary. Patients and their families endure the psychological impact and financial consequences.3 In Brazil, this assistance is en-sured by the Unified Health System (Sistema Único de Saúde, SUS, in Portuguese), which acts from prevention to screening, diagnosis, and treatment of breast cancer patients.3
Over time, in an effort to lessen these impacts, people diagnosed with cancer have acquired rights to legal benefits and exemptions. However, the lack of information leads women with breast cancer to remain unaware of the special rights mentioned in the legislation and end up not taking advantage of these benefits.3
Breast cancer patients experience a sense of uncertainty regarding the future, low self-esteem, and fear of death and mutilation. Although this situation compromises rational information absorption, technology can help.4
Information and the active, conscious participation of patients in the health-disease process are critical and posi-tively influence treatment outcomes. With the advent of the internet, searching for information has become easier; however, the data found is at times less reliable. Today, smart-phone use is virtually universal, and applications (apps) have become popular and beneficial tools.5
Well-designed and validated digital apps have proven to be potent tools to provide reliable and accessible information to patients, promoting autonomy in healthcare, and expand-ing knowledge about their rights. Recent studies6,7 highlight that health technologies, when evidence-based and devel-oped with adequate scientific validation, contribute significantly to health education and informed decision-making. The present study describes the development of Mama Lei, a free-access app to assist breast cancer patients, their fami-lies, and professionals responsible for their care, in becoming informed about, understanding, and securing their legally-mandated benefits.
Objectives
The current study aimed to develop and validate an algo-rithm to support the creation of an app to inform patients about their rights regarding breast cancer.
Materials and Methods
The present descriptive study of technological development was completed as part of a professional master’s degree pro-gram. The institutional Ethics Committee approved this study (under CAAE: 51511521.9.0000.5102, opinion no. 4.993.133).
We conducted a comprehensive review of the Brazilian literature and legislation regarding the rights of breast cancer patients. The literature search used the descriptors breast neoplasms, patient rights, mobile applications, and smartphone, as well as their Portuguese equivalents. The search included the following databases: Cochrane Library, Scientific Electronic Library Online (SciELO), Latin American and Caribbean Literature in Health Sciences (Literatura Latino Americana e do Caribe em Ciência da Saúde, LILACS, in Portuguese), and Medical Literature Analysis and Retrieval System Online (MEDLINE), accessed via PubMed, from the National Library of Medicine in the United States. Inquiries were made regarding Brazilian legislation on breast cancer through the Official Gazette of the Union (Diário Oficial da União, DOU) and various online search engines. The literature search was restricted to 5 years (2017-2022), while the legislation search focused on documents released since the promulgation of the Brazilian Federal Constitution, in 1988.
The algorithm was structured in a sequence of five stages: bibliographic research, collection of relevant information, writing of explanatory texts, development of the algorithm using the Canva (Canva Pty Ltd.) software, and review of the content.
The algorithm developed was then validated using the Delphi technique, a systematized method to obtain expert consensus on a given topic. By applying structured ques-tionnaires, the Delphi technique aims to reach a consensus among a group of experts or evaluators.6 The evaluation questionnaire was developed based on a Likert scale, which grades responses from 1 to 5, according to the level of agreement regarding the issue.8 The evaluators analyzed the algorithm in terms of the adequacy of the language, the sequence of the information, the ease of understanding, and the relevance of the content.
In total, 20 professionals, selected by convenience, were invited to participate in this stage and acted as evaluators.6,9 Physicians with specialist titles in Plastic Surgery or Mastol-ogy, in good standing with their respective Regional Medical Councils, and lawyers in good standing with the Brazilian Bar Association (Ordem dos Advogados do Brasil, OAB, in Portuguese) were considered for inclusion. Only professionals with at least 5 years of experience in their field were included.
An email was sent to the selected professionals, contain-ing an invitation letter and a link to a Google Forms (Alphabet Inc.) electronic form. The invited evaluator, if agreeing to participate, was required to enter their full name and email address at the end of the form, indicating their consent. The electronic form only allowed progress to the next stage after this explicit consent.
