

Original Article - Year 2026 - Volume 41Issue 1
Análise ultrassonográfica da lipoenxertia na reconstrução mamária
Ultrasound Analysis of Fat Grafting in Breast Reconstruction
RESUMO
Introdução A lipoenxertia é amplamente utilizada em procedimentos cirúrgicos mamários, tanto reconstrutivos quanto estéticos, com o intuito de aprimorar os resultados e corrigir imperfeições. Ainda há incertezas quanto à taxa de reabsorção da gordura, variando conforme a técnica aplicada. A ultrassonografia temsemostrado como uma ferramenta acessível para avaliação da reabsorção do enxerto de gordura.
Objetivo Examinar a integração dos enxertos de gordura em pacientes submetidas à reconstrução mamária por meio de exames ultrassonográficos.
Materiais e Métodos Realizou-se lipoaspiração seguida da reinjeção da gordura nas mamas. As pacientes foram divididas em dois grupos: em um, a gordura foi purificada por meio de filtragem; no outro, por decantação. A avaliação foi feita através da medição da espessura do tecido subcutâneo com ultrassom.
Resultados Participaram do estudo 12 pacientes, divididas igualmente entre os 2 grupos. A média de redução na espessura do tecido subcutâneo, após 6 meses, foi de 26,36% no grupo que recebeu gordura decantada e 22,80% no grupo com gordura filtrada.
Conclusão Verificou-se reabsorção parcial dos enxertos em ambos os grupos analisados. Não houve diferença significativa na taxa de reabsorção entre os dois grupos à análise com ultrassonografia.
Palavras-chave: reconstrução da mama; lipoaspiração; tecido adiposo; coleta de tecidos e órgãos; transplante autólogo; ultrassonografia; absorção
ABSTRACT
Introduction Fat grafting is widely used in breast surgical procedures, both reconstructive and esthetic, to enhance outcomes and correct imperfections. There are still uncertainties regarding the fat graft resorption rate, which varies depending on the technique used. Ultrasound has shown to be an accessible tool for evaluating fat graft resorption.
Objective To assess the integration of fat grafts in patients undergoing breast reconstruction using ultrasound scans.
Materials and Methods Following liposuction, fat was reinjected into the breasts of the patients. They were divided into two groups; one group had the fat purified through filtration, while the other group had it purified through decantation. Evaluation consisted of measuring subcutaneous tissue thickness with ultrasound.
Results The study included 12 patients, equally divided between the 2 groups. The average reduction in subcutaneous tissue thickness after 6 months was 26.36% in the decanted fat group and 22.80% in the filtered fat group.
Conclusion Both groups presented partial graft absorption. Ultrasound analysis revealed no significant difference in the resorption rate between the two groups.
Keywords: breast reconstruction; liposuction; adipose tissue; tissue and organ harvesting; transplantation; autologous; ultrasonography; absorption
Introduction
Using autologous fat as a filler material is an established practice in plastic surgery for treating soft-tissue defects. Its first description, by Gustav Neuber, dates to 1893. In 1985, Vincent Czerny performed the 1st breast reconstruction using fat grafting.1,2
In the past, fat grafting in the breasts was considered a risky procedure due to the possibility of malignancy and difficulty in breast cancer screening. However, studies have shown that fat grafting does not compromise radiological monitoring nor is it related to an increased risk of neoplasms.3,4 Fat is an abundant, readily available, biocompatible, and autologous material with low immunogenicity, making it ideal for filling procedures. Once seen as an experimental technique, fat grafting became a safe and effective approach in post-mastectomy breast reconstruction, as it allows correcting deformities, asymmetries, and skin retractions, improving breast contour.5,6
The increase in breast cancer incidence elevated the number of women undergoing breast reconstruction and, as a result, fat grafting.7 Despite the advantages, fat grafting outcomes remain variable, particularly due to the partial reabsorption of the grafted tissue, which can compromise the aesthetic result and require additional procedures.5,8
Recent advances in literature have elucidated significant biological aspects of grafted fat, highlighting the role of vascular stromal fraction (VSF) and adipose tissue-derived stem cells (ADSCs). These cellular structures exhibit immunomodulatory, angiogenic, and regenerative properties that favor graft vascularization and integration, and contribute to its viability and durability.9
Despite the satisfactory clinical outcomes, some doubts persist, such as the ideal fat processing technique and the methods for measuring graft integration. Magnetic resonance imaging, although effective, has practical and economic limitations for this measurement. Ultrasound emerges as a viable, safe, and accessible alternative for measuring post-grafting reabsorption.10,11
Objective
The present study aimed to evaluate the integration of fat grafts in patients undergoing breast reconstruction using ultrasound analysis, comparing the reabsorption of fat prepared by filtration or decantation.