The next stage of the form consisted of questions to collect the participant’s personal data and a link to access the algorithm for evaluation. The final stage of the electronic form consisted of a script to assess the algorithm, with spaces for suggestions. After 15 days, a reminder was sent by email to those who had not yet responded. After another 15 days from the second email, those who did not respond were excluded. After analyzing all the responses received, the necessary adjustments to the algorithm were made until the final text was drafted.10
Statistical analysis of the data used measures of central tendency for quantitative variables and absolute and relative frequencies for categorical variables. Cronbach’s alpha coef-ficient assessed the internal consistency of the question-naire,7,11 and the Content Validity Index (CVI) of the content of the app was calculated.6
Statistical analysis was performed in the Minitab (Mini-tab, LLC) software, version 18.1, and in the IBM SPSS Statistics for Windows (IBM Corp.) software, version 22.0. The level used to reject the null hypothesis was 5% (p ≤ 0.05) for all statistical tests.
The final version of the algorithm was transformed into an app by an Information Technology professional. After all revisions, the app was submitted to the Brazilian National Institute of Industrial Property (Instituto Nacional da Pro-priedade Industrial, INPI, in Portuguese) for registration.
Results
The response rate from the professionals invited to evaluate the reliability of the algorithm’s content was of 90%; 18 responded within the stipulated time or during deadline extensions. ►Table 1 presents the main characteristics of the evaluators.
| Variable | Values |
|---|---|
| Age (years) | |
| Minimum-maximum | 29-58 |
| Mean ± standard deviation | 40.1 ± 8.4 |
| Time of professional activity (years) | |
| Minimum-maximum | 1-26 |
| Mean ± standard deviation | 10.1 ± 8.8 |
| Profession: n (%) | |
| Plastic surgeon | 8 (44.4%) |
| Lawyer | 7 (38.9%) |
| Mastologist | 3 (16.7%) |
Table 2 presents the professionals’ evaluations, includ-ing the CVI for each aspect assessed and the overall score. The final Cronbach’s alpha coefficient, which determines the internal consistency of the questionnaire, was of 0.754, indicating substantial internal consistency.12
| Criteria evaluated | Classification | CVI: % |
|---|---|---|
| Information sequence | Adequate | 94.4% |
| Ease of understanding | Adequate | 94.4% |
| Language | Adequate | 100.0% |
| Relevance of the content | Adequate | 100.0% |
| Global CVI | 97.2% | |
| Cronbach’s alpha coefficient | 0.754 |
Regarding the observations made by the evaluators, there was overall agreement on the relevance of the content, and the algorithm and the development of the app were consid-ered of great use to patients, as much of the information provided is not widely known among the population. Fur-thermore, in the evaluators’ opinion, the algorithm uses accessible language, and the text is comprehensive.
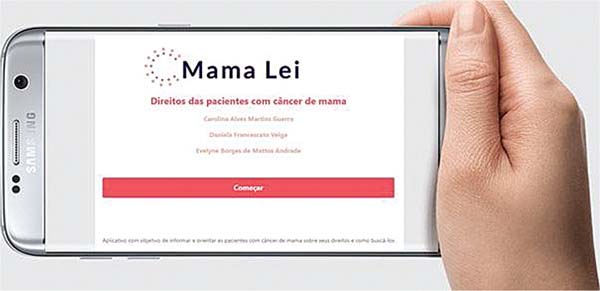
The app created was named Mama Lei (►Figs. 1-3), combining the Portuguese terms for breast (mama) and law (lei), the keywords of the study. The app was registered with INPI (BR512023001996-8) and is available for free at https://mama-lei.netlify.app.
Discussion
Breast cancer is a significant health problem in Brazil and worldwide, with a high prevalence and increasing inci-dence.1,2 In Brazil, several laws guarantee rights to women with breast cancer, focusing on early diagnosis, proper treatment, and social support, aiming to minimize the psychological and financial impacts of the disease.13 How-ever, most patients, and sometimes the team responsible for their treatment, are unaware of these benefits, meaning that many patients do not have access to their rights.
The development of apps based on reliable and validated algorithms provides a greater availability of high-quality information, facilitating access to it.6,7 Apps offer advan-tages, as they allow reaching more patients, saving time, and they are reliable, and have low costs. The Mama Lei app provides instant, clear information for breast cancer patients, healthcare teams, and citizens interested in the topic, helping them identify their rights and exercise them early on.
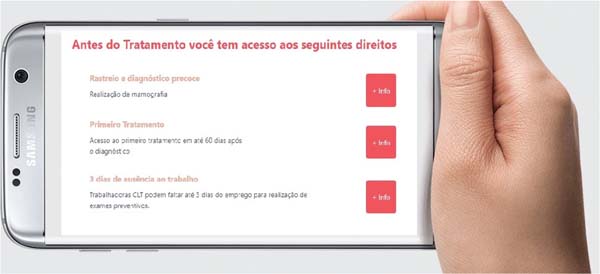
Disease screening through the SUS is guaranteed to patients under Brazilian Law No. 11,664/2008,14 updated by Law No. 14,335/2022.15 These laws ensure that mammo-grams are available starting from puberty and in subjects with suspicious clinical signs or a family history of breast issues, as well as the potential diagnostic complementation with ultrasound at the discretion of a medical professional. Ordinance No. 1,228/201216 of the Brazilian Ministry of Health (Ministério da Saúde, MS, in Portuguese)/Healthcare Department (Secretaria de Atenção à Saúde, SAS, in Portu-guese) establishes the provision of mammograms at mobile units, expanding access in hard-to-reach regions.
In Brazil, Law No. 12,732/2012, known as the “60-day Law,” establishes a maximum period of 2 months between histopathological diagnosis and the start of cancer treatment in SUS.17 Regarding social security and labor rights, Brazilian patients may withdraw the Severance Indemnity Fund (Fundo de Garantia do Tempo de Serviço, FGTS, in Portu-guese) and Social Integration Program/Public Servant Sav-ings Program (Programa de Integração Social/Programa de Formação do Patrimônio do Servidor Público, PIS/PASEP, in Portuguese),18,19 obtain sickness benefits if insured at the National Institute of Social Security (Instituto Nacional de Seguridade Social, INSS),20 and get time away from work supported by a medical certificate.21 Public servants may also obtain leave for their own treatment or to care for sick dependents, subject to medical examination.21
Out-of-Domicile Treatment (Tratamento Fora do Domi-cílio, TFD, in Portuguese), regulated by MS/SAS Ordinance No. 055/1999,22 constitutes an important strategy to enable access to specialized centers outside the city or state of residence, ensuring transportation, lodging, and food, in-cluding for a companion when necessary. Moreover, state laws, such as Law No. 4,510/2005 from Rio de Janeiro,23 guarantee free intermunicipal transportation for patients with severe chronic diseases.
Cancer patients may also obtain several tax and property benefits, including exemption from income tax on retire-ment or pension benefits, exemption from the Tax on Motor Vehicle Ownership (Imposto sobre a Propriedade de Veículos Automotores, IPVA, in Portuguese) (with state regulation), exemption from the Urban Property Tax (Imposto Predial e Territorial Urbano, IPTU, in Portuguese) (according to mu-nicipal legislation), exemption from the Tax on Industrialized Products (Imposto sobre Produtos Industria-lizados, IPI, in Portuguese) on the purchase of a vehicle for patients with disabilities resulting from the disease, and the settlement of housing financing in case of permanent dis-ability, provided it is stipulated in the contract.24-26 In the city of São Paulo, there is also an exemption from the municipal vehicle rotation program for patients undergoing treatment. Moreover, some states offer a social voucher (vale social, in Portuguese), a benefit for free intercity or intracity passenger transport under state administration, intended for people with disabilities or chronic diseases requiring con-tinuous treatment and whose interruption could pose a life-threatening risk.23
The provision of high-cost medications is a legal obligation of SUS, including reimbursement in case of unavailability.3 In the surgical context, breast reconstruction is ensured by a legal framework composed of Laws No. 9,797/1999 (SUS-funded reconstruction),27 No. 12.802/201328 (immediate reconstruc-tion when technically feasible),28 and No. 13.770/2018 (right to symmetrization of the contralateral breast and reconstruction of the areola-nipple complex),29 recognizing the integrality of esthetic and psychosocial rehabilitations. Recently, Law No. 15,171/2025 expanded the right to breast reconstruction, ensuring the procedure for all women who need reconstructive breast surgery, as a right to comprehensive health and physical and psychological reparations.30
The development of the Mama Lei app considered the fragmented nature of legal sources, and the challenges patients encounter when trying to access this information. This free and validated digital tool uses accessible language and works offline. Available for Android devices and as a web version (https://mama-lei.netlify.app/), the app provides a reliable, organized compilation of the main legal rights of breast cancer patients, contributing to the promotion of autonomy, citizenship, and quality of life for these women and their families.