Materials and Methods
The present prospective, interventional clinical study uses qualitative and quantitative analysis of the results. Patients who had undergone mastectomy with breast reconstruction (either immediate or delayed) and presented with complaints of asymmetries amenable to correction with lipofilling at the Plastic Surgery outpatient clinic of Hospital Universitário Clementino Fraga Filho (HUCFF) in Rio de Janeiro, RJ, Brazil, were selected. The study involved patients aged 18 and older who consented to participate by signing an informed consent form. The study occurred from December 2022 to May 2023.
All participants received treatment with autologous fat grafts to correct breast asymmetries according to the anatomical particularities of each case. A plastic surgeon at HUCFF conducted surgical planning and fat grafting at the surgical center and performed ultrasound measurements at the outpatient clinic. Tissue harvesting and grafting followed the same technique for all participants; only the processing differed according to the intervention group.
The selected patients were distributed alternately between two groups of equal sample size: one received autologous adipose tissue grafts treated by decantation and the other by sieve filtration. In addition, data regarding chronic diseases, breast reconstruction type, and oncological therapies were collected.
Preoperative marking and measurement point definition
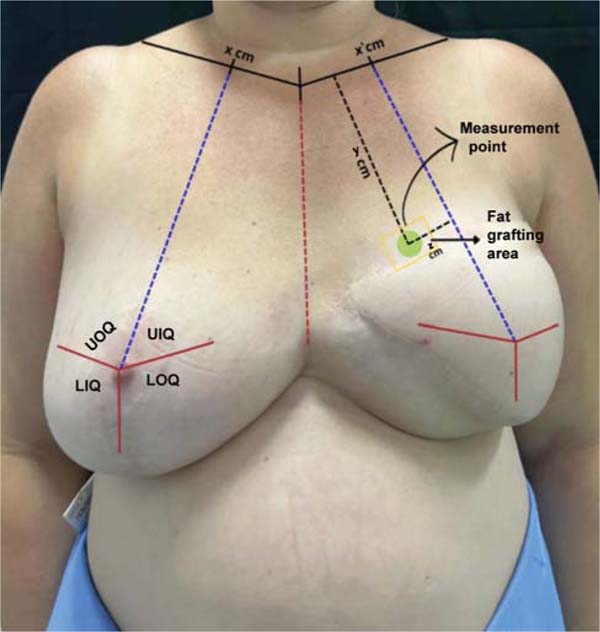
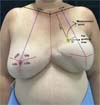
First, the non-reconstructed breast is divided into four quadrants, using the nipple as a horizontal reference and a line from the midpoint of the clavicle to the inframammary fold as the vertical one. This midpoint is also marked on the contralateral clavicle to orient the reconstructed breast. The sternal notch defines the center of the chest. Using a 20-cm compass, the point of the original nipple is transferred to the reconstructed breast, guided by the sternal line, allowing the marking of the four quadrants on the reconstructed breast.
Within the breast quadrant with perceived low volume, a fixed point is established for measuring subcutaneous thickness during follow-up. This point defines the central region of the quadrant, and the shortest distances from it to the clavicle and to the nearest division of the breast quadrant are recorded for reproduction in subsequent measurements (►Fig. 1).