The Mama Lei app has great potential to improve numer-ous aspects of the quality of life of breast cancer patients, including emotional and financial ones, by offering informa-tion not only to the patients themselves, but also to the healthcare teams that assist them and to the people within their family and professional lives, about the rights and benefits Brazilian law offers them and how to access them.
Conclusion
Mama Lei was developed using a validated algorithm. It is easy and free to access, and available to inform breast cancer patients, healthcare professionals, and the general public about the rights protecting these women.
REFERENCES
1. Instituto Nacional de Câncer (INCA). Estimativas da incidência e mortalidade por câncer. Rio de Janeiro: INCA; 2023 [citado 2025 jan 10]. Disponível em: https://www.inca.gov.br/numeros-de-cancer
2. Instituto Nacional de Câncer (INCA). Estimativa 2023: incidência do câncer no Brasil. Rio de Janeiro: INCA; 2023 [citado 2025 jan 9]. Disponível em: https://www.inca.gov.br/publicacoes/livros/estimativa-2023-incidencia-de-cancer-no-brasil
3. Brasil. Lei n° 8.080, de 19 de setembro de 1990. Dispõe sobre as condições para a promoção, proteção e recuperação da saúde, a organização e o funcionamento dos serviços correspondentes e dá outras providências. Brasília: Diário Oficial da União; 1990. Dispo-nível em: https://www.planalto.gov.br/ccivil_03/leis/l8080.htm
4. Miranda FD, Salomé GM, Costa MG, Alves JR. Mobile app for patient education about breast cancer surgical treatment. Fisioter Mov 2022;35:e35128. Doi: 10.1590/fm.2022.35128
5. Giunti G, Giunta DH, Guisado-Fernandez E, Bender JL, Fernandez-Luque L. A biopsy of Breast Cancer mobile applications: state of the practice review. Int J Med Inform 2018;110:1-9. Doi: 10.1016/ j.ijmedinf.2017.10.022
6. Silva BA, Silva PRR, Brito WR, et al. Processos de validação de instrumentos para área da saúde. Rev Eletr Acervo Saúde. 2024;24 (02):e14695. Doi: 10.25248/REAS.e14695.2024
7. Zakariya YF. Cronbach’s alpha in mathematics education research: Its appropriateness, overuse, and alternatives in esti-mating scale reliability. Front Psychol 2022;13:1074430. Doi: 10.3389/fpsyg.2022.1074430
8. Kusmaryono I, Wijayanti D, Maharani HR. Number of response options, reliability, validity, and potential bias in the use of the Likert scale in education and social science research: A literature review. Int J Educ Method 2022;8(04):625-637. Doi: 10.12973/ ijem.8.4.625
9. Alexandre NMC, Coluci MZO. Validade de conteúdo nos processos de construção e adaptação de instrumentos de medidas. Cien Saude Colet 2011;16(07):3061-3068. Doi: 10.1590/s1413-81232011000800006
10. Hohmann E, Cote MP, Brand JC, Lubowitz JH. Research pearls: expert consensus based evidence using the Delphi method. Arthroscopy 2018;34(12):3278-3282. Doi: 10.1016/j.arthro.2017.10.025
11. Tavakol M, Dennick R. Making sense of Cronbach’s alpha. Int J Med Educ 2011;2:53-55. Doi: 10.5116/ijme.4dfb.8dfd
12. Freitas ALP, Rodrigues SB. O uso do coeficiente alfa de Cronbach nos artigos da área de administração no Brasil: uma análise bibliométrica. Rev Contemp Contab. 2005;2(03):64-79
13. Instituto Nacional de Câncer (INCA). Diretrizes para a detecção precoce do câncer de mama no Brasil. Rio de Janeiro: INCA; 2023 [citado 2025 jan 8]. Disponível em: https://www.inca.gov.br/publicacoes/livros/diretrizes-para-deteccao-precoce-do-cancer-de-mama-no-brasil
14. Brasil. Lei n° 11.664, de 29 de abril de 2008. Dispõe sobre a efetivação de ações de saúde que assegurem a prevenção, a detecção, o tratamento e o seguimento dos cânceres do colo uterino e de mama, no âmbito do Sistema Único de Saúde - SUS. Brasília: Diário Oficial da União; 2008. Disponível em: https://www.pla-nalto.gov.br/ccivil_03/_ato2007-2010/2008/lei/l11664.htm
15. Brasil. Lei n° 14.335, de 10 de maio de 2022. Altera a Lei n° 11.664, de 29 de abril de 2008, para dispor sobre a atenção integral à mulher na prevenção dos cânceres do colo uterino, de mama e colorretal. Brasília: Diário Oficial da União; 2022. Disponível em: https://www.planalto.gov.br/ccivil_03/_ato2019-2022/2022/lei/l14335.htm#:~:text=LEI%20N%C2%BA%2014.335%2C%20DE%2010%20DE%20MAIO%20DE%202022&text=Altera%20a%20Lei%20n%C2%BA%2011.664,Art
16. Brasil. Ministério da Saúde. Secretaria de Atenção à Saúde. Portaria n° 1228, de 18 de outubro de 2012. Regulamenta a habilitação para o Programa de Mamografia Móvel, instituído pela Portaria n° 2.304/GM/MS, de 4 de outubro de 2012. Brasília: Diário Oficial da União; 2012. Disponível em: https://bvsms.saude.gov.br/bvs/saudelegis/sas/2012/prt1228_30_10_2012.html
17. Brasil. Lei n° 12.732, de 22 de novembro de 2012. Dispõe sobre o primeiro tratamento de paciente com neoplasia maligna com-provada e estabelece prazo para seu início. Brasília: Diário Oficial da União; 2012. Disponível em: https://www.planalto.gov.br/ccivil_03/_ato2011-2014/2012/lei/l12732.htm
18. Brasil. Lei n° 8.922, de 25 de julho de 1994. Acrescenta dispositivo ao art. 20 da Lei n° 8.036, de 11 de maio de 1990, para permitir a movimentação da conta vinculada quando o trabalhador ou qualquer de seus dependentes for acometido de neoplasia maligna. Brasília: Diário Oficial da União; 1994. Disponível em: https://www.planalto.gov.br/ccivil_03/leis/l8922.htm
19. Brasil. Resolução CD/PIS-PASEP n° 1, de 15/10/1996. Autoriza a liberação do saldo das contas do Programa de Integração Social - PIS e do Programa de Formação do Patrimônio do Servidor Público - PASEP, ao titular quando ele próprio ou qualquer de seus dependentes for acometido de neoplasia maligna. Brasília: Diário Oficial da União; 1996. Disponível em: https://www.normasbra-sil.com.br/norma/resolucao-1-1996_95224.html
20. Brasil. Lei n° 8.213, de 24 de julho de 1991. Dispõe sobre os Planos de Benefícios da Previdência Social e dá outras procidências. Brasília: Diário Oficial da União; 1991. Disponível em: https://www.planalto.gov.br/ccivil_03/leis/l8213cons.htm
21. Conselho Federal de Medicina. Resolução CFM n° 1.851, de 18 de agosto de 2008. Altera o art. 3° da Resolução CFM n° 1.658, de 13 de fevereiro de 2002, que normatiza a emissão de atestados médicos e dá outras providências. Brasília: Diário Oficial da União; 2008. Disponível em: https://sistemas.cfm.org.br/nor-mas/arquivos/resolucoes/BR/2008/1851_2008.pdf
22. Brasil. Ministério da Saúde. Secretaria de Atenção à Saúde. Portaria n° 55, de 24 de fevereiro de 1999. Dispõe sobre a rotina do Tratamento Fora de Domicilio no Sistema Único de Saúde - SUS, com inclusão dos procedimentos específicos na tabela de procedimentos do Sistema de Informações Ambulatoriais do SIA/ SUS e dá outras providências. Brasília: Diário Oficial da União; 1999. Disponível em: https://bvsms.saude.gov.br/bvs/saudelegis/sas/1999/prt0055_24_02_1999.html
23. Governo do Estado do Rio de Janeiro. Lei n° 4.510, de 13 de janeiro de 2005. Dispõe sobre a isenção do pagamento de tarifas nos serviços de transporte intermunicipal de passageiros por ônibus do estado do Rio de Janeiro, para alunos do ensino fundamental e médio da rede pública estadual de ensino, para as pessoas portadoras de deficiência e portadoras de doença crônica de natureza física ou mental que exijam tratamento continuado e cuja interrupção no tratamento possa acarretar risco de vida, e dá outras providências. Rio de Janeiro: Diário Oficial do Estado do Rio de Janeiro; 2005. Disponível em: https://www.legisweb.com.br/legislacao/?id=157565