Ultrasound assessment
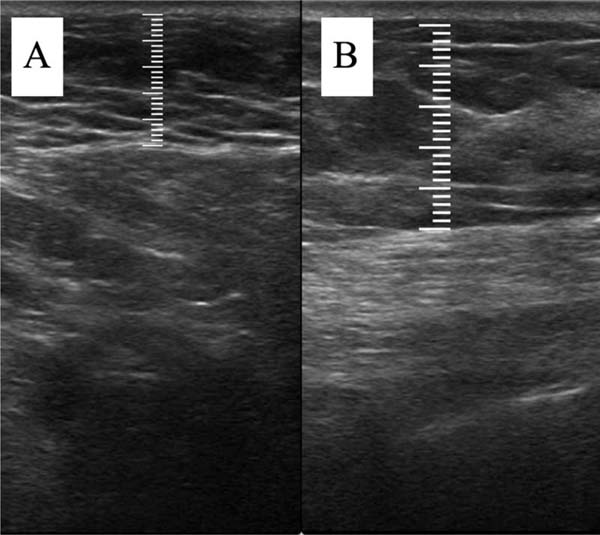
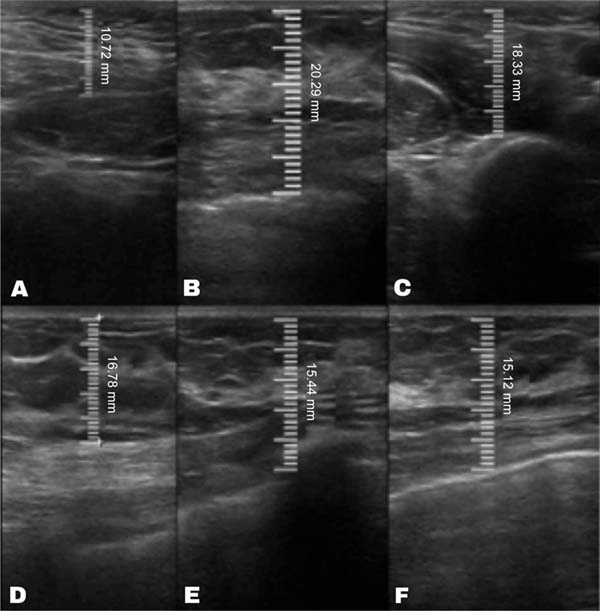
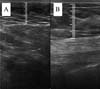
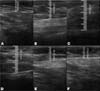
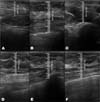
The measurement of subcutaneous tissue thickness is performed vertically, from the highest (right at the transition from the epidermis) to the lowest point (at the transition with the muscle tissue) of subcutaneous tissue thickness. This measurement is easily reproducible for those with basic ultrasound skills (►Fig. 2). In lipofilled breasts, the thickness of subcutaneous tissue was measured preoperatively and immediately after lipofilling, as well as at 15, 30, 90, and 180 days postoperatively. Measurements in millimeters were taken using ultrasound, including the vertical distance from the highest (closest to the epidermis) to the lowest point (closest to the adjacent musculature) of the subcutaneous tissue at the previously defined point. A GE LOGIQ P6 (GE HealthCare) ultrasound device with an 11L linear probe and a frequency range of 12 to 6 MHz was used, operated by a surgeon with postgraduate training in general ultrasonography (►Fig. 3).
Adipose tissue harvesting, preparation, and grafting
Adipose tissue was harvested from the abdominal region via a periumbilical incision preceded by local anesthesia with 2% lidocaine. Fat harvesting used 3-mm diameter and 200-mm long cannulas, along with 2-mL syringes, following subcutaneous infiltration of a saline solution containing epinephrine at a 1:500 thousand ratio. Next, adipose tissue treatment occurred according to the assigned group.
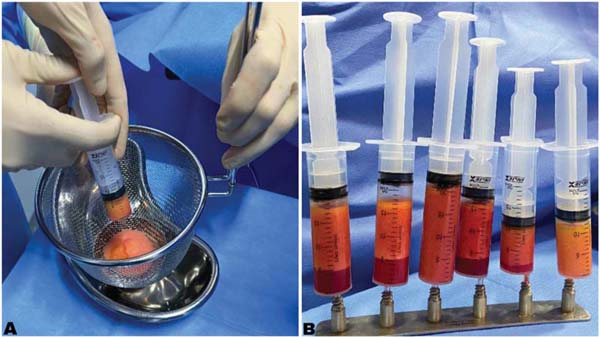
In the filtration group, adipose tissue was processed through a 16-cm stainless steel sieve. In the decantation group, the tissue was placed in vertical syringes and allowed to sit for 20 minutes. This process relied on gravity to separate adipose tissue components, that is, solids (fat) and liquids (oil, serum, saline solution). Next, the serum and oil separated from the fat were discarded, leaving only the fat ready for transplant.
The filtered or decanted fat was transferred to 3-mL or 10-mL syringes using 2-way stainless steel transfer devices. Next, fat was infiltrated into the breast through a small incision with simple single-hole cannulas (2 mm in diameter and 200 mm in length), establishing tunnels for fractional retroinjection into the subcutaneous tissue of the areas requiring correction (►Fig. 4).
Data analysis
Data analysis used Microsoft Excel (Microsoft Corp.) spreadsheets. Statistical correlations, including Chi-squared and odds ratio tests, with a 95%CI, were performed using the IBM SPSS Statistics for Windows software (IBM Corp.), version 23.0.
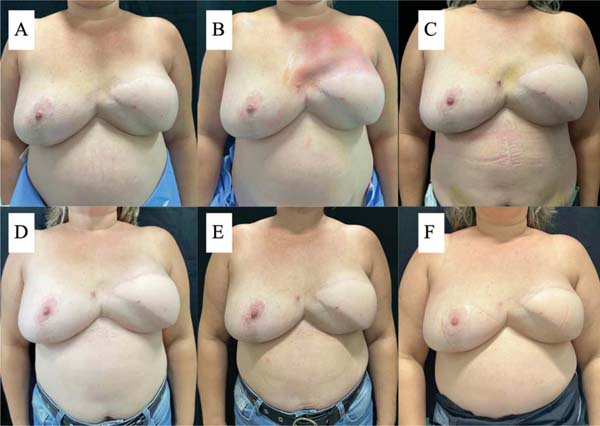
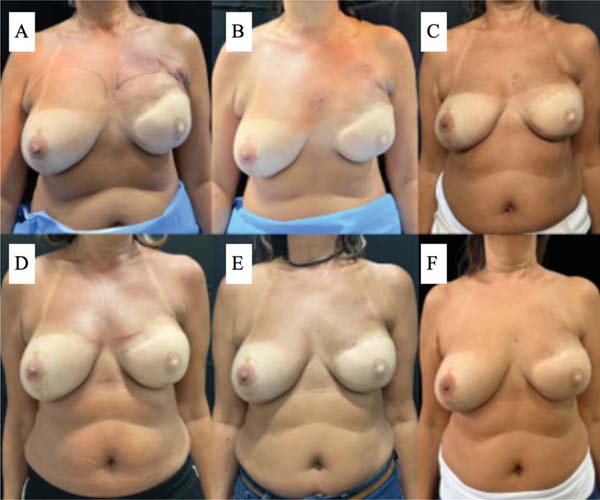
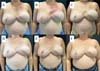
Standardized photographs were taken whenever patients underwent an ultrasound for visual monitoring. Images were obtained against a black background in anteroposterior, oblique (right and left), and lateral (right and left) views at a distance of 1.5 m using an iPhone 15 Pro (Apple, Inc.) camera.
All patients were discharged on the same day after surgery with a prescription for oral cephalosporin for 7 days and non-steroidal anti-inflammatory drugs. Patients were also given instructions to avoid applying pressure to the grafted breast.
The Research Ethics Committee of HUCFF approved the study in accordance with the ethical precepts for research in human subjects (CAAE 62113922.3.0000.5257).
Results
Twelve patients participated in the study, with half allocated to each of the two intervention groups. Among them, 8 under-went immediate reconstruction (66.7%), and 4 underwent delayed reconstruction (33.3%). Regarding the type of reconstruction, 7 used breast implants (58.3%) and five, myocutaneous flaps (41.7%). Regarding complications, there was 1 case of infection (8.3%) and 1 oil cyst (8.3%). In the decanted fat group, the average age of the participants was 53 years old; 2 subjects had systemic arterial hypertension (16.7%), 1 had diabetes mellitus (8.3%), and all underwent neoadjuvant chemotherapy and adjuvant radiotherapy (100%). In the filtered fat group, the average age was 55 years old; 2 subjects had systemic arterial hypertension (16.7%), 2 had diabetes mellitus (16.7%), and all underwent neoadjuvant chemotherapy and adjuvant radiotherapy (100%).
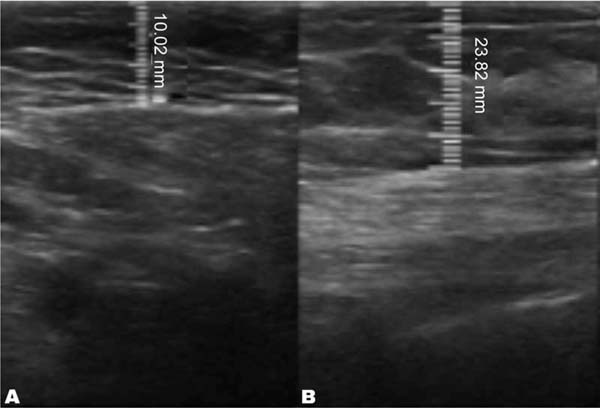
The average volume of fat grafted was 116.7 mL in the decanted graft group and 123.3 mL in the filtered graft group. The average initial thickness of the subcutaneous tissue was approximately 11 mm in both groups. This value increased to 22 mm in the immediate postoperative period in the decanted group and 23 mm in the filtered group.
Both groups presented progressive reabsorption. ►Table 1 and ►Fig. 5 show the change in mean subcutaneous thickness in both groups.
| Operative period | Decanted graft group (n=6) | Filtered graft group (n=6) |
|---|---|---|
| Preoperative | 11.27 | 11.23 |
| Immediate postoperative | 22.42 | 23.90 |
| 15 days after surgery | 20.20 | 22.05 |
| 1 month after surgery | 18.10 | 20.35 |
| 3 months after surgery | 17.13 | 18.96 |
| 6 months after surgery | 16.51 | 18.46 |
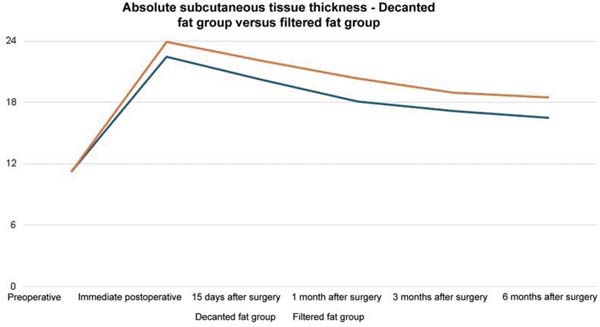
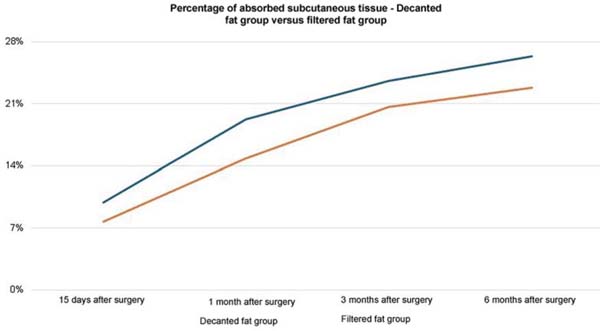
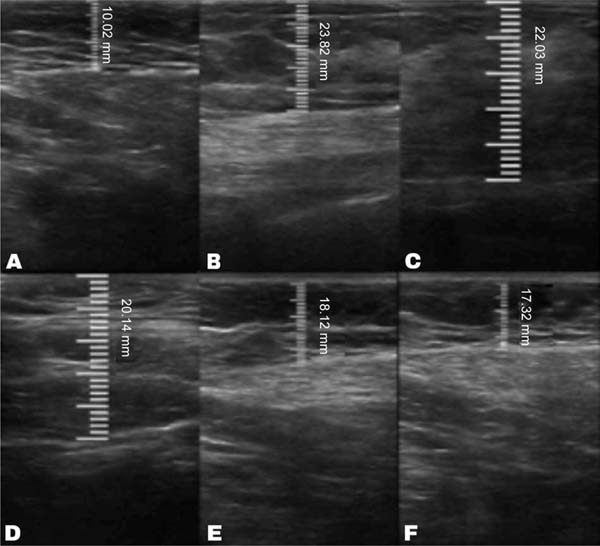
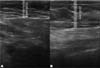
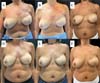
In the decanted graft group, the average reduction compared with the initial thickness after lipofilling was 9.90% on day 15, 19.27% on month 1, 23.59% on month 3, and 26.36% on month 6. In the filtered graft group, the average reduction compared to the initial thickness after lipofilling was 7.74% on day 15, 14.85% on month 1, 20.67% on month 3, and 22.80% on month 6 (►Fig. 6). ►Figs 7–8 reveal the ultrasound change in subcutaneous tissue’s thickness before and after lipofilling. ►Figs. 9–10 show the pre-and postoperative results of one patient from each group.
The comparison of subcutaneous thickness reduction at 6 months between the graft decantation and filtration techniques showed no statistically significant difference (p ¼ 0.59).
Discussion
Ultrasound has proven useful for accurately measuring subcutaneous thickness over time, allowing for an objective assessment of the absorption rate and detection of complications, such as necrosis or infection, which were not observed in the present study. Among radiological methods, the greater availability of ultrasound compared to magnetic resonance imaging and the lack of radiation exposure are key advantages. Other forms of analysis, such as photographic records, would be more susceptible to subjective interpretations.12,13
The fat preparation techniques resulted in no statistically significant difference between the groups. Reabsorption was similar, and the patients’ clinical factors did not significantly influence the outcomes.
These findings are consistent with existing literature, which indicates that the lipofilling technique, regardless of the fat preparation method, exhibits similar behavior in terms of graft absorption. Therefore, the choice between filtration and decantation can be based on the surgeon’s preferences, practicality, and resource availability, without compromising the outcome.13,14
Furthermore, it is important to consider that fat grafting has not only a volumetric role, but also a biological one. The presence of mesenchymal stem cells in adipose tissue can contribute to improving skin quality and tissue regeneration, especially in areas subjected to radiotherapy. The trophic effect of fat may even justify the repeated use of the technique to achieve more lasting and esthetically satisfactory outcomes.5,15
Another relevant aspect is the psychological impact on patients. The aesthetic improvement of reconstructed breasts contributes to the self-esteem and quality of life of women who had cancer. Lipofilling, as a less complex procedure with good results, represents an important tool in the physical and emotional rehabilitation process.15
Furthermore, the lack of complications noted in the present study highlights the safety of the technique even in patients with a history of radiotherapy, which is typically associated with a higher risk of necrosis or infection. This piece of data has major clinical relevance since many surgeons remain apprehensive when performing grafts on previously irradiated tissues.16,17
It is worth highlighting that quantifying the amount of fat retained over time is essential for planning future procedures. Due to the tendency for partial reabsorption, the surgeon must consider potential additional sessions to achieve the desired aesthetic outcome. Understanding the average absorption rate improves treatment planning and aligns patient expectations.5,10
Few large-sample longitudinal studies correlate variables such as age, body mass index, comorbidities, and tissue response to grafting. Exploring these relationships can help clarify which patient profiles exhibit greater or lesser absorption, refining the criteria for treatment indication and individualization.9
However, a robust validation of these findings would require studies with larger samples, randomized methodology, and long-term evaluation. Standardization of preparation, grafting, and measurement techniques is also fundamental to allow consistent comparisons between different studies and populations.17
Enriching grafts with SVF, ADSCs, and platelet-rich plasma has been studied as a promising strategy to optimize lipofilling outcomes. Nevertheless, standardization of these approaches in clinical practice requires more data.9,15,18 The most significant limitation of the present study is the small sample size, totaling 12 women who underwent breast lipofilling. Although the results obtained are encouraging and reinforce the safety and efficacy of the technique in the analyzed context, the restricted sample size compromises the statistical robustness of the findings, limiting their generalization to the female population. Additionally, including patients from a single center with specific eligibility criteria may introduce selection bias, which does not comprehensively reflect the diversity of clinical cases encountered in daily practice. Therefore, caution is recommended when extrapolating the results presented. Future studies with larger, multicenter samples and longer longitudinal follow-up are needed to validate the findings of this research and provide more consistent evidence regarding the applicability and durability of the results of breast lipofilling.
Conclusion
The volumes of grafted adipose tissue were partially absorbed over time in both processing techniques, with an average reabsorption rate of 25% after 6 months. There was no significant difference between the groups (filtered and decanted graft) regarding graft reabsorption.
REFERENCES
1. Mazzola RF, Mazzola IC. The fascinating history of fat grafting. J Craniofac Surg 2013;24(04):1069-1071. Doi: 10.1097/SCS.0b013e318292c447
2. Del Vecchio D, Fichadi H. Autologous Fat Transplantation - A Paradigm Shift in Breast Reconstruction. In: Salgarello M, editora. Breast Reconstruction - Current Techniques. Londres: InTechOpen; 2012. Disponível em: https://www.intechopen.com/chapters/27951
3. Petit JY, Botteri E, Lohsiriwat V, et al. Locoregional recurrence risk after lipofilling in breast cancer patients. Ann Oncol 2012;23(03): 582-588. Doi: 10.1093/annonc/mdr158
4. Delay E, Garson S, Tousson G, Sinna R. Fat injection to the breast: technique, results, and indications based on 880 procedures over 10 years. Aesthet Surg J 2009;29(05):360-376. Doi: 10.1016/j.asj.2009.08.01
5. Al Qurashi AA, Shah Mardan QNM, Alzahrani IA, et al. Efficacy of Exclusive Fat Grafting for Breast Reconstruction: An Updated Systematic Review and Meta-analysis. Aesthetic Plast Surg 2024;48(23):4979-4985. Doi: 10.1007/s00266-024-03978-3
6. Sarrami SM, Humar P, Marallo MJ, Roy E, Mehta M, De La Cruz C. High-Volume Fat Grafting With and Without Goldilocks Mastectomies Show Successful Clinical Outcomes for Breast Reconstruction. Ann Plast Surg 2025;94(4S, Suppl 2)S233-S237. Doi: 10.1097/SAP.0000000000004212
7. Instituto Nacional de Câncer (INCA). Estatísticas de câncer. Rio de Janeiro: INCA; 2022. Disponível em: https://www.gov.br/inca/pt-br/assuntos/cancer/numeros/
8. Dayal A, Bhatia A, Hsu JT. Fat grafting in aesthetics. Clin Dermatol 2021 ;40(01 ):35-44 Doi: 10.1016/j.clindermatol.2021.08.010 PubMed
9. Strong AL, Cederna PS, Rubin JP, Coleman SR, Levi B. The Current State of Fat Grafting: A Review of Harvesting, Processing, and InjectionTechniques. Plast Reconstr Surg 2015;136(04):897-912. Doi: 10.1097/PRS.0000000000001590
10. Kanchwala SK, Glatt BS, Conant EF, Bucky LP. Autologous fat grafting to the reconstructed breast: the management of acquired contour deformities. Plast Reconstr Surg 2009;124(02):409-418. Doi: 10.1097/PRS.0b013e3181aeeadd
11. Zocchi ML, Zuliani F. Bicompartmental breast lipostructuring. Aesthetic Plast Surg 2008;32(02):313-328. Doi: 10.1007/s00266-007-9089-3
12. Fraga MFP, Perin LF, Helene A Júnior, Aching NC. Volumetric assessment of breasts in patients undergoing fat grafting with pre-operative expansion (BRAVA®): experience of Santa Casa de São Paulo. Revista Brasileira de Cirurgia Plástica (RBCP). Brazilian Journal of Plastic Sugery [Internet] 2017;32(01):64-71. Doi: 10.5935/2177-1235.2017RBCP0009
13. Veber M, Tourasse C, Toussoun G, Moutran M, Mojallal A, Delay E. Radiographic findings after breast augmentation by autologous fat transfer. Plast Reconstr Surg 2011;127(03):1289-1299. Doi: 10.1097/PRS.0b013e318205f38f
14. Meruane MA, Rojas M, Marcelain K. The use of adipose tissue-derived stem cells within a dermal substitute improves skin regeneration by increasing neoangiogenesis and collagen synthesis. Plast Reconstr Surg 2012;130(01):53-63. Doi: 10.1097/PRS.0b013e3182547e04
15. Muntean MV, Pop IC, Ilies RA, Pelleter A, Vlad IC, Achimas-Cadariu P. Exploring the Role of Autologous Fat Grafting in Implant-Based Breast Reconstruction: A Systematic Review of Complications and Aesthetic Results. J Clin Med 2025;14(12):4073. Doi: 10.3390/jcm14124073
16. Papadopoulos S, Vidovic G, Tio J, et al. Fat grafting in breast surgery: a retrospective single-breast centre 6-year experience. Arch Gynecol Obstet 2024;310(05):2587-2594. Doi: 10.1007/s00404-024-07708-7
17. Gentile P, Cervelli V. Systematic review: Oncological safety of reconstruction with fat grafting in breast cancer outcomes. J Plast Reconstr Aesthet Surg 2022;75(11):4160-4168. Doi: 10.1016/j.bjps.2022.08.026
18. Debuc B, Gendron N, Cras A, et al. Improving Autologous Fat Grafting in Regenerative Surgery through Stem Cell-Assisted Lipotransfer. Stem Cell Rev Rep 2023;19(06):1726-1754. Doi: 10.1007/s12015-023-10568-4
1. Department of Plastic Surgery, Hospital Universitário Clementino Fraga Filho, Universidade
Federal do Rio de Janeiro (UFRJ), Rio de Janeiro, RJ, Brazil
2. Universidade Federal do Rio de Janeiro (UFRJ), Rio de Janeiro, RJ, Brazil
3. Afya Universidade Unigranrio, Rio de Janeiro, RJ, Brazil
4. Faculdade de Medicina de Petrópolis, Petrópolis, RJ, Brazil
5. Pontifícia Universidade Católica do Rio de Janeiro (PUC-Rio), Rio de Janeiro, RJ,
Brazil
Data Availability
Financial Support The authors declare that they did not receive financial support from agencies in the public, private or non-profit sectors to conduct the present study.
Address for correspondence Marcos Louro, PhD, Departmento de Cirurgia Plástica, Hospital Universitário Clementino Fraga Filho,, Universidade Federal do Rio de Janeiro (UFRJ), Rio de Janeiro, Rio de Janeiro, Brazil (e-mail: drmarcoslouro@gmail.com).
Article received: July 29, 2025.
Article accepted: December 16, 2025.
Conflict of Interests The authors have no conflict of interests to declare.
Editor-in-Chief: Dov Charles Goldenberg.







 Read in Portuguese
Read in Portuguese
 Read in English
Read in English
 PDF PT
PDF PT
 Print
Print
 Send this article by email
Send this article by email
 How to Cite
How to Cite
 Mendeley
Mendeley
 Pocket
Pocket
 Twitter
Twitter