24. Brasil. Lei n° 7.713, de 22 de dezembro de 1998. Altera a legislação do imposto de renda e dá outras providências. Brasília: Diário Oficial da União; 1988. Disponível em: https://www.planalto.gov.br/ccivil_03/leis/l7713.htm
25. Brasil. Lei n° 10.182, de 12 de fevereiro de 2001. Restaura a vigência da Lei no 8.989, de 24 de fevereiro de 1995, que dispõe sobre a isenção do Imposto sobre Produtos Industrializados (IPI) na aquisição de automóveis destinados ao transporte autônomo de passageiros e ao uso de portadores de deficiência física, reduz o imposto de importação para os produtos que especifica, e dá outras providências. Brasília: Diário Oficial da União; 2001. Disponível em: https://www.planalto.gov.br/ccivil_03/leis/leis_2001/l10182.htm
26. Brasil. Lei n° 8.989, de 24 de fevereiro de 1995. Dispõe sobre isenção do Imposto sobre Produtos Industrializados (IPI) na aquisição de automóveis para utilização no transporte autônomo de passageiros, bem como por pessoas portadoras de deficiência física e aos destinados ao transporte escolar, e dá outras provi-dências. Brasília: Diário Oficial da União; 1995. Disponível em: https://www.planalto.gov.br/ccivil_03/leis/l8989.htm
27. Brasil. Lei n° 9.797, de 6 de maio de 1999. Dispõe sobre a obrigatoriedade da cirurgia plástica reparadora da mama pela rede de unidades integrantes do Sistema Único de Saúde - SUS nos casos de mutilação decorrentes de tratamento de câncer. Brasília: Diário Oficial da União; 1999. Disponível em: https://www.pla-nalto.gov.br/ccivil_03/leis/l9797.htm
28. Brasil. Lei n° 12.802, de 24 de abril de 2013. Altera a Lei n° 9.797, de 6 de maio de 1999, que “dispõe sobre a obrigatoriedade da cirurgia plástica reparadora da mama pela rede de unidades integrantes do Sistema Único de Saúde - SUS nos casos de mutilação decorrentes de tratamento de câncer”, para dispor sobre o momento da reconstrução mamária. Brasília: Diário Oficial da União; 2013. Disponível em: https://www.planalto.gov.br/ccivil_03/_ato2011-2014/2013/lei/l12802.htm
29. Brasil. Lei n° 13.770, de 19 de dezembro de 2018. Altera as Leis n ° 9.656, de 3 de junho de 1998, e 9.797, de 6 de maio de 1999, para dispor sobre a cirurgia plástica reconstrutiva da mama em casos de mutilação decorrente de tratamento de câncer. Brasília: Diário Oficial da União; 2018. Disponível em:
30. Brasil. Lei n° 15.171, de 17 de julho de 2025. Altera a Lei n° 9.797, de 6 de maio de 1999, e a Lei n° 9.656, de 3 de junho de 1998, para ampliar o direito das mulheres à cirurgia plástica reparadora da mama em casos de mutilação total ou parcial. Brasília: Diário Oficial da União; 2025. Disponível em: https://www.planalto.gov.br/ccivil_03/_ato2023-2026/2025/lei/l15171.htm
1. Professional Postgraduate Program in Applied Health Sciences, Universidade do Vale
do Sapucaí (UNIVÁS), Pouso Alegre, MG, Brazil
2. School of Medicine, Universidade do Vale do Sapucaí (UNIVÁS), Pouso Alegre, MG,
Brazil
Address for correspondence Daniela Francescato Veiga, MD, PhD, Programa de Pós-graduação Profissional em Ciências Aplicadas à Saúde, Universidade do Vale do Sapucaí, Avenida Prefeito Tuany Toledo 470, Fatima I, Pouso Alegre, MG, CEP: 04023-900, Brazil (e-mail: danielafveiga@gmail.com).
Article received: September 16, 2025.
Article accepted: December 16, 2025.
Editor-in-chief: Dov Charles Goldenberg.
Conflict of Interests
The authors have no conflict of interests to declare.









 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter